Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of eptifibatide injection in China. Welcome to wholesale bulk high quality eptifibatide injection for sale here from our factory. Good service and reasonable price are available.
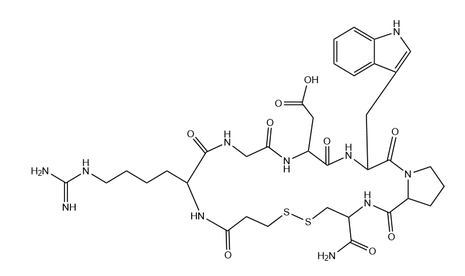
The main component of Eptifibatide Injection is eptibatide, with excipients including citric acid, sodium hydroxide, and injection water. The drug is a colorless or almost colorless clear liquid, with the chemical name N6- (amino iminomethyl) - N2- (3-mercaptopropionyl) - L-lysyl-glycyl-L - α - aspartyl-L-tryptoyl-L-prolyl-L-cysteine aminocyclic (1,6) - disulfide, molecular formula C35H49N11O9S2, and molecular weight 831.96. Mainly used for the treatment of acute coronary syndrome (ACS), including unstable angina and non ST segment elevation myocardial infarction.
Our Products Form






Eptifibatide COA
 |
||
| Certificate of Analysis | ||
| Compound name | Eptifibatide | |
| Grade | Pharmaceutical grade | |
| CAS No. | 188627-80-7 | |
| Quantity | 15g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090059 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.49% |
| Loss on drying | ≤1.0% | 0.36% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.90% |
| Single impurity | <0.8% | 0.58% |
| Total microbial count | ≤750cfu/g | 170 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 400ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
|
|
||
| Chemical Formula: | C35H49N11O9S2 |
| Exact Mass: | 831.32 |
| Molecular Weight: | 831.97 |
| m/z: | 831.32 (100.0%), 832.32 (37.9%), 833.31 (9.0%), 833.32 (7.0%), 832.31 (4.1%), 834.31 (3.4%), 833.32 (1.8%), 832.32 (1.6%), 833.32 (1.1%) |
| Elemental Analysis: | C, 50.53; H, 5.94; N, 18.52; O, 17.31; S, 7.71 |

As a highly effective antiplatelet drug in clinical practice, the core pharmacological effect of eptifibatide injection lies in the specific, competitive, and reversible blockade of platelet membrane glycoprotein (GP) IIb/IIIa receptors, which are the ultimate common pathway for platelet aggregation. Regardless of which agonist is activated upstream, such as ADP, collagen, thromboxane A ₂, thrombin, etc., all platelet aggregation signals need to converge to GP IIb/IIIa receptors, and achieve platelet cross-linking by binding to fibrinogen and von Willebrand factor (vWF). Etriptide mimics the KGD/RGD sequence of natural ligands with a cyclic heptapeptide structure, accurately blocking receptor binding sites and completely blocking platelet aggregation. It is one of the most potent antiplatelet drugs.
The physiological and pathological processes of platelet aggregation and the core position of GP IIb/IIIa receptors
Platelet aggregation is the core process of hemostasis and thrombus formation, which is divided into three consecutive stages: adhesion, activation, and aggregation, forming a complete signaling cascade reaction.
Initiation phase: Platelet adhesion


Endothelial injury → Exposure of subendothelial collagen and vWF → Platelets bind to vWF through GP Ib/IX/V complex → Initial adhesion is completed → Cell membrane receptor aggregation and intracellular signaling are initiated. This stage is locally triggered without large-scale platelet aggregation.
Amplification stage: Platelet activation (inside out signal)
After adhesion, ADP, 5-hydroxytryptamine, and thromboxane A ₂ (TXA ₂) are locally released, and thrombin is activated to form various agonist signals. These agonists bind to corresponding receptors on the surface of platelets (P2Y ₁₂, TP, PAR-1, etc.), triggering intracellular Ca ² ⁺ release, cAMP decrease, PKC activation, initiating signal transduction from the inside out, causing conformational remodeling of GP IIb/IIIa receptors from low affinity to high affinity activated state. This stage is signal amplification, which is a prerequisite for platelet aggregation.


Terminal stage: platelet aggregation (ultimate common pathway)
Activated GPIIb/IIIa receptors expose fibrinogen/vWF binding sites → plasma fibrinogen (divalent molecule) simultaneously binds to two adjacent platelet GPIIb/IIIa receptors → forms a platelet fibrinogen platelet cross-linking bridge → massive platelet aggregation forms white thrombus. This stage is not affected by the type of upstream agonist and is the only terminal pathway for platelet aggregation, as well as the most critical target for antiplatelet therapy.
GP IIb/IIIa receptors (integrin α IIb β ∝) are the most abundant and functionally core receptors on the surface of platelets, determining their ability to aggregate.
molecular structure
A heterodimer with a molecular weight of approximately 240 kDa is formed by non covalent bonding between two subunits, α IIb (GP IIb) and β ∝ (GP IIIa).
Extracellular region: containing ligand binding domains (RGD/KGD recognition site, fibrinogen gamma chain binding site), Ca ² ⁺/Mn ² ⁺ binding region (regulating conformation).


Transmembrane region: Single transmembrane, connecting intracellular and extracellular signals.
Intracellular region: Short peptide chains, combined with ankle protein and focal adhesion kinase, mediate outward in signaling, regulating further activation, contraction, and release of platelets.
Expression distribution
Specific expression: Only present on the surface of platelets/megakaryocytes, each resting platelet contains 80000 to 100000 receptors, which can increase to 120000 to 150000 after activation.
Conformal state: at rest, it is a bent low affinity conformation; When activated, it is an extended high affinity conformation, with ligand affinity increased by 1000-10000 times.
Core ligand
Fibrinogen: the main ligand that binds to GP IIb/IIIa at both ends and mediates platelet cross-linking.
VWF: The main ligand under high shear forces (such as arteries and narrowed blood vessels), mediating firm platelet aggregation.
Other: Fibronectin, Hyaluronidase (minor role).

The key role of uncontrolled platelet aggregation under pathological conditions and GP Ⅱ b/Ⅲ a

In acute coronary syndrome (ACS), PCI, atherosclerosis and other pathological conditions:
Plaque rupture/endothelial injury → strong and sustained platelet activation → extensive activation of GP IIb/IIIa → excessive aggregation → arterial thrombosis → vascular occlusion → ischemic events such as myocardial infarction and cerebral infarction.
Traditional antiplatelet drugs (aspirin, P2Y12 inhibitors) only block a single upstream agonist pathway (such as aspirin blocking TXA ₂, clopidogrel blocking ADP), and cannot inhibit aggregation induced by other agonists, leading to treatment evasion.
The GP IIb/IIIa receptor is the only common terminal target, and blocking this pathway can comprehensively inhibit all agonist induced platelet aggregation, achieving complete antithrombotic effects.

Reference information source:
- Chinese Medical Association Cardiovascular Disease Branch. Expert consensus on clinical application of platelet glycoprotein IIb/IIIa receptor antagonists (2024) Chinese Journal of Cardiovascular Disease, 2024
- Platelet glycoprotein Ⅱb/Ⅲ a receptor: Structure, function, and therapeutic targeting. Nature Reviews Cardiology, 2025.
- The signaling pathway of platelet aggregation and the regulation of GP IIb/IIIa receptors. Chinese Pharmacological Bulletin, 2024
- Pathophysiology of platelet aggregation and thrombosis. Circulation Research, 2024.
- GPⅡb/Ⅲa receptor as the final common pathway of platelet aggregation. Blood, 2024.
The molecular structure of eptifibatide and its structural basis for targeting GP IIb/IIIa receptors
The chemical nature and structural characteristics of eptipatide

Itibatide is an artificially synthesized cyclic heptapeptide derived from the rational design of disintegrin in rattlesnake venom, with a molecular formula of C35H49N11O9S2 and a molecular weight of 831.96.
Primary sequence: 3-mercaptopropionyl-high arginine glycine aspartic acid tryptophan proline cysteine amide (Mpr HArg Gly Asp Trp Pro Cys NH ₂).
Advanced structure: The intramolecular disulfide bond (Cys and Mpr thiol) forms a rigid cyclic conformation, fixing the spatial conformation of the core functional region.
Core functional motif: Modified KGD sequence (Lys Gly Asp, high arginine replaces lysine), which is a highly affinity and specific binding site for GP IIb/IIIa receptors.
The necessity of circular conformation
Linear peptides are easily degradable and conformally unstable, while circular structures resist protease hydrolysis and maintain precise spatial orientation of KGD, resulting in a 100 fold increase in receptor affinity.
The circular structure simulates the C-terminal circular binding domain of fibrinogen gamma chain, which binds to the receptor key and has strong specificity.
The core role of KGD motif
Natural fibrinogen contains two binding sites, RGD (Arg Gly Asp) and gamma chain KGD; The affinity of eptibatide KGD for activating GP IIb/IIIa is about 10 times higher than RGD, and the selectivity is stronger.


HArg (high arginine): The side chain guanidine enhances electrostatic binding with the negative pocket of the receptor, with an affinity (Kd=1.2 nM) much higher than fibrinogen (Kd=100 nM).
Gly (glycine): No side chains, provides conformational flexibility, and adapts to receptor binding slots.
Asp (Asp): carboxyl negatively charged, binds to the metal ion dependent site (MIDAS) of the receptor, and is an essential binding group.
Contribution of auxiliary residues
Trp (tryptophan): The hydrophobic side chain is embedded in the hydrophobic pocket of the receptor, enhancing binding stability.
Pro (proline): rigid ring, fixed conformation, reduces non-specific binding.
The precise simulation of GP IIb/IIIa binding epitopes of fibrinogen/vWF by eptibatide:
The spatial size, charge distribution, hydrophobic region, and natural ligand are highly homologous.
Compete with higher affinity to occupy the receptor ligand binding pocket, completely blocking fibrinogen/vWF binding without cross activation.
Non covalent binding with receptors (hydrogen bonding, electrostatic, hydrophobic interactions) lays the foundation for reversible blockade.

Reference information source:
- Eptifibatide Injection: Structural basis for GPⅡb/Ⅲa receptor antagonism. Journal of Medicinal Chemistry, 2024.
- Structure effect relationship of cyclic heptapeptide GP IIb/IIIa inhibitors. Pharmaceutical Journal, 2025
- FDA. Integralin (Epifibatide) Injection Official Manual (2011 Edition)
- RGD vs KGD mimetics: Selectivity for platelet GPⅡb/Ⅲa receptor. Biochemistry, 2024.
- Molecular design and targeting mechanism of eptipatide. Chinese Journal of New Drugs, 2024
Frequently Asked Questions
What is eptifibatide used for?
+
-
Integrilin: Package Insert / Prescribing Information / MOAEptifibatide (commonly sold as Integrilin) is a glycoprotein IIb/IIIa inhibitor (antiplatelet agent) used to prevent blood clots, heart attacks, or death in patients with acute coronary syndrome (ACS), including unstable angina and NSTEMI, or those undergoing percutaneous coronary intervention (PCI/stenting). It works by stopping platelets from sticking together.
Is eptifibatide an anticoagulant?
+
-
Eptifibatide is an anti-coagulant that selectively and reversibly blocks the platelet glycoprotein IIb/IIIa receptor.
What are the side effects of eptifibatide?
+
-
One of the most common side effects of eptifibatide is bleeding. This is not surprising given that the drug works by inhibiting platelets, which are crucial for blood clotting. Patients may experience minor bleeding issues such as nosebleeds, gum bleeding, or prolonged bleeding from cuts.
Hot Tags: eptifibatide injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale