Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of ziconotide injection in China. Welcome to wholesale bulk high quality ziconotide injection for sale here from our factory. Good service and reasonable price are available.
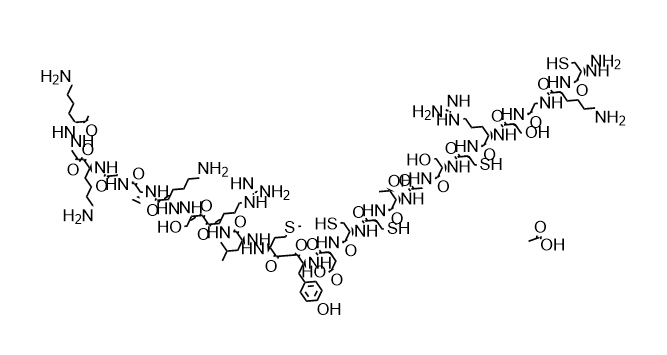
The active ingredient of ziconotide injection is ziconotide, a synthetic ω-conotoxin originally isolated and extracted from the venom of marine cone snails and chemically modified to produce an injectable formulation. It has a molecular weight of 2639.15 and a CAS number of 107452-89-1. The mechanism of action of ziconotide is completely different from that of traditional opioids: instead of acting on opioid receptors, it exerts an analgesic effect by selectively blocking N-type calcium channels in the central nervous system.
Our Products Form






Ziconotide Polyacetate COA
 |
||
| Certificate of Analysis | ||
| Compound name | Ziconotide Polyacetate | |
| Grade | Pharmaceutical grade | |
| CAS No. | 107452-89-1 | |
| Quantity | 30g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090088 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
|
|
||
| Chemical Formula: | C97H170N34O30S5 |
| Exact Mass: | 2511 |
| Molecular Weight: | 2513 |
| m/z: | 2511 (100.0%), 2513 (95.3%), 2511 (40.8%), 2513 (22.6%),2511 (21.5%), 2454 (17.2%), 2453 (12.6%), 2452 (12.0%), 2455 (11.7%), 2453 (11.2%), 2454 (6.5%), 2454 (6.2%), 2453 (5.9%), 2455 (4.5%), 2453 (4.0%), 2456 (3.9%), 2452 (3.8%), 2455 (2.8%), 2454 (2.7%), 2455 (2.5%), 2456 (2.0%), 2453 (2.0%), 2455 (1.9%), 2452 (1.9%), 2454 (1.6%), 2456 (1.5%), 2455 (1.4%), 2456 (1.4%), 2455 (1.3%), 2453 (1.1%), 2452 (1.1%), 2456 (1.1%), 2457 (1.0%) |
| Elemental Analysis: | C, 47.50; H, 6.99; N, 19.42; O, 19.57; S, 6.54 |

Cancer pain is one of the most common symptoms in patients with advanced cancer, severely impairing patients' quality of life, treatment adherence, and survival. As a new non‑opioid analgesic, Ziconotide Injection demonstrates significant advantages in the treatment of refractory cancer pain due to its unique mechanism of action, definite analgesic efficacy, and low abuse potential, serving as an important complement to clinical analgesic therapy.
Application Value in Refractory Cancer Pain

(I) Clear Target Population and High Specificity
The product is indicated for patients with refractory cancer pain whose pain is not adequately controlled (Visual Analogue Scale, VAS ≥ 5) despite standardized opioid therapy, or who cannot tolerate opioid‑related adverse reactions (e.g., severe respiratory depression, intractable constipation, severe nausea and vomiting). It is particularly suitable for patients with concomitant neuropathic cancer pain, which accounts for more than 50% of refractory cancer pain cases.Neuropathic cancer pain has a complex pathogenesis, mainly caused by tumor invasion of nerves, chemotherapy‑ or radiotherapy‑induced nerve injury, and manifests as burning, stabbing, or electric‑shock‑like pain with low sensitivity to traditional opioids.
The product provides marked analgesia for this type of pain.Clinical studies show that intrathecal infusion of it effectively reduces pain scores and improves sleep and quality of life in patients with refractory cancer pain who are opioid‑tolerant or non‑responsive to opioids.A case series of 8 patients with refractory cancer pain (5 with concomitant neuropathic pain) reported that all patients achieved significant pain reduction within 3–5 days after intrathecal administration of ziconotide combined with morphine. One patient who failed to respond to fentanyl still achieved satisfactory pain control after resuming ziconotide therapy, confirming its value in patients unresponsive to opioids.
(II) Significant Analgesic Efficacy and Non‑Addictive Properties
The analgesic potency of it is comparable to that of morphine and even superior in some patients, with a dose‑dependent analgesic effect; optimal analgesia can be achieved through slow dose titration. Multiple clinical studies confirm that intrathecal infusion of it reduces pain scores by more than 30% in patients with refractory cancer pain, with pain relief rates exceeding 70% in some individuals. In a randomized, double‑blind, placebo‑controlled clinical trial involving 169 patients with severe chronic pain (including those with refractory cancer pain), ziconotide treatment reduced the mean VAS score by 31.2%, compared with only 6.0% in the placebo group, representing a statistically significant difference (P ≤ 0.001).
Compared with opioids, the most prominent advantage of ziconotide injection is its lack of addiction potential. It does not act on opioid receptors, does not induce physical or psychological dependence, and does not cause withdrawal symptoms, allowing safe long‑term use. This feature is especially critical for advanced cancer patients requiring prolonged analgesia, as it avoids additional suffering and treatment burden associated with drug addiction. Furthermore, the drug does not cause respiratory depression, providing a higher safety profile for patients with refractory cancer pain and concurrent respiratory insufficiency, with no risk of drug‑induced respiratory failure.
(III) Synergistic Combination with Other Analgesics to Enhance Therapeutic Outcomes
In clinical management of refractory cancer pain, monotherapy often fails to achieve ideal analgesia, making combination therapy a common strategy. The product can be used in combination with opioids, local anesthetics (e.g., bupivacaine), and other analgesics to produce synergistic analgesia, reduce individual drug dosages, and lower the incidence of adverse reactions.
Clinical studies demonstrate that low‑dose ziconotide combined with intrathecal morphine rapidly controls opioid‑refractory cancer pain while reducing morphine dosage and its associated adverse reactions such as constipation and respiratory depression. A case series of 8 patients with refractory cancer pain reported that all patients had failed prior intrathecal morphine plus bupivacaine therapy, but achieved effective pain control after add‑on ziconotide therapy initiated at 0.5–1.0 μg/day and titrated upward by 0.5 μg every 4–7 days (maximum dose ≤ 10 μg/day). Morphine dosages were also reduced, confirming the efficacy and safety of combination therapy.
Information source: Intrathecal Ziconotide and Morphine for Pain Relief: A Case Series of Eight Patients with Refractory Cancer Pain, Including Five Cases of Neuropathic Pain; Intrathecal ziconotide in the treatment of chronic nonmalignant pain: a randomized, double‑blind, placebo‑controlled clinical trial.

Clinical Research Progress and Application Prospects

(I) Clinical Research Progress
In recent years, numerous domestic and international clinical studies have investigated ziconotide injection for refractory cancer pain, further validating its analgesic efficacy and safety. A multicenter, randomized, double‑blind, placebo‑controlled study enrolled 457 patients with severe chronic pain (including refractory cancer pain), randomized to ziconotide (268 patients) or placebo (189 patients) with slow dose titration for 21 days.
The ziconotide group showed a mean pain score reduction of 12%, compared with 5% in the placebo group (statistically significant), with a markedly lower incidence of adverse reactions than with rapid titration.Real‑world evidence also supports its clinical use. An 18‑month prospective, multicenter observational study (PRIZM Registry) included 93 patients with severe chronic pain treated with intrathecal ziconotide. Assessment via the Numeric Pain Rating Scale (NPRS) revealed significant pain reduction at 12 weeks and favorable long‑term safety, confirming its value in prolonged analgesia.
Additionally, studies on neuropathic cancer pain show ziconotide provides superior relief compared with traditional opioids, offering a more effective option for this patient population.
Ongoing clinical research is exploring optimized dosing regimens, including combination strategies and individualized dose titration, to further improve analgesia and minimize adverse reactions. Studies in special populations (e.g., elderly patients, those with hepatic or renal impairment) are also underway to support personalized clinical therapy.
(II) Application Prospects
With rising demand for refractory cancer pain management, the product, as a novel non‑opioid analgesic, has broad clinical prospects. On the one hand, its unique mechanism makes it a vital therapeutic option for patients with opioid tolerance or non‑response, filling a clinical gap and improving quality of life. On the other hand, its non‑addictive and non‑respiratory‑depressant properties reduce adverse events and treatment risks, especially for patients with respiratory insufficiency or high addiction risk.As clinical research advances, it will likely be further validated for specific subtypes of refractory cancer pain, with optimized regimens and expanded indications.

Meanwhile, wider adoption and standardization of intrathecal drug delivery will increase its clinical utilization, benefiting more patients. Development of long‑acting formulations will also be a future research direction, aiming to reduce dosing frequency and improve patient convenience and adherence.
Notably, it has current clinical limitations, including a relatively complex administration route (requiring intrathecal infusion), a high incidence of adverse reactions, and high cost, which restrict its widespread use. Future innovations in technology and cost control will address these barriers, improve clinical accessibility, and benefit more patients with refractory cancer pain.
Information source: Intrathecal Ziconotide and Morphine for Pain Relief: A Case Series of Eight Patients with Refractory Cancer Pain, Including Five Cases of Neuropathic Pain; Intrathecal ziconotide in the treatment of chronic nonmalignant pain: a randomized, double‑blind, placebo‑controlled clinical trial.
Additional Hypersensitivity and Contraindications
Hypersensitivity Contraindication: Contraindicated in patients with known hypersensitivity to ziconotide or any excipients (acetic acid, sterile water for injection, etc.).
Contraindications for Intrathecal Administration: Contraindicated in patients with infections at the infusion site, coagulopathy, spinal canal obstruction, or other disorders impairing cerebrospinal fluid circulation.
Information source: FDA Prescribing Information, DailyMed Database
FAQ
Is ziconotide stronger than morphine?
+
-
Pharmacodynamic studies in human. In a wide range of animal pain models, intrathecal ziconotide produced potent antinociceptive effects, which were at least ten times more potent than those of intrathecal morphine.
What is ziconotide used to treat?
+
-
Ziconotide is an intrathecal analgesic medication used for the treatment of chronic pain. Intrathecal therapy was introduced in the 1980s as a means to treat chronic refractory pain. More commonly used medications today include opioid analgesics such as morphine and local anesthetics such as bupivacaine.
Is ziconotide a peptide?
+
-
Ziconotide is a peptide with the amino acid sequence H-Cys-Lys-Gly-Lys-Gly-Ala-Lys-Cys-Ser-Arg-Leu-Met-Tyr-Asp-Cys-Cys-Thr-Gly-Ser-Cys-Arg-Ser-Gly-Lys-Cys-NH2 (CKGKGAKCSRLMYDCCTGSCRSGKC-NH2) and contains 3 disulfide bonds (Cys1-Cys16, Cys8-Cys20, and Cys15-Cys25).
Hot Tags: ziconotide injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale