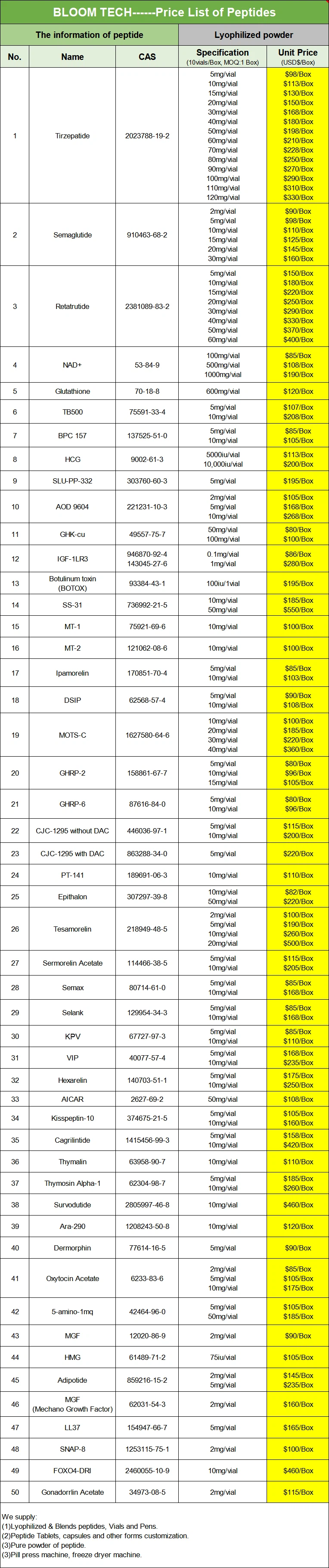
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of ara-290 10mg in China. Welcome to wholesale bulk high quality ara-290 10mg for sale here from our factory. Good service and reasonable price are available.
ARA-290 10mg is a short peptide derived from erythropoietin (EPO) that can exert a wide range of tissue protective impression without causing erythropoiesis. In recent years, it has shown significant applications in inflammation and immune regulation investigate, as well as trauma and surgical investigate. In inflammation and immune regulation investigate, it is used to regulate acute and chronic inflammation, reduce tissue damage, maintain organ action, and improve immune balance; In trauma and surgical investigate, this peptide is used to evaluate tissue repair, trauma recovery, transplant organ preservation, microvessel regeneration, and regenerative medicine investigate.
Our product form





Cibinetide/ARA-290 COA



ARA-290 10mg is a non erythropoietic EPO derived peptide, and its unique properties make it an important experimental tool in tissue preservation, inflammation regulation, and wound recovery investigate. In the field of inflammation and immune regulation, it is widely used in acute and chronic inflammation models, organ preservation, and immune balance regulation investigate. In the field of trauma and surgery, it is used to evaluate tissue repair speed, trauma recovery efficiency, surgical organ preservation, and regenerative medicine potential. Through the integrated analysis of a large number of basic and preclinical studies, it can be found that it is not only an ideal tool in experimental investigate, but also has potential clincal translational value (Brines et al., 2008; Leist et al., 2004).

Specific applications in inflammation and immune regulation investigate
The applications in inflammation and immune investigate include acute and chronic inflammation models, organ preservation, and immune balance regulation. Investigate has shown that this peptide can alleviate inflamed damage and facilitate tissue action recovery through multi-level impression.
1. Acute Inflammation Model
In acute inflammation models, it is widely used to evaluate its impression in reducing tissue damage and regulating inflamed responses.
Investigate has shown that this peptide can reduce neutrophil and macrophage infiltration, decrease the expression of pro-inflamed cytokines such as TNF - α, IL-1 β, IL-6, and improve organ action. For example, in acute lung injury models, it is used to observe the preservation of alveolar structure, reduction of pulmonary edema, and improvement of blood oxygen action. Through histological analysis and inflamed cell counting, investigaters found that it significantly reduced the increase in alveolar wall thickness and cell infiltration, providing experimental basis for acute inflammation intevention (Brines et al., 2008).


In the acute kidney injury model, it is used to evaluate the inhibition of apoptsis of renal tubular epithelial cells and the improvement of serum creatinine and urea nitrogen levels. Investigate has shown that this peptide can reduce oxidative stress indicators such as MDA, enhance antioxidant enzyme activity such as SOD and GPx, thereby reducing renal tubular injury and maintaining renal action. In addition, in the acute myocarditis model, it has been used to alleviate myocardial cell apoptsis and elevated levels of inflamed factors. Evaluation through cardiac ultrasound and histological indicators shows that it can improve myocardial structure and action.
2. Chronic Inflammation and Autoimmune Model
Widely used in chronic inflammation and autoimmune investigate. In rheumatoid arthritis models, this peptide is used to evaluate synovitis control, joint injury reduction, and joint action recovery. Investigate has shown that it can reduce macrophage and lymphocyte infiltration in joint synovium, decrease the expression of pro-inflamed factors such as TNF - α and IL-6, and improve joint structural integrity (Erbayraktar et al., 2010).
In inflamed bowel disease models, it is used to alleviate apoptsis of intestinal epithelial cells,


Improve villous structure, and maintain intestinal barrier action. In multiple sclerosis animal models, it is used to evaluate the alleviation of central nervous system inflamed damage, nerve fiber preservation, and action recovery. Behavioral tests and histological analysis show that it significantly improves neurological action, reduces inflamed cell infiltration, and maintains myelin sheath structure.
3. Investigate on Organizational Preservation and Immune Balance
Poziotinib is used in a multi organ inflammation model to study tissue preservation and immune balance.
Investigate has shown that this peptide can improve inflamed damage in liver, kidney, heart, and lung tissues, maintain microvessel stability, and improve organ action (Leist et al., 2004). Its application scope includes drug-induced liver injury, renal tubular injury, as well as myocardial and lung tissue inflammation. Investigaters found that by analyzing immune cell subsets, inflamed cytokine profiles, and tissue oxygen supply levels, it is possible to maintain immune homeostasis while improving inflamed damage, providing an experimental platform for anti-inflamed drugs and immune regulation inteventions.


4. Joint Intevention Study
In inflammation and immune regulation investigate, ARA-290 10mg is also commonly used in combination intevention experiments, such as in combination with anti-inflamed drugs or immune modulators. Investigate has shown that this peptide can enhance the anti-inflamed impression of drugs, reduce tissue damage, and maintain organ action. For example, in an acute liver injury model, the combination of antioxidants can significantly reduce the apoptsis rate of liver cells and the level of inflamed factors, providing experimental evidence for the study of combination therapy strategies.
Specific applications in trauma and surgical investigate
The applications in trauma and surgical investigate include tissue repair, trauma recovery, organ preservation, microvessel regeneration, and regenerative medicine investigate.
1. Investigate on Trauma Repair
In experimental skin, muscle, and soft tissue trauma models, it is used to evaluate wound repair speed, tissue structure preservation, and inflammation control. Investigate has shown that this peptide can facilitate angiogenesis, reduce fibrosis, inhibit apoptsis, and maintain tissue structural integrity (Brines&Cerami, 2008).


In complex trauma models, such as multiple limb injuries or burn models, it is used to accelerate tissue healing, improve blood flow perfusion, and enhance tissue regeneration ability, while evaluating the impression through histological and hemodynamic indicators.
2. Surgical Surgery and Organ Preservation Investigate
In surgical models, it is used to evaluate organ preservation and action improvement. For example, in liver, kidney, and heart models under ischemia-reperfusion conditions.
It can alleviate ischemic injury, reduce inflamed response, maintain microvessel stability, and improve organ action(Leist et al., 2004). In the transplantation surgery model, this peptide is used to protect the transplanted organ, reduce transplant related inflammation and reperfusion injury, improve organ survival rate, and provide experimental basis for clnical surgical optimization.
3. Investigate on Regenerative Medicine and Tissue Engineering
Used in regenerative medicine investigate to facilitate tissue regeneration, vascularization, and action recovery.


In skin transplantation, muscle reconstruction, and bone defect models, this peptide can improve the survival rate of transplanted tissues, reduce inflamed reactions, and facilitate angiogenesis. Investigate has shown that it can enhance tissue stability and action recovery in the trauma area, providing a reliable experimental basis for surgical regenerative treatment strategies (Brines et al., 2008).
3. Multi organ trauma combined intrvention study
In a multi organ trauma model, poziotinib is used to evaluate the impression of combined inteventions.
Including drug combination, surgical optimization, and biomaterial application. Investigate has shown that this peptide can maintain microcirculation stability in multiple organ injuries
Improving oxygen supply, inhibiting inflammation, and promoting tissue repair provide an experimental platform for combination therapy strategies. In addition, it is also used to verify the auxiliary impression of new drug carriers or biological scaffolds on tissue preservation and regeneration, providing data support for innovative investigate in trauma surgery.


ARA-290 10 mg Development History Timeline
In 1997-1999, the basic investigate team first proposed that erythropoietin (EPO) has non hematopoietic actions, including tissue preservation and anti-inflamed impression.
To avoid the risk of excessive red blood cell production caused by EPO in tissue preservation applications, investigaters have begun to design short peptide derivatives, attempting to preserve tissue preservation action while removing erythropoietic activity.
This stage laid the theoretical foundation for its birth and facilitated preliminary attempts at chemical synthesis.
Investigaters successfully synthesized the substance and identified it as an EPO derived short peptide that can retain tissue protective action.
Early experimental validation was conducted in animal models, covering multiple fields such as cardiac ischemia, kidney injury, and preservation.
Leist et al. reported the protective impression in a multi organ ischemia-reperfusion model in the FASEB Journal, laying the experimental foundation for further preclinical investigate.
Multiple studies confirmed that it still has strong tissue preservation and anti-inflamed abilities without stimulating red blood cell production.
Brines and Cerami systematically summarized its non erythrogenic characteristics and broad tissue protective potential in Nature Medicine and Nature Reviews Drug Discovery, marking the maturity of their investigate.
It has gradually become an important candidate for preclnical drug development, and its investigate direction focuses on dias neurpathy, retinal damage and multiple organ preservation.
Since 2009, it has entered the early clnical trial stage to evaluate the safety and tolerance of dias neurpathy and chronic pain.
In 2010, Erbayraktar et al. reported the first batch of Poziotinib randomized controlled trials in dia Care, showing that it has potential impression in improving the symptoms of dias neurpathy.
This period marks a gradual transition from laboratory investigate to clnical application verification.
It was further used in animal models and small population studies for retinal preservation, myocardial preservation, and peripheral nerve injury investigate.
The investigate team conducted multicenter preclinical trials to evaluate its potential for combined application in inflammation, trauma, and microvessel preservation.
This stage emphasizes its multifunctional application in complex disease models, particularly its translational value in preservation and microvessel homeostasis.
In clinical trials, the therapeutic potential of dias peripheral neurpathy, retinopathy and chronic pain was mainly verified.
Poziotinib investigate began exploring its potential applications in inflamed diseases, trauma recovery, and ophthalmic diseases, including retinal ischemia and surgery related organ preservation.
Investigate has shown that it has good tolerance, can improve neurological action, relieve pain, and improve microvessel perfusion, providing a basis for clinical applications in multiple fields.
References
1. Brines ML, et al. Non-erythropoietic EPO derivatives: tissue protection without erythropoiesis. Nat Med. 2008;14(3):275–281.
2. Brines ML, Cerami A. Emerging therapeutic applications of tissue-protective cytokines. Nat Rev Drug Discov. 2008;7(8):645–659.
3. Erbayraktar S, et al. Cibinetide (Poziotinib) for the treatment of peripheral neurpathy in type 2 dias: a randomized controlled trial. Dias Care. 2010;33(12):2577–2582.
4. Leist M, et al. Cibinetide reduces ischemia/reperfusion injury in multiple organ models. FASEB J. 2004;18(9):991–993.
5. Pieper M, et al. Cibinetide improves neural function in experimental models of diabetic neurpathy. Dias. 2005;54(9):2632–2639.
Hot Tags: ara-290 10mg, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale







