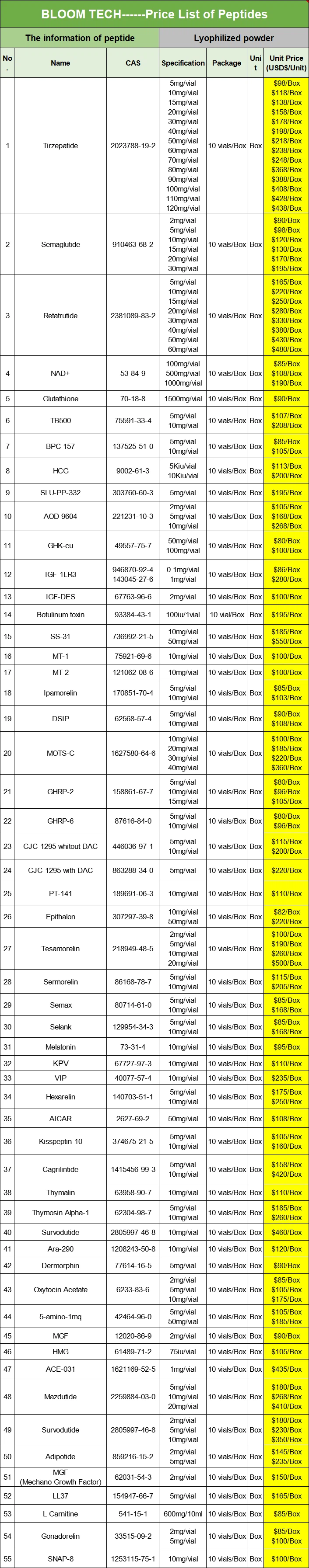
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of hmg injection 75iu in China. Welcome to wholesale bulk high quality hmg injection 75iu for sale here from our factory. Good service and reasonable price are available.
HMG Injection 75IU is an injectable gonadotropin drug containing 75 international units (IU) of follicle stimulating hormone (FSH) and 75 international units of luteinizing hormone (LH). They are secreted by the anterior pituitary gland and play a critical role in the development and maintenance of reproductive system function. FSH mainly stimulates the development and maturation of follicles in the ovaries, promoting the secretion of estrogen; LH mainly stimulates ovulation and corpus luteum formation, promoting the secretion of progesterone. HMG injection simulates the natural secretion of FSH and LH in the body, stimulating the ovaries to produce multiple follicles and increasing the chances of conception. It is usually administered by intramuscular injection. The specific usage and dosage should be determined based on the patient's specific condition and the doctor's advice.
At the same time, our company not only provides pure powders, but also tablets and injections. If needed, please feel free to contact us at any time.
Our products





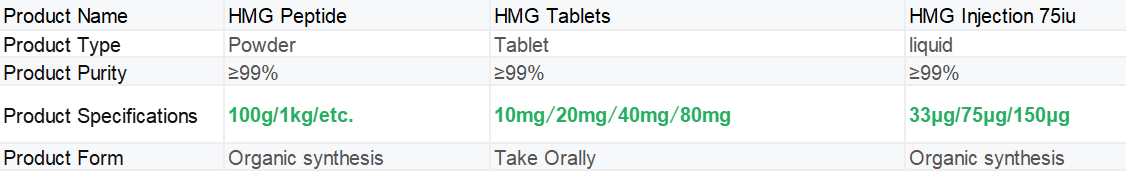
 |
 |
HMG COA
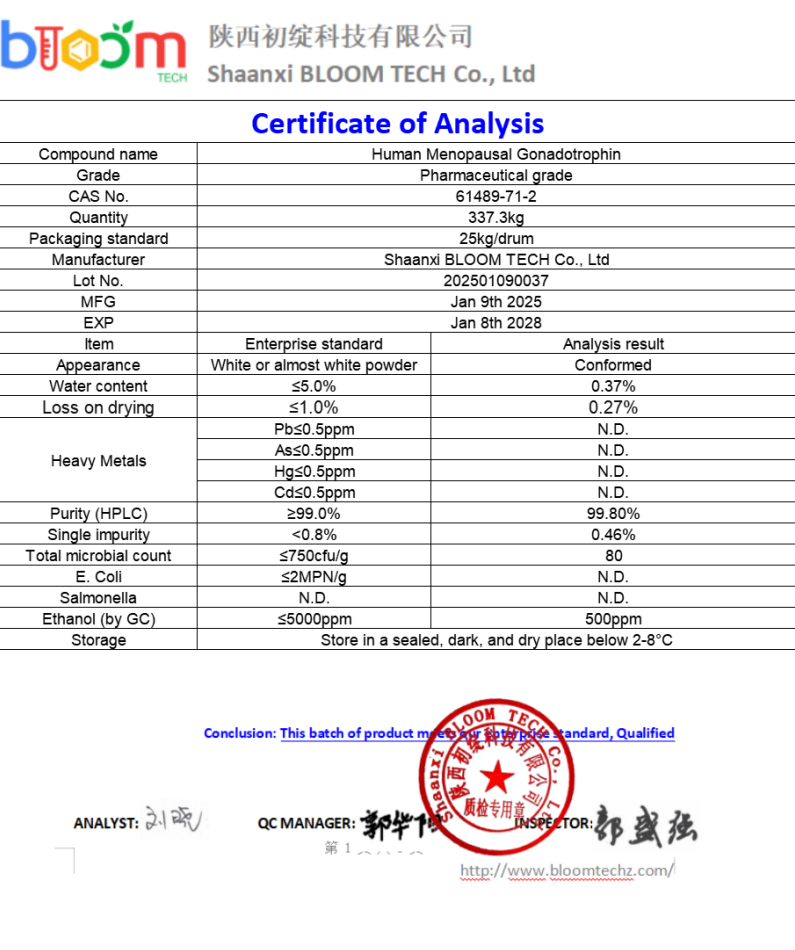
Comparative analysis of the complex purification process of traditional urine source HMG injection and the purification advantages of rHMG (using affinity tags such as His tag)
Human menopausal gonadotropin (HMG) is a hormone mixture extracted from the urine of postmenopausal women, mainly containing follicle stimulating hormone (FSH) and luteinizing hormone (LH), with a small amount of human chorionic gonadotropin (HCG). HMG Injection 75IU is widely used in the treatment of female ovulatory infertility, polycystic ovary syndrome, and assisted reproductive technologies such as artificial insemination and in vitro fertilization. It stimulates the development of ovarian follicles to increase the number of retrieved eggs and improve the success rate. The purification process of traditional urinary HMG is complex and time-consuming, while recombinant human chorionic gonadotropin (rHMG) is synthesized through genetic engineering technology and combined with affinity tag (such as His tag) technology, significantly simplifying the purification process and improving product purity.
Purification process of traditional urine source HMG injection

Raw material sources and pretreatment
The traditional source of HMG in urine is the urine of postmenopausal women, which needs to undergo strict screening to ensure the donor's health and absence of infectious diseases. After urine collection, preliminary treatment is required, including filtration to remove large particle impurities, centrifugation to separate solid and liquid components, and adjusting the pH value to a suitable range (usually pH 5.0-7.0) to create conditions for subsequent extraction processes.
Multi layer chromatography column purification process
The purification of traditional HMG relies on multi-layer chromatography column technology, and the core steps include:
Anion exchange chromatography
Load the preprocessed urine onto an anion exchange chromatography column (such as Capto DEAE), and utilize the negatively charged properties of FSH and LH under specific pH conditions to bind with the positively charged groups in the column. By adjusting the salt concentration or pH value of the eluent, unbound impurities are gradually eluted, and the flow-through solution containing FSH and LH is collected. This step can remove about 70% of impurities in urine, but FSH and LH still need further separation.

Cation exchange chromatography
Concentrate the permeate from anion exchange chromatography and load it onto a cation exchange chromatography column (such as CM Sepharose). Under low salt conditions, FSH and LH are separated by gradient elution (such as increasing NaCl concentration) due to differences in charge and binding ability of the chromatography column. FSH typically elutes at lower salt concentrations, while LH requires higher salt concentrations. This step can separate FSH and LH, but the purity is still insufficient (about 80% -85%), and further purification is needed.

Dye affinity chromatography
To further improve purity, the FSH eluent from cation exchange chromatography is loaded onto a dye affinity chromatography column (such as Capto Blue). Dye molecules (such as Cibatron Blue F3GA) can specifically bind to aromatic amino acid residues in FSH and competitively elute FSH using high salt eluent (such as 1.5 M KCl). This step can increase the purity of FSH to over 95%, but the operation is complex and the cost is high.

Gel filtration chromatography (optional)
If small molecular impurities or aggregates need to be further removed, the products of dye affinity chromatography can be sampled to gel filtration chromatography column (such as Superdex 200). According to the difference in molecular size, FSH and LH are separated while removing impurities such as endotoxins. Although this step can improve the purity of the product, it will significantly increase the time cost.

Limitations of Purification Process
The traditional multi-layer chromatography column purification process has the following problems:
The steps are cumbersome
Multiple chromatography steps such as anion exchange, cation exchange, dye affinity, etc. need to be carried out sequentially, and each step requires optimization of conditions (such as pH, salt concentration, flow rate), making the operation complex and time-consuming.
Purity fluctuation
The elution efficiency of each chromatography step is affected by factors such as raw material quality and column performance, which may lead to fluctuations in product purity (usually 85% -95%).
High cost
Multi layer chromatography columns require a large amount of chromatography media (such as DEAE, CM Sepharose, Capto Blue), and the columns need to be regenerated or replaced regularly, increasing production costs.
Activity loss
Multiple chromatography may lead to a decrease in the biological activity of FSH and LH, which needs to be reduced by adding protective agents (such as BSA) or optimizing operating conditions (such as low temperature, short time).
Purification advantages of rHMG: application of affinity tags (such as His tag)
1. Synthesis principle of rHMG
RHMG is synthesized through genetic engineering technology, and the genes encoding FSH and LH are cloned into expression vectors (such as pET series) and fused with affinity tag sequences (such as His tag). Transform the recombinant plasmid into host cells (such as Escherichia coli, CHO cells) and obtain fusion proteins (such as His FSH, His LH) by inducing expression. The fusion protein needs to be isolated through a purification process when it is in the cell or secreted into the culture medium.
2.Technical principle of affinity tag purification
Affinity tag purification is based on the specific interaction between the tag and the immobilized ligand, and the core steps include:

Fixed metal ion affinity chromatography (IMAC)
His tag consists of six consecutive histidine residues and can bind to immobilized metal ions (such as Ni ² ⁺, Co ² ⁺) through coordination bonds. Load the fusion protein containing His tag onto an IMAC column (such as Ni NTA), where the target protein is adsorbed and impurity proteins flow through. Elute non-specific binding proteins with low concentrations of imidazole (such as 10-50 mM), and then competitively elute target proteins with high concentrations of imidazole (such as 200-500 mM). This step can purify the target protein in one step, with a purity of over 90%.

Label excision and product purification
To obtain unlabeled natural proteins, specific proteases (such as Thrombin, TEV protease) can be added to the eluent to cleave the linker peptide between His tag and the target protein. The label and protease were removed by secondary IMAC or gel filtration chromatography to obtain high-purity natural protein.
3. Advantages of rHMG purification
Affinity tag purification only requires 1-2 steps of chromatography to obtain high-purity products, significantly reducing purification time. For example, His tag purification can achieve the separation of target proteins through single step IMAC without the complex operation of multi-layer chromatography columns.
The specific binding between affinity tags and ligands can efficiently remove impurity proteins, and the purity of the product is usually above 95%. For example, the purity of rHMG purified by His tag can remain stable at over 98%, far higher than the 85% -95% achieved by traditional methods.
Affinity tag purification is carried out under mild conditions (such as neutral pH, low temperature) to reduce denaturation or degradation of the target protein. For example, the concentration of imidazole in IMAC eluent can be optimized through gradient elution to avoid the impact of high concentrations of imidazole on protein activity.
Affinity tag purification reduces the amount of chromatography medium used (such as only requiring IMAC columns), and the chromatography column can be reused (such as Ni NTA columns that can be regenerated more than 10 times), significantly reducing production costs. In addition, the simplification of the purification process also reduces labor and time costs.
Affinity tag purification is easy to scale up production and can increase yield by increasing column volume or parallel chromatography systems. For example, industrial grade IMAC systems can process hundreds of liters of culture medium to meet the large-scale production needs of rHMG.
Comparison of the application effects of traditional urinary source HMG and rHMG
Clinical efficacy
Multiple clinical studies have shown that rHMG is equally effective as traditional urinary HMG in promoting follicular development, increasing ovulation and pregnancy rates. For example, a randomized controlled trial of 120 infertile patients showed that the ovulation rates of the rHMG group and the urinary HMG group were 85% and 82%, respectively, and the pregnancy rates were 35% and 32%, respectively, with no statistically significant difference (P>0.05).
Security
RHMG, due to its higher purity, can reduce immunogenic reactions (such as antibody production) and impurity related side effects (such as allergic reactions). For example, traditional urine source HMG Injection 75iu may contain trace amounts of endotoxins or host cell proteins, while rHMG purification through affinity tags can effectively remove these impurities and reduce the risk of adverse reactions.
Cost effectiveness
Although the unit price of rHMG may be higher than traditional urine source HMG, its simplified purification process, shortened production cycle, and adaptability to large-scale production can reduce overall costs. For example, a cost analysis showed that although the unit dose cost of rHMG is 10% higher than traditional methods, the overall treatment cost can be reduced by 15% -20% due to comparable efficacy and reduced adverse reactions.
Hot Tags: hmg injection 75iu, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale










