Tesamorelin Tablets are a pharmaceutical formulation containing the active ingredient Tesamorelin, mainly used to treat specific metabolic diseases. It is an artificially synthesized growth hormone releasing hormone (GHRH) analog that stimulates the secretion of growth hormone (GH) from the hypothalamus, thereby regulating physiological processes such as fat metabolism, protein synthesis, and bone growth. It can directly act on the GHRH receptor in the anterior pituitary gland, promoting the release of endogenous growth hormone, rather than directly supplementing exogenous growth hormone. Tesamorelin is the only FDA approved drug for treating abdominal fat accumulation (fat metabolism disorder) in HIV infected individuals. HIV patients may experience fat redistribution (such as increased abdominal fat and decreased limb fat) due to long-term antiretroviral therapy (ART). This medication can significantly reduce subcutaneous fat volume in the abdomen and improve body shape.
Our products



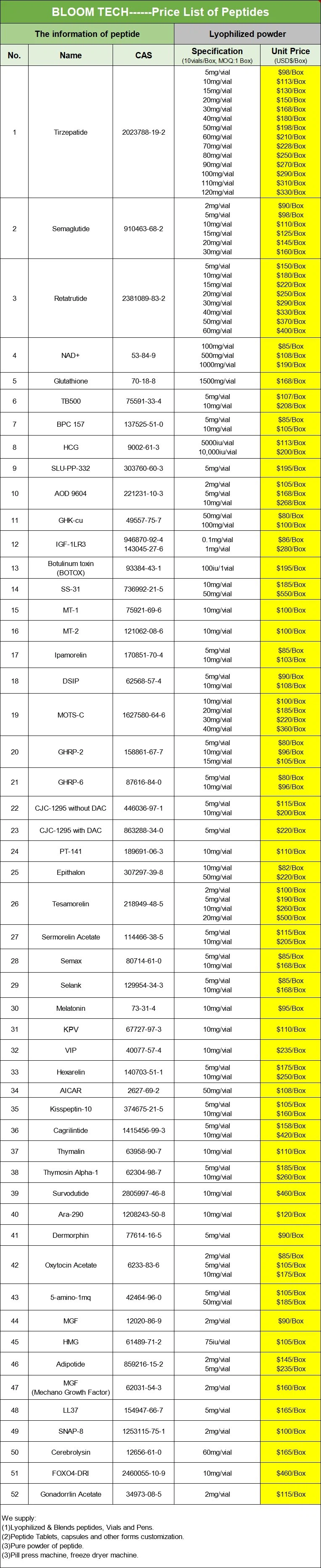
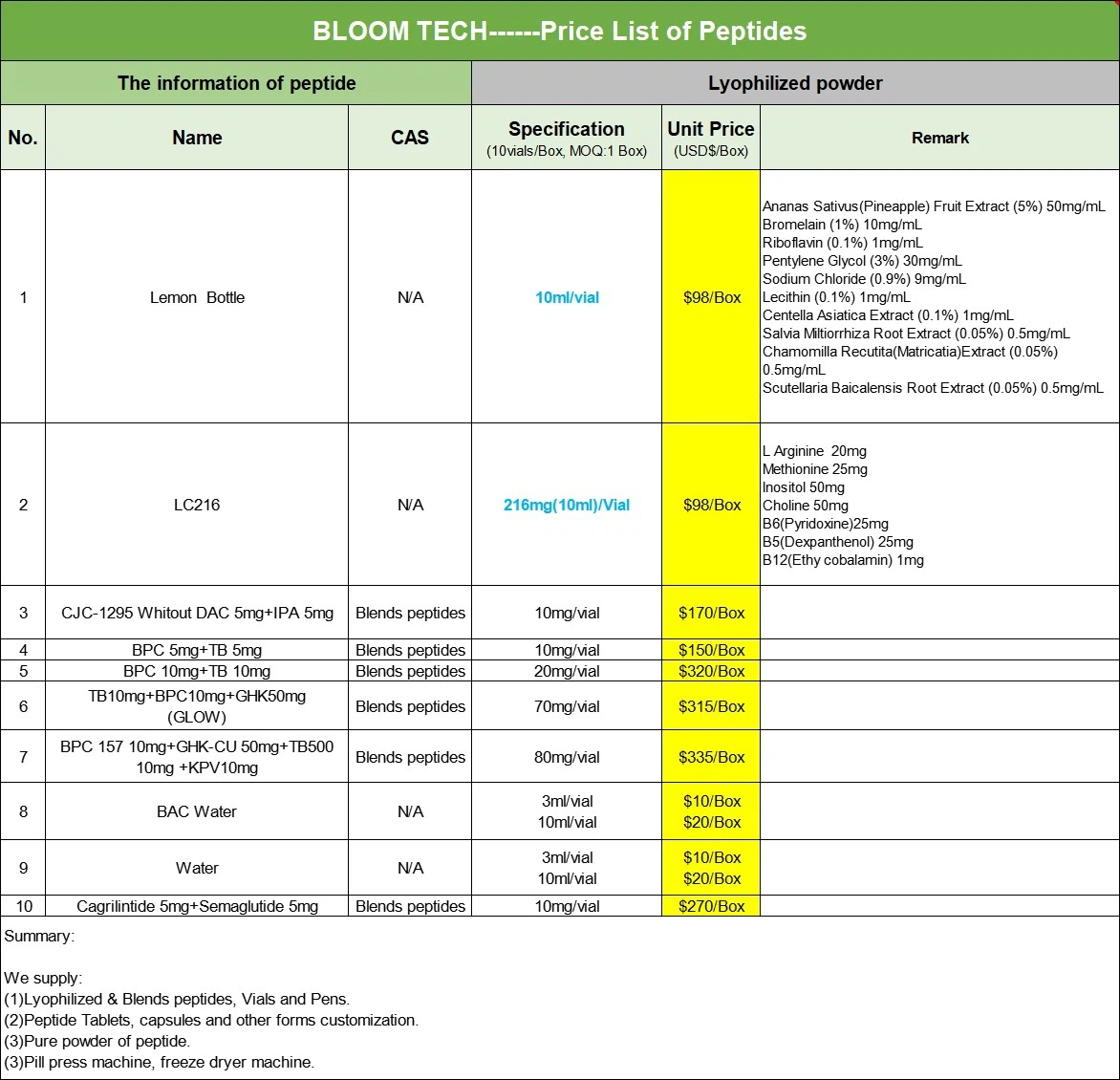

| Product Name | Tesamorelin Powder | Tesamorelin Tablets | Tesamorelin Injection |
| Product Type | Powder | Tablet | liquid |
| Product Purity | ≥99% | ≥99% | ≥99% |
| Product Specifications | 100g/1kg/etc. | 12.5mg/20mg | 2mg/2ml |
| Product Form | Organic synthesis | Take Orally | Organic synthesis |
Tesamorelin COA
 |
||
| Certificate of Analysis | ||
| Compound name | Tesamorelin | |
| Grade | Pharmaceutical grade | |
| CAS No. | 218949-48-5 | |
| Quantity | 337.3kg | |
| Packaging standard | 25kg/drum | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202501090033 | |
| MFG | Jan 9th 2025 | |
| EXP | Jan 8th 2028 | |
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.39% |
| Loss on drying | ≤1.0% | 0.28% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.80% |
| Single impurity | <0.8% | 0.44% |
| Total microbial count | ≤750cfu/g | 80 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage | Store in a sealed, dark, and dry place below 2-8°C | |
|
|
||

Tesamorelin Tablets is an artificially synthesized growth hormone releasing hormone (GHRH) analog primarily used to treat abdominal fat metabolism disorders caused by antiretroviral therapy (ART) in HIV infected individuals.
Technical Challenges and Solutions for Tesamorelin Oral Tablets
Gastric acid degradation: Gastric acid (pH 1.5-3.5) can hydrolyze amide and ester bonds in peptides, leading to drug inactivation.
Intestinal enzymatic hydrolysis: Trypsin, chymotrypsin, and other enzymes can specifically cleave polypeptide chains, disrupting drug structures.
Moisture absorption: Tesamorelin is prone to absorbing environmental moisture, leading to clumping or degradation.

Stability Issues and Protection Strategies

Enteric coating technology: pH dependent polymers (such as hydroxypropyl methylcellulose phthalate, HPMCP) are used to coat the tablets, making them insoluble in the stomach and releasing the drug after entering the small intestine (pH>5.5).
Enzyme inhibitor co delivery: Combining intestinal enzyme inhibitors (such as Camostat, soy trypsin inhibitors) or penetration enhancers (such as sodium cholate, sodium lauroyl sulfate) to reduce drug degradation and promote absorption.
Stabilizing excipients: Adding fillers such as mannitol and lactose, as well as adhesives such as hydroxypropyl cellulose (HPC), to improve tablet hardness and moisture resistance.
Excessive molecular weight: The molecular weight of Tesamorelin (5111.6 Da) far exceeds the threshold for active transport by intestinal cells (usually<500 Da), resulting in extremely low passive diffusion efficiency.
Polar surface charge: The charged groups of peptides (such as arginine and lysine) interact with the phospholipid bilayer of the cell membrane, hindering transmembrane transport.

Penetration Enhancement Technology

Nanocarrier technology: Encapsulating drugs in liposomes, polymer nanoparticles, or solid lipid nanoparticles (SLNs) to promote absorption through endocytosis or membrane fusion mechanisms. For example, the use of polylactic acid hydroxyacetic acid copolymer (PLGA) to prepare nanoparticles can significantly improve the retention time of drugs in the intestine.
Chemical modification: Increasing drug lipophilicity through polyethylene glycol (PEG) or fatty acid conjugation modification. For example, covalently combining Tesamorelin with lauric acid to form a prodrug and improve membrane permeability.
Ionic liquid technology: using ionic liquids (such as 1-butyl-3-methylimidazolium hexafluorophosphate) to dissolve drugs and form low viscosity solutions to enhance intestinal mucosal permeability.
First pass effect: After oral medication is absorbed through the intestine, some of it is metabolized by the liver, resulting in a decrease in systemic exposure.
Individual differences: There are individual differences in intestinal pH, enzyme activity, and transporter protein expression, which affect the consistency of drug absorption.

Optimization of bioavailability

Micro needle patch assisted delivery: Combined with transdermal micro needle technology, microchannels are formed on the surface of the intestinal mucosa to promote direct drug absorption.
3D printing technology: customized tablet structures (such as multi-layer tablets, sustained-release cores) to achieve targeted release of drugs in specific parts of the intestine. For example, using fused deposition modeling (FDM) technology to print inner layer tablets containing enzyme inhibitors and outer layer tablets containing drugs.
Artificial intelligence (AI) assisted design: using machine learning models to predict drug excipient interactions and process parameters, optimizing tablet formulations. For example, using the random forest algorithm to screen for the optimal coating material thickness and nanoparticle loading capacity.
Manufacturing process of Tesamorelin oral tablets

Preparation of raw materials
Solid phase synthesis: Using Wang resin as a carrier, amino acids are gradually coupled through Fmoc/tBu strategy to synthesize Tesamorelin linear peptide chains. Cut the resin with trifluoroacetic acid (TFA) to obtain crude peptides, and purify them by high performance liquid chromatography (HPLC) (purity>98%).
Freeze drying: Freeze the purified peptide solution to -80 ℃, vacuum sublime to remove moisture, and obtain a white powdery raw material. Store at -20 ℃ to avoid degradation.
preparation technology

Enteric coated nanoparticle tablets
Preparation of nanoparticles: Emulsification solvent evaporation method was used: Tesamorelin (5% w/v) and PLGA (10% w/v) were dissolved in dichloromethane and mixed with a water phase containing polyvinyl alcohol (PVA, 2% w/v). After ultrasonic emulsification, the organic solvent was evaporated, and the nanoparticles were collected by centrifugation.
Tablet compression: Mix nanoparticles (30% w/w), microcrystalline cellulose (50% w/w), and magnesium stearate (2% w/w), dry compress to obtain core tablets. Coating: Immerse the core tablet in an ethanol solution containing HPMCP (10% w/v) and dry to form an enteric coating layer.
Ionic liquid pellet tablets
Preparation of microspheres: Extrusion rolling method: Dissolve Tesamorelin (5% w/v) in 1-butyl-3-methylimidazolium hexafluorophosphate ionic liquid, mix with hydroxypropyl methylcellulose (20% w/v), extrude into strips, and roll into microspheres (diameter 1-2 mm).
Tablet assembly: Mix microspheres (40% w/w), lactose (40% w/w), and cross-linked carboxymethyl cellulose sodium (10% w/w), wet granulation, tablet pressing, and coating.

Process validation and optimization
Dissolution test
USP Type II paddle method was used to determine the drug release curve under pH 1.2 (gastric fluid simulation) and pH 6.8 (intestinal fluid simulation) conditions to ensure the integrity of the enteric coating.
Stability study
Under accelerated testing (40 ℃/75% RH) and long-term testing (25 ℃/60% RH), regularly check the appearance, content, and related substances of the tablets to confirm the expiration date (usually ≥ 24 months).
Bioequivalence (BE) test
Through a cross study of healthy volunteers, the blood concentration time curves of oral tablets and injections are compared to verify whether the bioavailability meets the standard (usually requiring a relative bioavailability of ≥ 80%).
The unique positioning of Tesamorelin: different from the targeting effect of ordinary growth hormone releasing peptides (GHRH)
Growth hormone releasing peptide (GHRH) is a neuropeptide secreted by the hypothalamus that regulates metabolism, growth, and immune function by activating the secretion of growth hormone (GH) in the anterior pituitary gland. Traditional GHRH analogues, such as Sermorelin, have limited clinical applications due to their short half-life and low bioavailability. Tesamorelin Tablets, as an artificially synthesized GHRH analog, have shown significant differences in their mechanism of action, pharmacokinetics, and clinical indications through structural optimization and targeted design, especially in the treatment of HIV related lipid metabolism disorders.
Molecular structural differences: from natural peptides to highly selective analogues
Natural GHRH is composed of 44 amino acids (GHRH (1-44)), with its core active region located at the N-terminal amino acids 1-29, of which 1-21 are essential sequences and 22-29 enhance receptor binding affinity. GHRH activates the G protein coupled signaling pathway by binding to the extracellular domain of the GHRH receptor (GHRH-R) in the anterior pituitary gland, promoting GH synthesis and release. However, natural GHRH is easily degraded by dipeptidyl peptidase IV (DPP-IV) in the blood and has a certain affinity for GHRH-R in non pituitary tissues such as fat and muscle, leading to off target effects.
Tesamorelin achieves targeted enhancement through the following structural modifications:
N-terminal acetylation modification: Introducing an acetyl group at the N-terminus of histidine (His) to block the cleavage site of DPP-IV and prolong the half-life to 2-3 hours (natural GHRH only takes 5-10 minutes).
D-amino acid substitution: Replace L-tryptophan at position 2 with D-tryptophan to enhance stereoselective binding with GHRH-R and reduce cross activation of other G protein coupled receptors (such as VIP receptors).
C-terminal truncation and functionalization: retaining the 1-29 core sequence of natural GHRH, but introducing non natural amino acids (such as α - methyllysine) through chemical synthesis to optimize the interaction with the receptor transmembrane domain and improve signal transduction efficiency.
| Characteristic | Natural GHRH (1-44) | Tesamorelin |
| Amino acid composition | 44 L-amino acids | 29 amino acids (including D-tryptophan) |
| Half-life | 5-10 minutes | 2-3 hours |
| Receptor selectivity | Low (easily cross activated VIP receptors) | High (only activating GHRH-R) |
| Enzyme stability | Easily degraded by DPP-IV | Resistant to enzymatic hydrolysis |
Targeted differences in receptor binding and signal transduction
Distribution and function of GHRH receptors
GHRH-R belongs to the G protein coupled receptor (GPCR) family and is mainly expressed in anterior pituitary growth hormone cells, but also at low levels in adipose tissue, skeletal muscle, pancreas, and immune cells. Traditional GHRH analogs, such as Sermorelin, may activate non pituitary tissue receptors simultaneously due to their low affinity, leading to off target effects such as insulin resistance or lipolysis.
Targeted binding mechanism of Tesamorelin
High affinity binding
The binding affinity of Tesamorelin to GHRH-R (Kd ≈ 0.2 nM) is significantly higher than that of natural GHRH (Kd ≈ 1.5 nM), due to the enhanced hydrophobic interaction between its D-tryptophan and the extracellular loop of the receptor.
Molecular docking simulations showed that the C-terminal α - methyllysine of Tesamorelin can be inserted into the hydrophobic pocket of the receptor transmembrane domain, stabilizing the receptor conformation and promoting G α s protein activation.
Organizational specific activation
Pituitary priority targeting: The concentration of Tesamorelin in the pituitary gland is 10-20 times higher than in plasma, due to the high permeability of the pituitary capillary bed to large molecular peptides and the significantly higher local GHRH-R expression density compared to peripheral tissues.
Peripheral tissue desensitization: Long term use of Tesamorelin does not upregulate GHRH-R expression in fat or muscle, while natural GHRH may cause receptor downregulation due to sustained stimulation, leading to drug resistance.
Signal pathway selectivity
Tesamorelin Tablets mainly activates the G α s-cAMP PKA pathway, promotes GH gene transcription, and has weaker activation effects on the G α i or G α q pathways (which may mediate fat breakdown or inflammatory responses).
Animal experiments have shown that Tesamorelin treatment does not increase the release of free fatty acids from rat adipose tissue, while equal doses of natural GHRH can significantly increase plasma free fatty acid levels.
Hot Tags: tesamorelin tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale