Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of trilostane capsule in China. Welcome to wholesale bulk high quality trilostane capsule for sale here from our factory. Good service and reasonable price are available.
Trilostane capsules are one of the commonly used dosage forms in clinical practice, typically with specifications such as trilostane 60mg, to meet the treatment needs of different patients. Capsule dosage form is convenient for patients to take orally, with accurate dosage, and suitable for long-term medication management. In addition, there are other dosage forms such as tablets, but capsule dosage forms may have specific advantages in drug release and absorption.
Chemical Structure and Physical and Chemical Properties:
The appearance of pure Qulostan is usually white to brown powder or yellow brown crystals. Under different preparation processes and storage conditions, there may be certain differences in its appearance. For example, high-purity troxostat may be closer to a white powder, while troxostat containing certain impurities or stored under specific conditions may appear as brown or yellow brown crystals. Poor water solubility, almost insoluble in water; Slightly soluble in dimethylformamide; Extremely soluble in methanol and ethanol; Insoluble in diethyl ether and chloroform. The poor water solubility of troxostat poses certain challenges to the preparation of its formulations, as many drug formulations require administration in solution form. In order to improve the solubility and bioavailability of troxostat, researchers usually use formulation techniques such as making liposomes, nanoparticles, cyclodextrin inclusion complexes, or selecting appropriate solvents and excipients to prepare formulations.
Our product
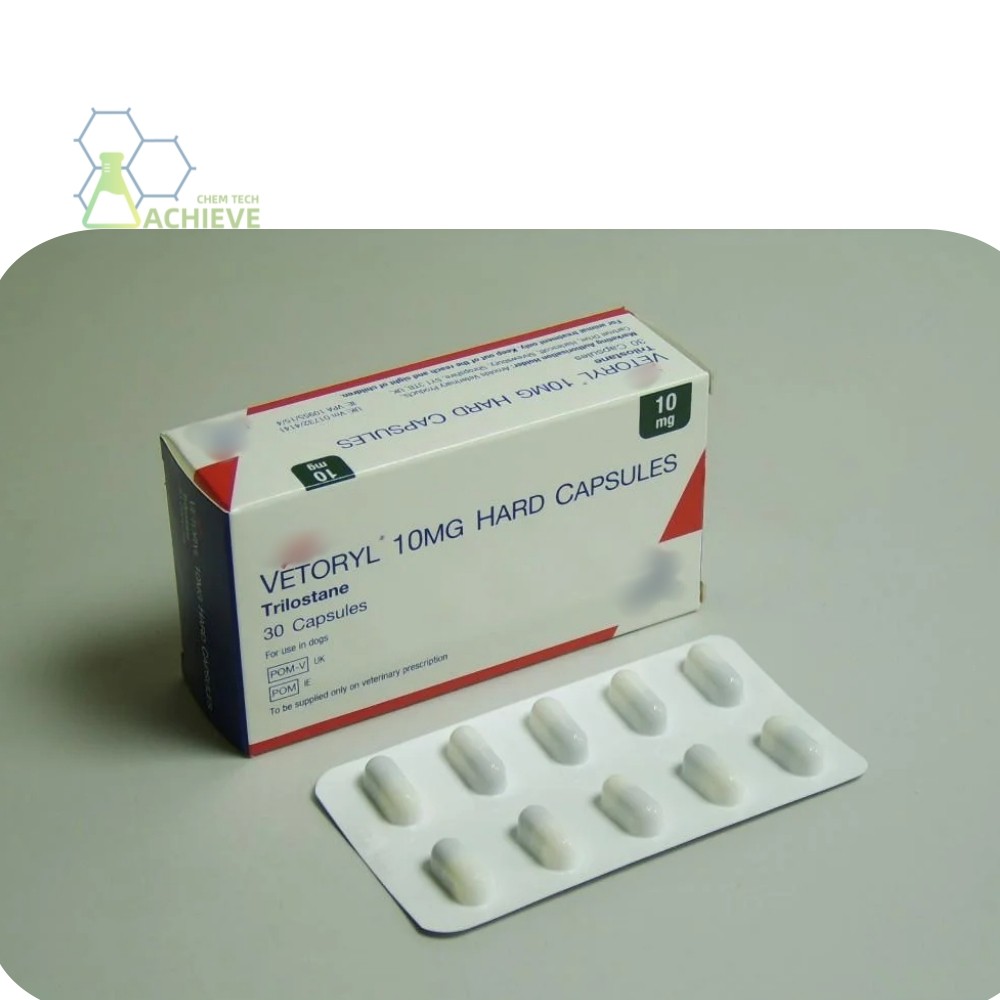



Additional information of chemical compound:

|
|
|



Clinical application and efficacy
Treatment of respiratory system diseases
Trilostane can improve the respiratory status of patients with chronic obstructive pulmonary disease (COPD) by dilating airway smooth muscle and reducing airway spasms. It can help patients reduce the feeling of shortness of breath and difficulty breathing, and also has a certain effect on alleviating acute attacks in patients with chronic obstructive pulmonary disease. However, troxostat is not suitable for treating patients with asthma or acute heart failure.
Other Potential Applications
The anti-inflammatory, immunomodulatory, hypotensive, diuretic, cholesterol lowering, triglyceride lowering, and blood glucose lowering effects of trilostane make it potentially valuable for the treatment of various diseases. For example, it can be used to treat respiratory dysfunction caused by chronic bronchitis and emphysema, as well as regulate blood lipid metabolism and blood glucose levels. However, further clinical studies are needed to validate the efficacy and safety of these applications.


Introduction to the production of trilostane capsule
Raw material preparation
types of CNC machining
Lorem ipsum dolor sit amet consectetur adipisicing elit.
Main raw materials
The main active ingredient of Quluositan capsuls is Quluositan, chemically named 4 α, 5 α - epoxy-2-cyano-androst-2-ene-3,17 β - diol, with a molecular formula of C20H27NO3 and a molecular weight of 329.43. The preparation of troxostat usually requires multiple chemical reactions, starting from specific steroid compounds and synthesizing them through a series of steps such as hydroxyl protection, oxidation reaction, cyclization reaction, deprotection reaction, etc. In the production process, the quality requirements for the raw material trilostane are extremely high, and its purity, content, and impurity level need to be strictly controlled to ensure the quality and efficacy of the final product.
Accessory selection
Accessories play an important role in capsul formulations, as they can affect the solubility, stability, release rate, and capsul formability of drugs. The commonly used excipients for Quluositan capsuls include fillers, disintegrants, lubricants, and capsul shell materials.
Fillers:
Commonly used fillers include starch, microcrystalline cellulose, etc. Starch has good compression molding and moisture absorption properties, which can increase the volume of capsuls and ensure accurate drug dosage. Microcrystalline cellulose has good fluidity and compressibility, which helps to improve the uniformity of capsul contents.
Disinfectant:
The function of a disintegrant is to rapidly disintegrate the capsul in the gastrointestinal tract, releasing the drug. Common disintegrants include carboxymethyl starch sodium and cross-linked polyvinylpyrrolidone. These disintegrants can rapidly expand after absorbing water, breaking the structure of the capsul shell and promoting drug dissolution.
Lubricant:
Lubricants can reduce the friction between materials and molds, ensuring smooth capsul filling and tablet pressing processes. Common lubricants include magnesium stearate, talcum powder, etc.
Capsule shell material:
Capsul shells are usually made of gelatin, which has good film-forming and plasticity properties and can protect drugs from external environmental influences. In addition, some auxiliary materials such as plasticizers, light blocking agents, etc. can be added as needed to improve the performance of the capsul shell.
Production process flow
Raw material pretreatment
Before production, it is necessary to perform pre-treatment on the raw material, including crushing, sieving, etc., to ensure that the particle size of the raw material meets the requirements. Crushing can make the raw material particles smaller, increase their specific surface area, and facilitate the dissolution and absorption of drugs. Screening can remove large particles and impurities from the raw materials, ensuring their uniformity.
Mixed
Mix the pre treated trilostane with various excipients in a certain proportion. The purpose of mixing is to evenly distribute the drug and excipients, ensuring consistent drug content in each capsul. Common mixing equipment includes V-type mixer, 3D motion mixer, etc. During the mixing process, it is necessary to control parameters such as mixing time and speed to ensure the mixing effect.
Granulation (optional)
Depending on the nature of the drug and the formulation requirements, granulation operations may sometimes be necessary. Granulation can turn powdered drugs and excipients into granules, improving the flowability and compressibility of materials. The commonly used granulation methods include wet granulation and dry granulation. Wet granulation is the process of mixing drugs and excipients with a binder solution to produce soft materials, which are then passed through a sieve to form granules. Dry granulation is the process of directly pressing drugs and excipients into thin sheets under high pressure, and then crushing them into granules.
Fill in
Fill the uniformly mixed or granulated material into the capsul shell. The filling process requires the use of a capsul filling machine, which can accurately control the filling amount and ensure that the drug content in each capsul meets the regulations. During the filling process, it is also important to avoid issues such as material leakage and damage to the capsul shell.
Polishing
The surface of the filled capsul may be coated with some powder or impurities, and needs to be polished to make the surface of the capsule smooth and tidy. Polishing usually uses a polishing machine to remove stains on the surface of capsuls through friction and collision.
Quality inspection
The produced Tromestane capsuls need to undergo strict quality testing to ensure that the product meets quality standards. The quality inspection items include appearance, uniformity of content, solubility, microbial limits, etc.
Appearance inspection: Check whether the shape, size, and color of the capsul are consistent, whether the surface is smooth, and whether there is any damage, deformation, or other conditions.
Content uniformity detection: High performance liquid chromatography and other methods are used to determine the content of trilostane in each capsul, calculate the content uniformity, and ensure that the drug content in each capsul is within the specified range.
Dissolution detection: Simulate the human gastrointestinal environment, measure the dissolution rate and amount of trilostane in capsuls to evaluate the drug release performance.
Microbial limit testing: Detecting the number of microorganisms in the capsul to ensure that the product meets the microbial limit standards and ensures medication safety.
Packaging
Trilostane capsule that have passed quality inspection need to be packaged. The packaging materials usually use aluminum-plastic blister packaging or bottled packaging. Aluminum plastic blister packaging has good sealing and moisture resistance, which can protect drugs from external environmental influences. Bottled packaging is convenient for storage and carrying. During the packaging process, it is necessary to label the product's name, specifications, batch number, expiration date, and other information.
Production equipment
Common crushing equipment includes universal crushers, airflow crushers, etc. The universal crusher is suitable for crushing materials with moderate hardness or below, and has the advantages of simple structure and easy operation. The airflow pulverizer uses high-speed airflow to collide and pulverize materials, which is suitable for the pulverization of heat sensitive, easily oxidizable and other materials, and can obtain finer particle sizes.
V-shaped mixer and three-dimensional motion mixer are commonly used mixing equipment. The V-shaped mixer achieves the purpose of mixing by causing convection, diffusion, and shear of materials inside the container through its rotation. The 3D motion mixer enables the container to perform complex movements in 3D space, resulting in better mixing effects, especially suitable for mixing materials with high viscosity or density differences.
Capsule filling machine is one of the key equipment for producing Tromestane capsuls. According to the different filling principles and structures, capsul filling machines can be divided into fully automatic capsul filling machines and semi-automatic capsul filling machines. The fully automatic capsul filling machine has the advantages of high production efficiency, high filling accuracy, and high degree of automation, and is suitable for large-scale production. The semi-automatic capsul filling machine is relatively easy to operate and suitable for small batch production or laboratory research.
Polishing machine is mainly used to remove stains on the surface of capsuls, making the surface of capsuls smooth. Common polishing machines include drum polishing machines and centrifugal polishing machines. The drum polishing machine achieves the purpose of polishing by causing the capsuls to rub and collide with each other inside the drum through the rotation of the drum. Centrifugal polishing machine uses centrifugal force to make the capsul move at high speed in the polishing chamber, resulting in higher polishing efficiency.
Quality inspection requires the use of various advanced testing equipment, such as high-performance liquid chromatography, ultraviolet spectrophotometer, dissolution tester, microbiological detection equipment, etc. High performance liquid chromatography can be used to determine the content and uniformity of trilostane; UV spectrophotometer can be used for some simple quantitative analysis; The dissolution tester is used to simula`te the dissolution process of drugs in vivo; Microbial detection equipment is used to detect the number of microorganisms in capsuls.
Production environment control
The production of Quluositan capsuls needs to be carried out in a clean workshop to prevent contamination by microorganisms and particles. Cleanrooms are usually classified according to different cleanliness levels, such as A, B, C, and D. In the production process, it is necessary to select appropriate clean level areas for operation according to different production processes. For example, processes such as capsul filling and packaging need to be carried out in areas with higher cleanliness levels.
The temperature and humidity of the production environment have a significant impact on the quality and stability of drugs. Generally speaking, the suitable temperature for the production of Tromestane capsuls is 18-26 ℃, and the relative humidity is 45% -65%. It is necessary to install air conditioning systems and dehumidification equipment to accurately control the temperature and humidity of the production environment.
The hygiene condition of production personnel directly affects the quality of products. Production personnel need to strictly comply with hygiene regulations. Before entering the clean room, they need to change into clean clothes, wear masks, hats, gloves, etc., and disinfect their hands. At the same time, production personnel also need to undergo regular health checks, and those who suffer from infectious diseases or other diseases that may contaminate products are not allowed to engage in direct contact with drug production work.
Key points of quality control in the production process
Raw material quality control
Strictly control the procurement channels of the raw material Tromestane and select suppliers with reliable quality. Inspect each batch of raw materials, including appearance, content, impurities, etc. Only materials that pass the inspection can be put into production.
Control of production process parameters
In the production process, it is necessary to strictly control various process parameters, such as mixing time, speed, filling amount, granulation pressure, etc. Small changes in these parameters may affect the quality of the product. Therefore, it is necessary to develop detailed production process regulations and provide training to operators to ensure that they strictly follow the regulations for operation.
Intermediate product quality control
During the production process, it is necessary to conduct quality inspections on intermediate products, such as mixed materials and granulated particles. The testing items include content uniformity, particle size, moisture, etc. Only when the quality of intermediate products meets the requirements can they proceed to the next process.
Finished product quality traceability
Establish a comprehensive product quality traceability system, and number and record each batch of Trilostane capsule. The recorded content includes information such as raw material batches, production process parameters, quality inspection results, etc. Once product quality issues are discovered, they can be quickly traced back to specific production processes and raw material batches, and timely measures can be taken to address them.
Trilostane capsules represent a paradigm shift in the management of cortisol-related disorders, offering a reversible, enzyme-targeted approach with a favorable safety profile. Its efficacy in Cushing's syndrome and CAH is well-established, while emerging applications in PCOS, prostate cancer, and depression highlight its therapeutic versatility. As research progresses, trilostane may transition from a niche adrenal inhibitor to a broad-spectrum modulator of steroidogenesis, addressing unmet needs in endocrinology and oncology. Clinicians must remain vigilant about monitoring for adrenal insufficiency and drug interactions, but with judicious use, trilostane stands as a cornerstone of modern adrenal pharmacotherapy.
Hot Tags: trilostane capsule, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 2 3 phenanthren 9 yl 5 pyridin 3 yl phenyl 4 6 diphenyl 1 3 5 triazine, 9 bromospiro benzo c fluorene 7 9 thioxanthene , 12H benzofuro 2 3 a carbazole, 4 4 4 4 6 diphenyl 1 3 5 triazin 2 yl phenyl naphthalen 1 yl biphenyl 4 carbonitrile, CAS 1548470 92 3, Methylamine hydrochloride powder









