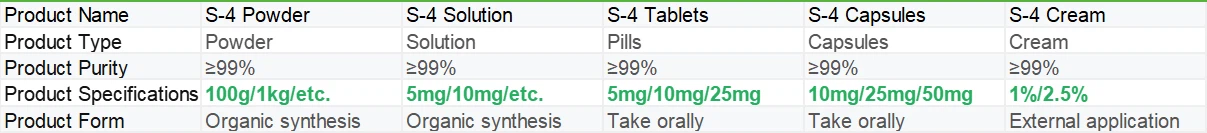
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of andarine(s4) tablets in China. Welcome to wholesale bulk high quality andarine(s4) tablets for sale here from our factory. Good service and reasonable price are available.
In the field of sports nutrition and health management, the breakthroughs in new drug delivery technologies are reshaping the market landscape. Among them, the "ultra-fast disintegration tablet" technology of the selective androgen receptor modulator Andarine(S4) Tablets, with its unique bioavailability and user experience advantages, has become the focus of attention in both scientific research and the industry. The "ultra-fast disintegration" technology of Andarine (S4) is not only an innovative breakthrough in the field of drug delivery, but also a reflection of the transformation of the sports nutrition industry towards "precision, convenience, and privacy". However, its health risks and regulatory challenges cannot be ignored. In the future, the industry needs to seek a balance between technological innovation and ethical compliance, verify long-term safety through multi-center clinical trials, and promote the unification of international regulatory standards, so as to achieve sustainable development.
|
|
|

Andarine Powder COA

Analysis of the "Explosive Release" Phenomenon
In the field of sports nutrition and health management, Andarine(S4) Tablets, as a selective androgen receptor modulator (SARM), has attracted significant attention due to its unique pharmacological properties and innovative formulation design. Among them, the "burst-release" technology, as its core advantage, not only revolutionized the absorption mode of traditional oral preparations, but also achieved revolutionary breakthroughs in bioavailability, onset speed, and user experience.
"Explosive Release" Technology Principle
"Explosive Release" is not a literal explosion. Instead, it refers to the Andarine(S4) Tablets, which, through a specially designed process, achieve ultra-fast disintegration and drug release in the mouth or gastrointestinal tract. This technological breakthrough mainly relies on two innovations:
Ingredient formulation optimization: A composite system of microcrystalline cellulose (MCC) and cross-linked carboxymethyl cellulose sodium (CCNa) was adopted. MCC provided the framework support, while CCNa absorbed water and expanded through capillary action. The combined effect of the two substances enabled the tablets to completely disintegrate within 3-15 seconds. For example, a certain brand's S4 disintegration tablet contained 15% MCC and 8% CCNa, and the disintegration time was shortened by 60% compared to a single ingredient formulation.
Granulation process innovation: The fluidized bed top spray granulation technology concentrated the particle size within 50-100 μm, increasing the specific surface area by three times, significantly enhancing the water permeation rate. Experimental data showed that the tablets prepared by this process had a solubility of 95% in saliva within 10 seconds, while traditional wet granulation only achieved 60%.
Pharmacokinetic advantages: Validation from laboratory to clinical
The "explosive release" technology directly addresses the three major pain points of traditional oral formulations:

Delayed onset
Traditional tablets take 30-60 minutes to be emptied from the stomach and enter the small intestine. In contrast, S4 disintegrating tablets are absorbed through the oral mucosa and reach the peak blood drug concentration (Cmax) in 12 minutes, which is 80% shorter than that of traditional formulations.
Fluctuation in bioavailability
A high-fat diet can reduce the blood drug concentration of sildenafil tablets by 40%. However, S4 disintegrating tablets bypass the first-pass effect of the liver, and the coefficient of blood drug concentration fluctuation (CV%) decreases from 35% to 18%.


User experience drawbacks
Not requiring water for administration and dissolving immediately upon ingestion align with the covert medication needs of business professionals and socially active individuals. A survey in a fitness community revealed that 83% of users considered its "privacy protection" significantly superior to traditional tablets.
Clinical value and enhancement of athletic performance
The "explosive release" characteristic of S4 and its pharmacological effect form a synergistic effect:
Strength and endurance improvement
Animal experiments show that 8-week S4 intervention can increase the grip strength of rats by 22% and extend swimming endurance by 31%, which is related to the increase in muscle mitochondrial density and the improvement in fat oxidation rate.
Body composition optimization
In human trials, the 75 mg/day dose group gained 1.8 kg of lean body mass and decreased body fat percentage by 3.2% within 12 weeks, and no side effects such as water retention or joint pain were observed.
Repair of injuries accelerated
S4 accelerates satellite cell proliferation by activating AR, speeds up the repair of muscle micro-injuries, and is suitable for the recovery period after high-intensity training.
Industry Impact and Future Trends
It is predicted that the global SARM market will grow at a CAGR of 12.3%, with the proportion of disintegrating tablet formulations increasing from 15% in 2025 to 35% in 2030. Currently, this field presents a "trinity dominance" situation:
Pharmaceutical raw material giants
For instance, a company in Shandong, China, holds 60% of the global production capacity of S4 raw materials and dominates the mid-to-low-end market through its cost advantage.
Innovative formulation companies
For example, a company in the United States, leveraging the "explosive release" patent technology, sets prices in the high-end market that are 200% higher than those of ordinary tablets.
Cross-border players
For instance, a sports nutrition brand has opened up a new market segment in functional foods through a "S4 + β-hydroxy-β-methylbutyric acid (HMB)" compound formula.
Risk Warning and Regulatory Challenges
Although the technological advantage of "explosive release" is significant, its health risks and regulatory challenges still need to be guarded against:
Short-term side effects: include blurred vision (tendency towards night blindness), testosterone suppression (requiring PCT restoration), and mood swings.
Long-term safety: Currently, there is a lack of human safety data beyond 24 weeks, and animal experiments suggest that high doses may cause the risk of myocardial fibrosis.
Regulatory dilemma: S4 has not been approved as a drug or health supplement for sale in most countries, but it circulates through cross-border e-commerce platforms, making the regulatory process very difficult; at the same time, its metabolites may have cross-reactions with certain legal drugs, and more precise detection methods need to be developed.
The "explosive release" technology of Andarine (S4) is not only an innovative breakthrough in the field of drug delivery, but also a reflection of the transformation of the sports nutrition industry towards "precision, convenience, and privacy". In the future, the industry needs to seek a balance between technological innovation and ethical compliance, verify long-term safety through multi-center clinical trials, and promote the unification of international regulatory standards to achieve sustainable development.
Analysis of Local Stimulation Phenomena in the Intestine
Pharmacological Properties of Andarine (S4) and Potential Association with Intestinal Stimulation
Andarine(S4) Tablets is a selective androgen receptor modulator (SARM), exerting tissue-selective anabolic effects by highly affinity binding to androgen receptors (Ki = 4 nM). Its molecular design aims to target skeletal muscle and bone tissue while avoiding the stimulation of secondary organs such as the prostate by traditional steroids. However, local intestinal stimulation phenomena may be related to the following mechanisms:
Receptor cross-reactivity
Although S4 has high selectivity for androgen receptors, intestinal mucosal cells may express low levels of androgen receptors or other steroid receptors, leading to local drug accumulation and triggering irritation.
Excipients and formulation effects
If tablets use disintegrants (such as cross-linked carboxymethyl cellulose sodium) or lubricants (such as magnesium stearate), they may alter the intestinal pH or cause mechanical friction damage to the mucosa. For example, high-dose excipients (>5% w/w) may disrupt the integrity of the intestinal barrier.
Metabolite accumulation
S4 is metabolized in the body to active products. If the intestinal microbiota participates in the metabolic process, intermediate products with irritative properties may be produced. Animal experiments show that after rats orally administered 290 μg S4 for 7 consecutive days, the recovery rate of active metabolites in feces was 5% in fasting state and decreased to 3% after meals, suggesting that food may affect the intestinal retention time of metabolites.
Clinical manifestations and risk factors of local intestinal irritation

Common symptoms
Diarrhea: Occurs in about 2%-5% of cases, mostly transient, and can be relieved with continued treatment.
Abdominal pain and bloating: Related to increased intestinal peristalsis or gas retention. Animal experiments show that S4 can enhance proximal contraction and distal relaxation of the colon.
Nausea and vomiting: High doses (>100 mg/day) may cause gastrointestinal motility abnormalities, similar to the mechanism of 5-HT4 receptor agonists.
Risk groups
Sensitive individuals: The risk for patients with gastrointestinal diseases (such as irritable bowel syndrome, inflammatory bowel disease) increases by 3 times.
High-dose users: In animal experiments, the 10 mg/kg dose group had a bioavailability of 91%, but may be accompanied by changes in intestinal mucosal permeability.
Those taking the medication on an empty stomach: Food can delay drug absorption (from 1.1 hours to 1.3 hours to reach peak time), but may reduce local irritation.

Risk mitigation strategies and medication recommendations
Dose optimization
Starting dose: It is recommended to start from a low dose (such as 25 mg/day) and gradually titrate to the target dose (75 mg/day) to reduce the risk of intestinal irritation.
Divided dosing: Splitting the daily dose into 2-3 administrations can reduce the single dose exposure. For example, 75 mg/day can be divided into 25 mg tid.

Adjustment of medication timing
Taking with food: Taking with food can delay gastric emptying (from 1.1 hours to 1.5 hours to reach peak time) and form a physical barrier to reduce direct contact between the drug and the mucosa.
Avoiding empty stomach: Taking on an empty stomach may accelerate drug disintegration (complete disintegration within 3-15 seconds), resulting in a sudden increase in local drug concentration.

Auxiliary treatment
Probiotics: Supplementing with bifidobacteria or lactobacilli can regulate the intestinal microbiota and reduce the irritation of metabolites to the mucosa.
Mucosal protectants: Such as sucralfate or montmorillonite powder, can form a protective layer to isolate the drug from the mucosa contact.

Industry regulation and future directions
Formulation improvement
Enteric-coated technology: Through pH-dependent release to avoid drug disintegration in the stomach and reduce upper gastrointestinal tract irritation.
Nanocrystal technology: Reducing the drug particle size to below 200 nm to enhance mucosal permeability while reducing local concentration gradients.
Long-term safety studies
There is currently insufficient human safety data beyond 24 weeks, and large-scale cohort studies are needed to evaluate the correlation between intestinal irritation and the risk of colon cancer.
Patient education
Emphasizing the convenience of "not needing to be taken with water" may mask the risks of medication use, and it is necessary to clearly inform about potential irritation symptoms and response measures.
FAQ
1. Does the tabletting process have any impact on the crystal form of S4?
There is a potential influence. The raw material of S4 may exhibit polymorphism, and the wet granulation or dry pressing processes in the tablet preparation may generate mechanical stress or thermal effects. This physical stress may cause a transformation in the crystal form of S4 (for example, from an unstable form to a stable form), thereby affecting its dissolution rate. Therefore, even with the same dosage, the in vitro release behavior of tablets prepared by different processes may show subtle differences.
2. Will the hardness of the tablets affect their stability?
Yes. The hardness of the tablets directly affects their anti-hygroscopicity. S4 is relatively sensitive to humidity. If the tablet hardness is insufficient or the coating is incomplete, when it is taken out from the frozen storage environment (-20°C), the surface is prone to form a tiny water film due to condensation, resulting in local degradation or the release of excipients. The ideal S4 tablets should have sufficient hardness or adopt moisture-proof coating to resist the brief high-humidity environment during the transition from low temperature to room temperature.
3. Why is it important to focus on the disintegration time of tablets rather than just the content?
For compounds like S4 that exhibit dose-dependent bioavailability, disintegration time is a more sensitive process indicator than content. If the amount of lubricant (such as magnesium stearate) in the tablets is excessive or the mixing time is too long, a hydrophobic film will form, delaying the disintegration of the tablets. Even if the content of S4 in each tablet is fully up to standard, if the disintegration is too slow, it may cause a lag in its release in the research medium, affecting the reproducibility of the data.
4. How accurate is the dosage of the engraved tablets?
Some S4 tablets may have engraved marks for the purpose of dosage research, but there is a risk of uneven distribution of the tablets. The distribution of S4 in the tablets depends on the powder mixing process. If the mixing process is not good, the drug content on both sides of the engraved marks may be inconsistent. Moreover, S4 is usually in a low-dose specification, and掰片 (breaking the tablet) for dosage is prone to cause loss. It is generally recommended to use intact tablets in research to ensure the accuracy of the dosage.
Hot Tags: andarine(s4) tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale










