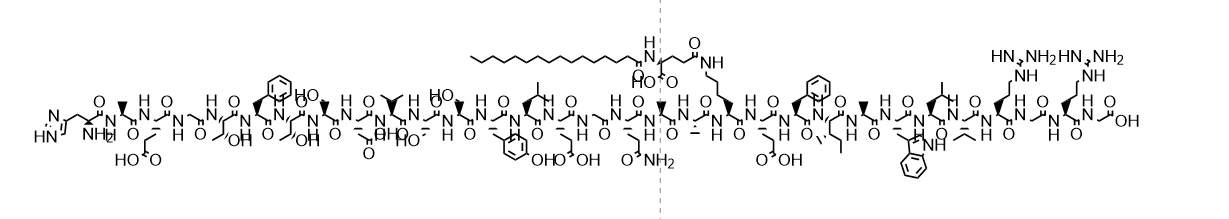
Liraglutide tablets are a glucagon like peptide-1 (GLP-1) analogue, belonging to long-acting GLP-1 receptor agonists. Although the current mainstream dosage form is injection, theoretically, if the tablet form is successfully developed, it will still retain its core characteristics as a GLP-1 receptor agonist.
The molecular structure of Liraglutide is produced through genetic recombination technology and shares 97% sequence homology with human GLP-1. Its unique molecular design, such as fatty acid side chain modification, extends the half-life of the drug in the body, making it only necessary to administer once a day. If developed into tablets, special formulation techniques such as enteric coating and osmotic pump technology are required to ensure stable release and absorption of the drug in the gastrointestinal tract. If developed as tablets, the specifications may be designed for different doses according to clinical needs (such as 0.6mg, 1.2mg, 1.8mg, etc.), which need to be adjusted based on the absorption characteristics and bioavailability of the drug in the gastrointestinal tract.
Our product


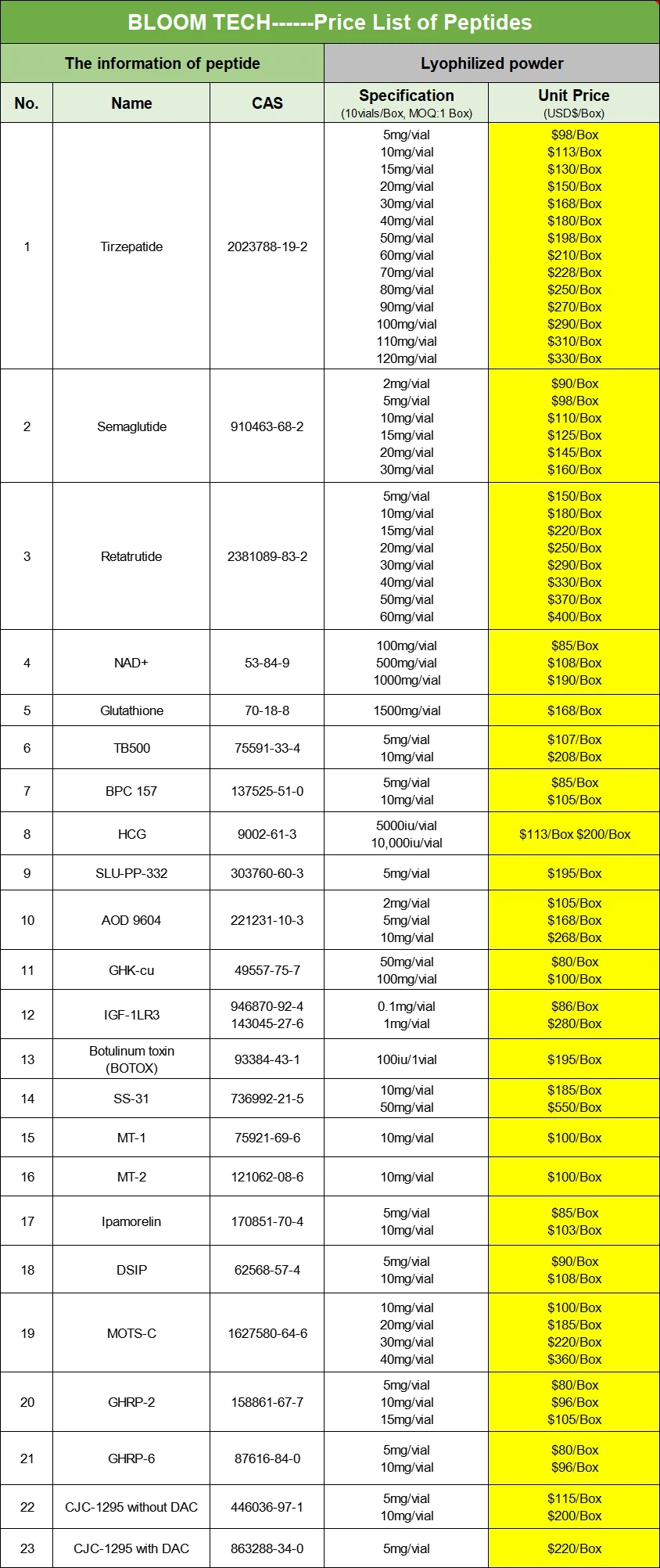

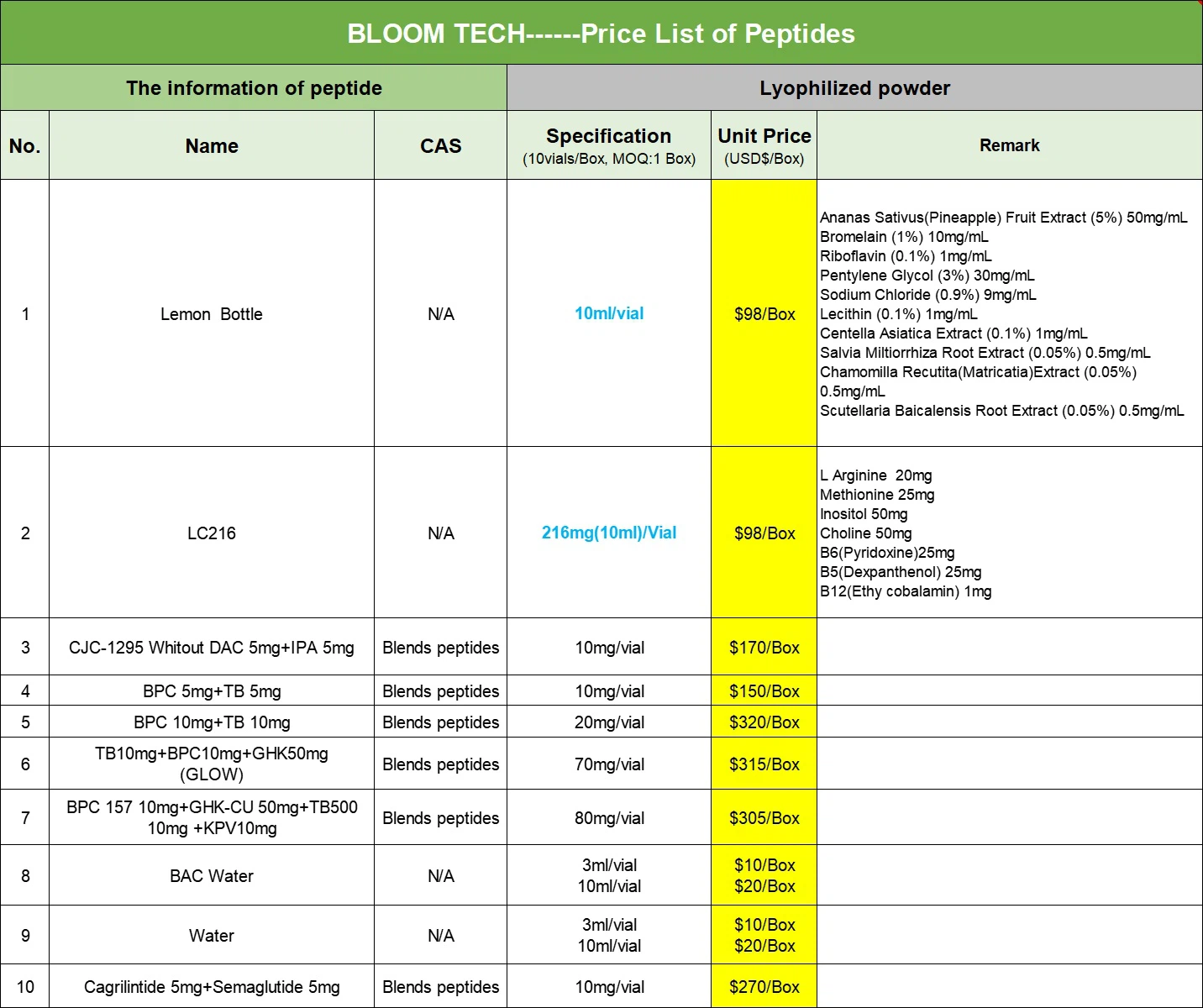

Additional information of chemical compound:
| Product Name | Liraglutide Capsule | Liraglutide Injection | Liraglutide Tablets |
| Product Type | Capsule | Injection | Tablets |
| Product Purity | ≥99% | ≥99% | ≥99% |
| Product Specifications | Customizable | Customizable | Customizable |
| Product Package | Customizable | Customizable | Customizable |
Our Product



Liraglutide+. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Liraglutide | |
|
CAS No. |
204656-20-2 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|
|
|
||

Drug interactions and precautions
Drug Interactions
In vitro studies have confirmed that the likelihood of pharmacokinetic interactions between liraglutide tablet and other active substances related to cytochrome P450 and plasma protein binding is extremely low. However, the mild delay in gastric emptying caused by liraglutide may affect the absorption of other drugs taken orally at the same time. Therefore, if developed into tablets, special attention should be paid to the interactions with other oral medications, especially those that are sensitive to absorption rates.
Precautions
Dose adjustment: When adjusting the dose of liraglutide, blood glucose monitoring is not required (unless in combination with sulfonylurea therapy and adjusting the sulfonylurea dose). However, when combined with sulfonylurea drugs for treatment, blood glucose monitoring may be necessary to adjust the dosage of sulfonylurea drugs.
Omission treatment: If it is a missed dose of an injection, the next day's dose should be used directly according to the prescription requirements, without the need to give additional doses or increase the dose to compensate for the missed dose. If more than 3 days have passed since the last administration of liraglutide, patients should restart at a dose of 0.6mg according to a dose escalation schedule to alleviate any gastrointestinal adverse reactions associated with resuming treatment. If developed as tablets, the handling principles for missed doses may be different from those for injections. A specific supplementation plan should be developed based on the half-life and duration of action of the drug.
Efficiency leap Precision and stability
Injection site: Injection should be injected into the abdomen, thigh, or upper arm, while tablets do not need to consider injection site issues, but attention should be paid to medication timing and gastrointestinal reactions.
Special Symptoms: It has been found that the use of other GLP-1 analogs is associated with the risk of developing pancreatitis. Therefore, patients should be informed of the characteristic symptoms of acute pancreatitis (such as persistent and severe abdominal pain). If pancreatitis is suspected, liraglutide and other potentially suspicious drugs should be discontinued.


The absorption of Liraglutide after subcutaneous injection is relatively slow, reaching its maximum concentration 8-12 hours after administration. Single subcutaneous injection of Liraglutide 0 After 6mg, the estimated maximum concentration of liraglutide is 9 4nmol/L (average weight about 73kg). At a dose level of 1.8mg of liraglutide, the average steady-state concentration (AUCT/24) of liraglutide reached approximately 34nmol/L (average body weight of approximately 76kg). After administering 3mg to obese subjects (BMI 30-40kg/m2), the average steady-state concentration (AUCT/24) of Liraglutide reached 31nmol/L. The exposure level of liraglutide decreases with increasing body weight. The exposure level of liraglutide increases proportionally with the dose. The intra individual coefficient of variation of the area under the drug time curve (AUC) for a single dose of liraglutide is 11%. The absolute bioavailability of Liraglutide after subcutaneous injection is about 55%.
The apparent distribution volume after subcutaneous injection is 11-17L, and the average distribution volume after intravenous injection is 0.07L/kg. For a person weighing approximately 100kg, the average apparent volume of distribution after subcutaneous injection is 20-25L. Liraglutide can bind extensively to plasma proteins.
Within 24 hours of a single administration of radiolabeled [3H]-liraglutide to healthy subjects, the main component in plasma is the liraglutide prototype drug. Two small amounts of plasma metabolites were detected (≤ 9% and ≤ 5% of total plasma radioactive exposure, respectively). Liraglutide is metabolized in a manner similar to macromolecular proteins, and no specific organ has been identified as the main elimination pathway.
After administration of [3H]-liraglutide, no intact liraglutide tablet was detected in urine and feces. Only a small portion of the radiation given is excreted as Liraglutide related metabolites in urine or feces (6% and 5%, respectively). The radioactivity in urine and feces is mainly excreted within the first 6-8 days, corresponding to three small amounts of metabolites. The average clearance rate of Liraglutide after a single subcutaneous injection is about 0.9-1.4 L/hour, and the elimination half-life is about 13 hours.

(1) Gender: Based on pharmacokinetic analysis of overweight and obese patient populations, women have a 24% lower weight adjusted clearance rate than men. The group pharmacokinetic study results of male and female patients with type 2 diabetes and a pharmacokinetic study conducted in healthy subjects showed that gender would not have a clinically significant impact on the pharmacokinetics of lilalutide. According to the exposure response data, there is no need to adjust the dosage based on gender.
(2) Racial origin: The results of a pharmacokinetic analysis of a population including white, black, Asian, and Spanish subjects showed that racial differences did not have clinically relevant effects on the pharmacokinetics of liraglutide.
(3) Weight: The population dynamics analysis of adult type 2 diabetes suggests that body mass index (BMI) will not have a significant impact on the pharmacokinetics of rilalutide. In the exposure response assessment of weight management clinical trials, a daily dose of 3.0mg of liraglutide can provide appropriate systemic exposure within the weight range of 60-234kg. The exposure to liraglutide decreases with increasing baseline body weight. There has been no study on the exposure level of liraglutide for patients weighing over 234kg.
(4) Liver function impairment: A single dose clinical trial evaluated the pharmacokinetics of liraglutide in subjects with varying degrees of liver function impairment. Compared to healthy subjects, subjects with mild to moderate liver dysfunction had a 13-23% reduction in exposure to liraglutide. The exposure to liraglutide was significantly reduced (44%) in subjects with severe liver dysfunction (ChildPugh score>9).


(5) Renal dysfunction: Compared to subjects with normal renal function, subjects with renal dysfunction have reduced exposure to liraglutide. Mild (creatinine clearance rate, CrCl 50-80ml/min), moderate (CrCI 30-50ml/min), and severe (CrCl<30ml/min) renal impairment and end-stage renal disease subjects requiring dialysis reduced liraglutide exposure by 33%, 14%, 27%, and 28%, respectively. Similarly, in a 26 week clinical trial, the liraglutide exposure of type 2 diabetes patients with moderate renal impairment (creatinine clearance rate 30-59ml/min) was 26% lower than that of a trial of type 2 diabetes patients with normal renal function or mild renal impairment.
(6) Pregnant women: This product should not be used during pregnancy, and insulin is recommended at this time. If the patient plans to become pregnant or is already pregnant during the treatment period, the treatment with this product should be stopped. Breastfeeding women: Due to lack of relevant experience, this product should not be used during lactation. Children's medication: No relevant data has been obtained yet, and this product is not recommended for use in adolescents.

Challenges and Prospects of Tablet Development
Development Challenges
Formulation technology: The development of Liraglutide requires solving the problem of stable release and absorption of the drug in the gastrointestinal tract. Special formulation techniques such as enteric coating and osmotic pump technology are required to ensure the stability and bioavailability of the drug in the gastrointestinal tract.
Clinical trials: New clinical trials are required to validate the efficacy, safety, and tolerability of the tablets. Clinical trials should include different doses, different dosing regimens, and long-term safety studies.
Market competition: Currently, the Liraglutide injection market is relatively mature, and the tablet form needs to face competition with the injection. We need to win market share through differentiation strategies such as medication convenience, patient compliance, etc.
Development prospects
Medication convenience: Tablet form is more convenient to administer compared to injection, which can improve patient compliance.
Market demand: With the increasing number of patients with type 2 diabetes and obesity, the demand for new hypoglycemic and weight reducing drugs is also growing. If Liraglutide tablets are successfully developed, they will meet the market demand for new hypoglycemic and weight loss drugs.
Policy support: The government's increasing support for the biopharmaceutical industry has provided a favorable policy environment for the development of Liraglutide tablets.
Frequently Asked Questions
Does liraglutide come in pill form?
+
-
Liraglutide, the active ingredient in Saxenda, is available only as an injection. But a similar medication called semaglutide comes in injectable (Ozempic, Wegovy) and pill (Rybelsus) forms.
What is the use of liraglutide tablet?
+
-
Liraglutide is used to lower the risk of cardiovascular events (heart attack, stroke, or death) in people who have type 2 diabetes and heart disease. Liraglutide may also be used for other conditions as determined by your healthcare provider. Liraglutide is also available in a different form for weight management.
Can liraglutide be given orally?
+
-
Highlights. Developed an oral liraglutide nanomicelle for enhanced insulin resistance reduction. Utilized ASBT-mediated transport to enhance absorption of the oral formulation. Achieved 4.63-fold higher oral bioavailability than unformulated liraglutide.
Who cannot take liraglutide?
+
-
Tell your doctor if you or anyone in your family has or has ever had thyroid cancer or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2; condition that causes tumors in more than one gland in the body). If so, your doctor will probably tell you not to use liraglutide injection.
Which is better, Ozempic or liraglutide?
+
-
Ozempic® (semaglutide) typically leads to more weight loss than liraglutide medications like Saxenda® or Victoza®, making it the stronger option for most people. Many people also prefer Ozempic's once-weekly injection schedule over liraglutide's daily dosing.
Hot Tags: liraglutide tablet, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale