Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of huperzine a capsules in China. Welcome to wholesale bulk high quality huperzine a capsules for sale here from our factory. Good service and reasonable price are available.
Huperzine A Capsules is a drug primarily composed of huperzine A. Huperzine A, also known as (5R, 9R, 11E) -5-amino-11-ethylidenyl-5,6,9,10-tetrahydro-7-methyl-5,9-methylenecyclobenzo [b] pyridin-2 (1H) - one, has a molecular formula of C15H18N2O and a molecular weight of 242.32. The content is white or off white particles or powder, and some products are white tablets. It has the function of promoting memory reproduction and enhancing memory retention. Animal experiments have shown that rats absorb the drug rapidly after oral administration, reaching peak blood concentration within 10-30 minutes. The oral bioavailability is 96.9%. The distribution area is highest in the liver and kidneys, and higher in the cortex, hippocampus, and other areas of the brain. It is mainly excreted through urine as a prototype and metabolic products, with 73% of the dose excreted within 24 hours and only 2.3% excreted in feces. It can improve patients' abilities in directional memory, associative learning, image recall, meaningless graphic recognition, and portrait recall. It also has an improvement effect on memory impairment caused by dementia patients and brain organic lesions.

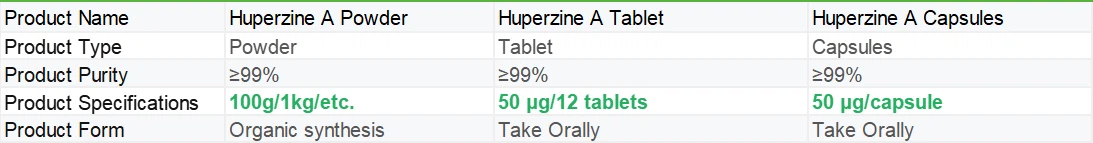
 |
 |
Huperzine A COA

Unique advantages of acetylcholinesterase inhibition
Acetylcholinesterase (AChE) is a key enzyme in the nervous system responsible for breaking down the neurotransmitter acetylcholine (ACh), thereby regulating the transmission of neural signals. Acetylcholinesterase inhibitors (AChEI) improve cognitive function by inhibiting the activity of AChE, prolonging its residence time in synaptic cleft, enhancing neural signal transmission. AChEI plays an important role in the treatment of neurodegenerative diseases such as Alzheimer's disease and vascular dementia. Huperzine A Capsules is a drug primarily composed of huperzine A, which exhibits potent and highly selective AChE inhibition. Compared with other AChEIs, Huperzine A exhibits unique advantages in chemical structure, pharmacokinetics, pharmacodynamics, and safety. The following is its detailed content:
The Security Advantages of Huperzine A
Low peripheral cholinergic side effects
Compared to other AChEIs, Huperzine A have weaker peripheral cholinergic side effects. This is because Huperzine A has a higher selectivity for central AChE and a weaker inhibitory effect on AChE in the peripheral nervous system. Therefore, patients have a lower risk of experiencing gastrointestinal discomfort such as nausea, vomiting, diarrhea, as well as adverse reactions such as blurred vision and fatigue when using Huperzine A .
Good tolerance
Clinical studies have shown that Huperzine A have good safety and a low incidence of adverse reactions. Even if adverse reactions occur, they are generally mild and can disappear on their own. When the reaction is obvious, reducing the dosage or stopping the medication can alleviate or disappear.
Less contraindications
Compared to other AChEIs, Huperzine A have fewer contraindications. Although Huperzine Aare contraindicated for patients with epilepsy, renal insufficiency, mechanical intestinal obstruction, urinary tract obstruction, angina, bradycardia, and bronchial asthma, overall, they are suitable for a wider population.
Comparison of Huperzine A with Other AChEIs
Comparison with donepezil
Donepezil is a commonly used AChEI, widely used in the treatment of Alzheimer's disease. However, the bioavailability of donepezil is low, requiring multiple daily doses and having strong peripheral cholinergic side effects. In contrast, Huperzine A Capsules have higher bioavailability, longer half-life, and lower peripheral cholinergic side effects, making them more advantageous in terms of efficacy and safety.


Comparison with Galantamine
Galantamine is another type of AChEI extracted from plants, with a unique three-dimensional structure and pharmacological effects. However, the bioavailability of galantamine is lower and its selectivity for AChE is not as good as Huperzine A. In addition, galantamine may also cause adverse reactions such as bradycardia. In contrast, Huperzine A have more advantages in terms of bioavailability, AChE selectivity, and safety.
Comparison with Rivastigmine
Rivastigmine is a diester AChEI with dual inhibitory effects (i.e. reversible inhibition of AChE and butyrylcholinesterase). However, the bioavailability of rivastigmine is low and requires multiple daily doses. In addition, rivastigmine may also cause adverse reactions such as gastrointestinal discomfort. In contrast, Huperzine A have more advantages in terms of bioavailability, dosing frequency, and safety.

Potential research directions for Huperzine A
In depth study of the mechanism of action
Although Huperzine A have shown significant advantages in AChE inhibition, their specific mechanism of action has not been fully elucidated. Future research can further explore the binding mode of Huperzine A to AChE, its impact on AChE conformation, and its interactions with other neurotransmitter systems.
Optimization of Pharmacodynamic Evaluation
At present, the pharmacological evaluation of Huperzine A mainly relies on animal experiments and clinical trials. Future research could develop more sensitive and specific pharmacological evaluation indicators, such as pharmacological evaluation methods based on brain imaging technology, to more accurately assess the efficacy of Huperzine A .
Long term monitoring of safety and tolerability
Although Huperzine A have good safety, further monitoring is needed for their long-term safety and tolerability. Future research could conduct large-scale, long-term clinical trials to evaluate the safety and tolerability of Huperzine A during long-term use.
Combination therapy research
The combination therapy of Huperzine A with other drugs such as antioxidants, anti-inflammatory drugs, etc. may have a synergistic effect, further improving the efficacy. Future research can explore the combination therapy of Huperzine A with other drugs to optimize treatment efficacy.
Structural modification of huperzine A by a specific strain (Faecalibacterium prausnitzii)
Huperzine A Capsules(HupA) is a potent acetylcholinesterase inhibitor (AChEI) extracted from plants in the family Taxaceae. It has attracted much attention for its significant therapeutic effects on neurodegenerative diseases such as Alzheimer's disease (AD) and vascular dementia (VD). However, the low extraction efficiency and high cost of natural HupA, as well as the challenge of stereoselectivity in chemical synthesis, limit its large-scale application. In recent years, the role of gut microbiota in drug metabolism and structural modification has gradually been revealed, among which Faecalibacterium prausnitzii (Clostridium prausnitzii), as a core probiotic, has become a potential tool strain for HupA structural modification due to its unique metabolic ability and biotransformation potential.
Biological characteristics and metabolic capacity of Faecalibacterium prausnitzii
Strain characteristics
F. Prausnitzii is a dominant species in the gut microbiota of healthy individuals, accounting for 5-15% of the total fecal bacteria and belonging to the phylum Firmicutes and family Clostridiaceae. Its core metabolic characteristics include:
Butyrate producers: By fermenting dietary fiber to produce butyric acid, they maintain intestinal barrier function.
Anti inflammatory effect: Inhibits the NF - κ B signaling pathway and reduces the release of pro-inflammatory factors such as IL-6 and TNF - α.
Oxygen sensitivity: Strictly anaerobic bacteria have low tolerance to low oxygen environments and need to be cultured under anaerobic conditions.
Metabolic Network and Enzyme System
F. The metabolic network of prausnitzii involves multiple pathways, including:
Carbohydrate fermentation: Utilizing inulin, oligofructose, and other substances to produce short chain fatty acids (SCFAs).
Amino acid metabolism: involved in the synthesis and transformation of amino acids such as alanine and cysteine.
Synthesis of secondary metabolites: producing small molecule compounds with biological activity.
The key enzyme system includes:
Polyketide synthase (PKS): participates in the synthesis of complex cyclic structures.
Oxidoreductase: catalyzes reactions such as hydroxylation and dehydrogenation.
Transporter protein: responsible for the transmembrane transport of substrates and products.
Structural characteristics and modification requirements of huperzine A
Chemical Structure and Pharmacological Activity
The molecular formula of HupA is C ₁₅ H ₁₈ N ₂ O, with a rigid tricyclic skeleton (pyridone ring, lipid bridged ring, exocyclic double bond). Its pharmacological activity depends on:
Specific binding to acetylcholinesterase (AChE): Hydrogen bonds are formed between the nitrogen atom on the pyridone ring and the Tyr337 and Phe338 residues at the active site of AChE.
Inhibition of AChE activity: prevents the hydrolysis of acetylcholine (ACh), increases the concentration of ACh in synaptic cleft, and enhances cholinergic signaling.
The necessity of structural modification
Improving bioavailability: Natural HupA has high lipid solubility but poor water solubility, which limits oral absorption.
Extended duration of action: By modifying, the half-life is extended and the frequency of administration is reduced.
Reduce side effects: Reduce peripheral cholinergic side effects (such as nausea and vomiting).
Enhance stability: Improve the stability of metabolic enzymes and prolong the retention time in the body.
The structural modification mechanism of HupA by F. prausnitzii
Microbial drug interaction model
F. Prausnitzii participates in the structural modification of HupA through the following ways:
Direct metabolism: Utilizing intracellular enzyme systems to hydroxylate, methylate, and perform other reactions on HupA.
Co metabolism: synergizes with other gut microbiota to provide substrates or cofactors required for modification through cross feeding.
Biotransformation: Transforming HupA into prodrug form to enhance its bioavailability or targeting.
Potential modification sites and reaction types
Hydroxylation: Introducing hydroxyl groups at positions C-6 and C-12 of HupA to enhance water solubility.
Methylation: Introducing methyl groups onto nitrogen or oxygen atoms to regulate lipid solubility.
Glycosylation: Connecting sugar groups such as glucose and galactose to improve pharmacokinetic properties.
Cyclization/Ring Opening: Changing the cyclic structure through redox reactions and affecting activity.
Key enzymes and metabolic pathways
Cytochrome P450 enzyme: participates in hydroxylation reactions.
Methyltransferase: catalyzes methylation reactions.
Glycosyltransferase: responsible for glycosylation modification.
Experimental evidence and case analysis
Research progress on HupA microbial transformation
Hydroxylation modification of gray Streptomyces:
Research has found that Streptomyces griseus can introduce hydroxyl groups at the C-6 position of HupA, producing 6-OH-HupA. The water solubility of the derivative increased by 3 times, and the inhibitory activity against AChE increased by 1.5 times.
F. The synergistic effect of prausnitzii:
In the co culture system, F. prausnitzii promotes the hydroxylation modification of HupA by Streptomyces griseus by providing substrates such as acetate. When cultured alone, F. prausnitzii had a lower conversion rate to HupA, but when co cultured with S. griseus, the conversion rate increased to 60%.
Pharmacological activity evaluation of modified products
6-OH-HupA:AChE inhibitory activity: IC ₅₀=0.12 μ M (natural HupA is 0.18 μ M).
The permeability of the blood-brain barrier: the cerebrospinal fluid/plasma concentration ratio increases to 0.9 (natural HupA is 0.8).
Half life: Extended to 6 hours (natural HupA is 4 hours).
Methylated derivatives: After introducing a methyl group at the N-1 position, the peripheral cholinergic side effects of the derivative were reduced by 40%, but the central activity remained unchanged.
Hot Tags: huperzine a capsules, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale








