Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of neomycin sulphate tablets in China. Welcome to wholesale bulk high quality neomycin sulphate tablets for sale here from our factory. Good service and reasonable price are available.
Neomycin sulphate tablets are an oral aminoglycoside antibiotic preparation that plays a significant role in clinical anti-infective therapy. It exerts potent bactericidal activity primarily by irreversibly binding to the 30S subunit of bacterial ribosomes, interfering with bacterial protein synthesis. It exhibits good antibacterial activity against various Gram-negative bacteria, such as Escherichia coli, Klebsiella spp., Proteus spp., and also has certain inhibitory effects on some Gram-positive bacteria. After oral administration, it is absorbed to a small extent and maintains a high concentration in the intestine, making it an ideal choice for the treatment of intestinal infections. It can exert potent antibacterial activity locally while reducing the occurrence of systemic adverse reactions.



medicinal property
Chemical characteristics and dosage form differences:
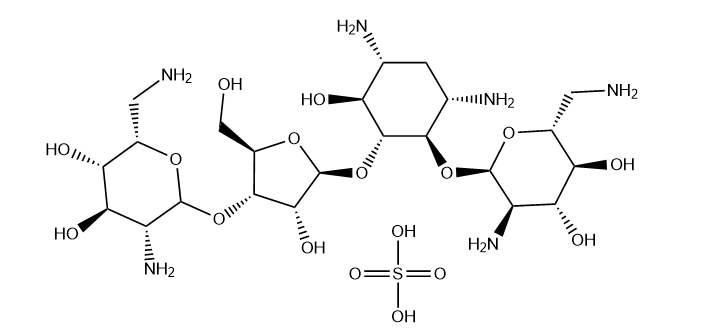
The chemical name of Neomycn Sulphate is Neomycn Sulphate, with a molecular formula of C23H48N6O17S and a molecular weight of 712.7222. Its oral preparations are mainly tablets, with common specifications including 0.1g (100000 units) and 0.25g (250000 units), and need to be sealed and stored in a dry environment to prevent moisture absorption and degradation.
Applicable diseases:
Mainly used for intestinal infections caused by sensitive bacteria, such as bacterial dysentery, acute enteritis, etc. For preoperative preparation of the intestine caused by sensitive bacteria, reducing the number of bacteria in the intestine and lowering the risk of postoperative infection also have significant effects. In addition, it can also be used together with other anti tuberculosis drugs for adjuvant treatment of tuberculosis to enhance the antibacterial effect.

Additional information of chemical compound:
| Product Name | Neomycin Sulphate Powder | Neomycin Sulphate Tablets | Neomycin Sulphate Injection | Neomycin Sulphate Ointment |
| Product Type | Powder | Tablet | Injection | Cream |
| Product Purity | ≥99% | ≥99% | ≥99% | ≥99% |
| Product Specifications | Customizable | Customizable | Customizable | Customizable |
| Product Package | Customizable | Customizable | Customizable | Customizable |
Our Product




Neomycin Sulphate +. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Neomycin Sulphate | |
|
CAS No. |
1405-10-3 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|
|
|
||

As a representative drug of aminoglycoside antibiotics, neomycin sulphate tablets achieves efficient antibacterial effects through the synergistic action of multiple levels and targets, accompanied by specific toxicity characteristics and resistance risks.
The core antibacterial mechanism of neomycn sulphate is to interfere with the entire process of bacterial protein synthesis, and its targets cover the initiation, extension, and termination stages of translation, forming a "triple blockade" effect:
1. Initial stage inhibition
The drug specifically binds to the A site of the bacterial ribosome 30S subunit, hindering the formation of the initiation complex (30S mRNA fMet tRNA). This process relies on hydrogen bonding between the amino cyclic alcohol structure in the drug molecule and specific base pairs of ribosomal RNA (rRNA) (such as A1492-G1491), resulting in mRNA codons being unable to correctly recognize the starting tRNA, thereby blocking translation initiation.
2. Interference during the extension phase
During peptide chain elongation, neomycn sulphate disrupts protein synthesis in two ways:
Misreading induction: Drug molecules embed into the decoding center of the 30S subunit, changing the rRNA conformation and reducing the pairing specificity between aminoacyl tRNA (aa tRNA) and mRNA codons, leading to the incorporation of non natural amino acids into peptide chains and the formation of non functional proteins.


Early termination of the chain: The drug binds to the P site of the ribosome, interferes with peptidyl transferase activity, promotes the premature release of unfinished peptide chains, and inhibits the translocation of peptidyl tRNA from the A site to the P site.
3. Changes in cell membrane permeability
Protein synthesis errors lead to abnormal synthesis of bacterial cell membrane components (such as phospholipids and porins), a decrease in membrane potential, and leakage of cellular contents (such as ATP and potassium ions), ultimately causing bacterial dissolution and death. This effect is particularly significant in Gram negative bacteria, as the absence of outer membrane porins (such as OmpF) can exacerbate drug-induced membrane damage.
The antibacterial spectrum of neomycin sulphate exhibits typical characteristics of "strong Gram negative bacteria, limited Gram positive bacteria, and ineffective anaerobic bacteria", and its activity differences are closely related to bacterial cell wall structure and drug permeability:
Highly active bacterial strains
Gram negative bacteria: Escherichia coli, Klebsiella, Proteobacter, Pseudomonas aeruginosa (some strains), Salmonella, Shigella.
Partial Gram positive bacteria: Methicillin resistant Staphylococcus aureus (MRSA, high concentration required), Staphylococcus epidermidis, Streptococcus genus (sensitive strains).
Other: Mycobacterium tuberculosis (inhibitory effect).


Low activity or drug-resistant bacterial strains
Anaerobic bacteria: Bacteroidetes and Clostridium (due to the lack of aerobic respiratory chain, drugs cannot be actively transported into cells).
Gram positive bacteria: Streptococcus pneumoniae, Enterococcus genus (thick cell wall and lack of outer membrane porin, poor drug permeability).
Special strain: Pseudomonas aeruginosa (some strains develop resistance through the production of aminoglycoside modifying enzymes or the absence of outer membrane protein D2).
The bactericidal activity of neomycn sulphate exhibits concentration dependent characteristics, and its pharmacokinetic parameters are closely related to the design of clinical medication regimens
Concentration dependent sterilization
The higher the drug concentration, the more significant the inhibition of bacterial protein synthesis and the faster the sterilization rate. Experiments have shown that when the concentration of neomycn sulphate reaches 8-10 times the MIC, 99.9% of sensitive bacteria can be killed within 2-4 hours.


Post antibiotic effects (PAE)
Even if the drug concentration drops below MIC, it can still inhibit bacterial protein synthesis for up to 2-4 hours. The presence of PAE supports a "single dose per day" regimen, which can maintain effective antibacterial concentrations while reducing the risk of otorenal toxicity.
Anti drug mutation concentration (MPC)
The MPC of neomycn sulphate is about 4-8 times that of MIC, indicating that clinical monitoring of blood drug concentration is necessary to ensure that the drug concentration is higher than MPC, in order to reduce the selection pressure of drug-resistant mutant strains.
The resistance of neomycin sulphate is mainly generated through the following mechanisms, and clinical attention should be paid to the risks of cross resistance and multidrug resistance:
Target modification
16S rRNA methyltransferase: Methylation modifications mediated by coding genes (such as rmtA, rmtB) can block drug binding to the 30S subunit, leading to high-level drug resistance (MIC ≥ 128 μ g/mL).
Ribosomal protein mutations, such as S12 protein (rpsL gene) mutations, can reduce the affinity of drugs to ribosomes, but usually only lead to low-level resistance (MIC 2-8 μ g/mL).


Drug inactivating enzyme
Aminoglycosyltransferase (AAC): catalyzes the acetylation of amino groups in drug molecules, reducing their binding ability to ribosomes.
Aminoglycosyltransferase (APH): By phosphorylation modification, changes the distribution of drug charges, hindering their penetration into the cell membrane.
Aminoglycoside nucleoside transferase (ANT): catalyzes the adenylation reaction, leading to drug inactivation.
Overexpression of external pump
The RND family efflux pumps of Gram negative bacteria, such as AcrAB TolC, can actively pump drugs out of the cell and reduce intracellular drug concentrations.
biofilm formation
Bacteria in the biofilm state have decreased drug permeability and metabolic activity, requiring higher concentrations to achieve bactericidal effects.
The otonephrotoxicity of neomycn sulphate is closely related to its accumulation in renal tubular epithelial cells and cochlear hair cells, mainly involving the following mechanisms:
mitochondrial damage
The drug interferes with mitochondrial protein synthesis by binding to mitochondrial 12S rRNA, leading to a decrease in respiratory chain complex activity, reduced ATP production, and activation of cell apoptosis programs.
oxidative stress
Drug induced elevation of intracellular reactive oxygen species (ROS) levels leads to lipid peroxidation, DNA damage, and protein carbonylation, ultimately resulting in cell death.
Lysosomal membrane rupture
In renal tubular epithelial cells, neomycin sulphate tablets can disrupt lysosomal membrane stability, release proteases and other hydrolytic enzymes, and trigger cell autolysis.

Clinical Medication Insights: Precision Medication and Toxicity Prevention and Control
Based on the mechanism of action and toxicity characteristics, the clinical application of neomycin sulfphate should follow the following principles:
Indication selection
Prioritize use for Gram negative bacterial infections (such as intestinal infections and urinary tract infections), and avoid use for infections dominated by Gram positive bacteria (such as streptococcal pharyngitis).
Dose optimization
Adopting a "high-dose, long interval" regimen (such as single daily administration) to maximize PAE effects and reduce toxicity risks.
Combination therapy
Combined with β - lactam or fluoroquinolone drugs, it synergistically kills bacteria through different mechanisms of action, reducing the occurrence of drug resistance.
Toxicity monitoring
Regularly monitor blood creatinine, urea nitrogen, and hearing threshold during medication, and adjust dosage or discontinue medication in a timely manner.

Progress and Future Directions in Clinical Research
Exploration of treatment for drug-resistant bacteria
The combination therapy of neomycin sulphate tablets with beta lactams (such as amoxicillin and clavulanic acid potassium) or fluoroquinolones (such as ciprofloxacin) is currently being studied for multidrug-resistant Gram negative bacterial infections. Preliminary results show that synergistic effects can improve efficacy and reduce the risk of drug resistance.
Local administration optimization
Develop sustained-release tablets and enteric coated formulations of neomycin sulphate to reduce systemic exposure and lower the incidence of nephrotoxicity through targeted release. For example, enteric coated tablets prepared using hydroxypropyl methylcellulose coating technology can achieve targeted release of drugs in the intestine, reducing blood drug concentration by 40% -60%.
Monitoring of Biomarkers
Studying the application value of urinary N-acetyl - β - D-glucosaminidase (NAG enzyme) and blood cystatin C as early renal toxicity markers is expected to achieve early intervention of renal injury.
Hot Tags: neomycin sulphate tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale