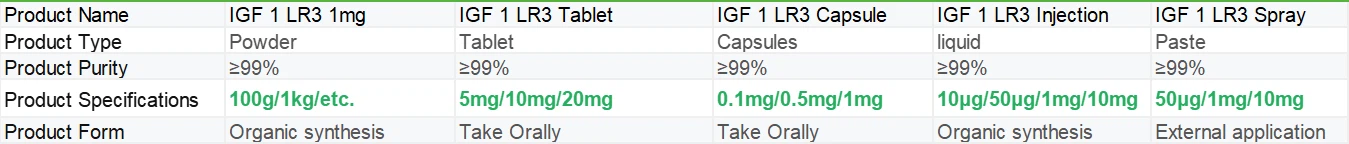
The core component of IGF 1 LR3 Tablet, IGF-1 LR3 (Long R3-IGF-1), is an artificially synthesized insulin-like growth factor-1 analog commonly used in scientific research or specific medical settings in the form of injections. It is a single chain alkaline polypeptide composed of 70 amino acids, mainly synthesized by the liver and regulated by growth hormone (GH). Its structure is similar to insulin, but its growth promoting activity is much higher than insulin. It activates downstream signaling pathways (such as AKT and MAPK) by binding to tyrosine kinase type receptors (IGF-1R), promoting cell proliferation, differentiation, and metabolic regulation. As a growth additive, IGF-1 LR3 significantly increases the proliferation rate and recombinant protein production of HEK293, CHO and other cells, and is widely used in the biopharmaceutical industry. In transgenic mouse models, LR3 is used to study muscle development, metabolic regulation, and aging mechanisms. For example, long-term injection of LR3 can delay muscle atrophy and improve the motor ability of elderly mice.
|
|
|

IGF 1 LR3 COA


IGF-1 LR3 (long-chain human insulin-like growth factor) is a highly active peptide that has been artificially modified. By modifying the amino acid sequence based on human IGF-1, it significantly enhances biological stability and activity, and has important application value in fields such as biomedical research and development, cell culture, etc. The manufacturing of IGF 1 LR3 Tablet (long-chain human insulin-like growth factor tablets) is a complex system engineering that integrates multiple fields of technology such as genetic engineering, formulation processes, and quality control
Core manufacturing process of IGF-1 LR3 tablets
The manufacturing process of IGF-1 LR3 tablets can be divided into three core stages: raw material preparation, formulation molding, coating, and packaging. Each stage includes multiple fine chemical processes and requires strict control of process parameters to ensure product quality uniformity.
Raw material preparation: Gene engineering expression and purification
IGF-1 LR3, as a peptide substance, cannot be efficiently produced on a large scale through chemical synthesis. The mainstream method is to use genetic engineering recombinant expression technology for preparation, and its process has achieved mature transformation from laboratory small-scale testing to 150L fermentation tank pilot testing. The specific steps are as follows:
Construction of expression system
Priority should be given to using eukaryotic expression systems, with the commonly used Pichia pastoris GS115 strain as the host cell. By using DNA recombination technology, the nucleic acid molecule encoding IGF-1 LR3 was cloned into the pPICZ α A plasmid to construct the pPICZ-LR3IGF-1 expression vector. The vector needs to contain elements such as promoters and selective marker genes (such as Zeocin resistance genes) to ensure stable expression of the target gene. Some companies also use the E. coli expression system, such as the products of Wuhan Pronosai, which achieve the expression of target proteins through E. coli.

High density fermentation
The fermentation process is the key to raw material production, and it is necessary to achieve fed batch fermentation in 10-150L fermentation tanks. The optimized core parameters are an inoculation volume of 4% and an IPTG concentration of 0.8 mmol/L. The induction method involves the binding of lactose and IPTG, with an induction time of 4 hours. During the fermentation process, the dissolved oxygen is controlled at 40% -60%, and the pH value is maintained stable. After 12 hours of fermentation, the wet bacterial weight can reach 22-24g/L, and the expression level of the target protein accounts for 25% of the total protein. During the fermentation process, real-time monitoring of parameters such as bacterial biomass, dissolved oxygen, pH, etc. is required to maintain nutrient supply through an automatic feeding system and avoid the accumulation of metabolites that inhibit protein expression.

Bacterial fragmentation and inclusion body treatment
After fermentation, the bacterial cells were collected by centrifugation and sonicated to break down the cells, releasing inclusion bodies containing the target protein. To improve the purity of the inclusion bodies, three gradient washes are required. First, wash twice with 2mol/L urea, and then wash once with PBS buffer. After treatment, the proportion of target protein in the inclusion bodies can be increased to 50%. Subsequently, the inclusion bodies were dissolved in a denaturing solution (pH 8.0) containing 8mol/L urea, 0.1mol/L Tris HCl, and 10mmol/L DTT to ensure complete protein unfolding and prepare for subsequent refolding.

Protein renaturation and purification
The dilution refolding method is used for refolding, and the most suitable refolding solution formula is 0.1mol/L Tris HCl, 1mmol/L EDTA, 3mmol/L oxidized glutathione, 1mmol/L reduced glutathione, and 1% glycine (pH 8.0). After dilution, the protein is stirred at 4 ℃ for 12 hours to promote correct folding. In the purification process, the chromatography combination process is used. First, small molecular impurities are removed by Sephadex G -50 gel chromatography, and then further purified by SP - Sepharose F. F ion exchange chromatography. Finally, high-purity IGF -1 LR3 protein is obtained. Its molecular weight and amino acid sequence are completely consistent with the theoretical value through detection.

Preparation of freeze-dried powder
The purified protein is treated with freeze-drying technology, and the water is directly sublimated from solid to gas under low temperature and low pressure environment, forming a stable white crystalline powder. This process ensures that proteins remain active during storage and transportation. Freeze dried powder can be stored stably for several weeks at room temperature and can be stored for several years at -80 ℃.

Formulation molding: a key process in tablet preparation
The peptide properties of IGF 1 LR3 Tablet make it sensitive to the gastrointestinal environment, and tablet formulations need to address issues such as low bioavailability and susceptibility to enzymatic degradation. Therefore, targeted optimization is required in prescription design and process selection. The molding process of its formulation refers to the general process of peptide tablets and is adjusted according to its own characteristics, as follows:

1
Prescription design and excipient screening
The prescription design should take into account protein stability, dissolution rate, and formability. Microcrystalline cellulose and lactose should be used as diluents, with a ratio of about 3:1 to ensure tablet hardness and facilitate dissolution; The adhesive uses a 5% polyvinylpyrrolidone K30 aqueous solution to avoid damaging the protein structure with organic solvents; The disintegrant selected is cross-linked carboxymethyl cellulose sodium, with an addition amount of 3% -5%, to ensure rapid disintegration of the tablet in the gastrointestinal tract; The lubricant used is magnesium stearate, with a dosage of 0.5%, to reduce friction during compression; Simultaneously adding 1% -2% meglumine as a solubilizer to improve the water solubility of IGF-1 LR3.

2
Crushing and sieving
Grind the IGF-1 LR3 freeze-dried powder and excipients separately through an air flow crusher, controlling the particle size D90 of the raw material at 20-30 μ m, and matching the particle size of the excipients with the raw material to avoid uneven mixing due to particle size differences. After crushing, pass through an 80 mesh sieve to remove clumped particles and impurities, ensuring the fluidity of the material.

3
Mixing and granulation
The IGF-1 LR3 raw material was initially mixed with diluent and disintegrant using an equal incremental method for 30 minutes to ensure uniform dispersion of the raw material. Subsequently, polyvinylpyrrolidone K30 adhesive was added and wet granulation was performed using a swing granulator (16 mesh sieve) for granulation. Wet particles should be dried at 40-45 ℃ for 2-3 hours, with a controlled moisture content of 3% -5%. The drying temperature should be strictly controlled to avoid protein denaturation caused by high temperatures. After drying, the particles are sieved through a 20 mesh sieve to remove large particles and fine powder.

4
Tablet pressing
Select an 8mm shallow concave die for tablet pressing, with a pressing force controlled at 18-20KN, tablet hardness maintained at 60-80N, and tablet weight difference controlled within ± 3%. During the tablet pressing process, real-time monitoring of tablet weight, hardness, and brittleness is required, with sampling and testing every 15 minutes to avoid quality defects caused by fluctuations in equipment parameters. At the same time, the tablet press needs to undergo aseptic treatment, and the production environment should be controlled in a D-level clean area with a temperature of 18-26 ℃ and a relative humidity of 45% -65%.

5
Coating
To protect IGF-1 LR3 from damage by gastrointestinal enzymes, a thin film coating process is used. The coating solution uses OBA premix, diluted with purified water to a concentration of 15%, and coated using an efficient coating machine. During the coating process, the inlet air temperature should be controlled at 45-50 ℃, the outlet air temperature at 30-35 ℃, the spraying rate at 5-8ml/min, and the coating weight should increase by 2-3%. The surface of the coated tablets is smooth, which can delay gastric dissolution, rapidly release drugs in the intestine, and improve bioavailability.
Production equipment and environmental control
Core production equipment and functions
The production equipment for IGF 1 LR3 Tablet covers multiple fields such as biological fermentation, formulation molding, and inspection and testing. The accuracy and stability of the equipment directly affect product quality. The specific equipment list and functions are as follows:
| Equipment category | Specific equipment | Core functionality | Key parameter requirements |
| fermentation equipment | 10-150L fermentation tank | High density cultivation of recombinant engineering bacteria | Temperature control accuracy ± 0.5 ℃, dissolved oxygen control range 0-100%, pH control range 5.0-9.0 |
| Cell processing equipment | Ultrasonic cell disruptor | Breaking bacterial cells releases inclusion bodies | Power 500-1000W, continuous working time ≥ 4 hours |
| Purification equipment | Gel chromatography column, ion exchange chromatography system | Separate and purify the target protein | Pressure tolerance ≥ 0.5MPa, adjustable flow rate range 0.1-10ml/min |
| Freeze drying equipment | Vacuum freeze dryer | Preparation of protein freeze-dried powder | Vacuum degree ≤ 10Pa, condensation temperature ≤ -50 ℃ |
| Preparation equipment | Airflow crusher, swing pellet machine | Material crushing and granulation | The particle size can be controlled during crushing, and the mesh size of the granulation sieve can be replaced |
| Tablet pressing equipment | Rotary tablet press | Press the particles into shape | Compression speed 5000-10000 tablets/hour, tablet weight accuracy ± 1% |
| Coating equipment | Efficient coating machine | Tablet film coating | Coating uniformity deviation ≤ 2% |
| Packaging equipment | Aluminum plastic blister packaging machine, vacuum packaging machine | Product sealed packaging | Packaging qualification rate ≥ 99.9% |
Hot Tags: igf 1 lr3 tablet, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale








