Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of afoxolaner powder in China. Welcome to wholesale bulk high quality afoxolaner powder for sale here from our factory. Good service and reasonable price are available.
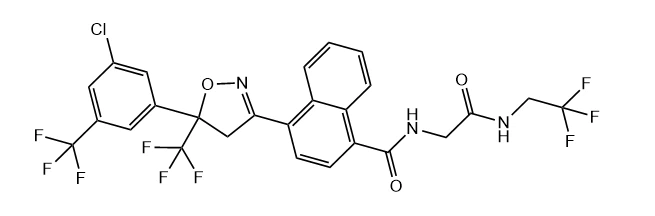
Afoxolaner powder, as a new type of isoxazoline compound, has attracted much attention in the field of veterinary medicine due to its excellent control effect on ectoparasites such as fleas and ticks in dogs and cats. The molecular formula is C26H17ClF9N3O3, CAS 1093861-60-9, and the molecular weight is 625.87 g/mol. Its chemical structure contains an isoxazoline ring core, which is modified with specific substituents (such as 3-chloro-5-trifluoromethylphenyl, trifluoromethyl, trifluoromethylamino) to endow it with unique biological activity. This compound belongs to a white to off white crystalline solid with high melting point characteristics, and exhibits a stable solid state at room temperature.
Our product




Additional information of chemical compound:
| Product Name | Afoxolaner Tablet | Afoxolaner Powder |
| Product Type | Tablet | Powder |
| Product Purity | ≥99% | ≥99% |
| Product Specifications | Customizable | Customizable |
| Product Package | Customizable |
Customizable |
Afoxolaner +. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Afoxolaner | |
|
CAS No. |
1093861-60-9 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|
|
|
||

Afoxolaner powder is an orally active isoxazoline insecticide/acaricide mainly used for the prevention and control of parasites on dogs. Its synthesis methods are diverse, and several common synthesis routes will be introduced below.
(1) Synthesis steps
Aldol condensation:
Starting from 1-naphthalenedione, intermediate I is obtained by aldol condensation reaction with 3,5-dichloro-trifluoroacetophenone, and the yield of this step can reach 82.2%.
Ring reaction:
Intermediate I undergoes ring reaction with hydroxylamine to obtain key intermediate II, with a yield of 87.5% in this step.
Formylation reaction:
Key intermediate II undergoes further formylation reaction to obtain key intermediate III with a yield of 88.4%.
Final reaction:
Intermediate III reacts with 2-amino-N - (2,2,2-trifluoroethyl) acetamide hydrochloride and oxidant to obtain Aflana.
(2) Advantages and disadvantages analysis
Advantages:
This synthetic route starts with common naphthophenone as the raw material, which is relatively easy to obtain. The yield of each step is high, and the overall synthesis efficiency is good, which is conducive to industrial production.
Disadvantages:
The synthesis process involves multiple chemical reactions, requiring precise control of reaction conditions such as temperature, reaction time, reagent dosage, etc., which places high demands on experimental operations. Meanwhile, the oxidants and other reagents used may pose certain risks and require strict safety precautions.
(1) Synthesis steps
Aldol condensation:
Starting from 1- (4-bromo-1-naphthyl) ethanone, it undergoes aldol condensation reaction with 3,5-dichloro-trifluoroacetophenone to obtain intermediate IV (i.e. 1- (4-bromo-naphthyl-1-yl) -3- (3-chloro-5- (trifluoromethyl) phenyl) -4,4,4-trifluoro-2-buten-1-one), with a yield of 85.6%.
Ring reaction:
Intermediate IV undergoes ring reaction with hydroxylamine to obtain the key intermediate 4- {5- [3-chloro-5- (trifluoromethyl) phenyl] -5-trifluoromethyl-4,5-dihydroisoxazole-3-yl} naphthalene-1-carboxaldehyde (intermediate V) with a yield of 83.2%. Further reaction steps may be required to obtain Afurana.




(2) Advantages and disadvantages analysis
Advantages:
The starting material 1- (4-bromo-1-naphthyl) ethanone has certain reactivity, which is beneficial for the subsequent reaction. The yield of aldol condensation and cyclization reactions is also considerable, providing a feasible pathway for the synthesis of Aflana.
Disadvantage:
The price of bromonaphthyl ethyl ketone may be relatively high, which increases the synthesis cost. Moreover, this synthesis route also involves multiple reaction steps, requiring precise control of reaction conditions, and the synthesis process is relatively complex.
(1) A synthetic route starting from 4-acetyl-1-naphthoic acid and 2-amino-N - (2,2,2-trifluoroethyl) acetamide
Condensation reaction:
4-acetyl-1-naphthoic acid and 2-amino-N - (2,2,2-trifluoroethyl) acetamide are condensed to obtain 4-acetyl-N - (2-oxo-2- (2,2,2-trifluoroethyl) amino) ethyl) -1-naphthalenamide.
Subsequent reaction:
The intermediate undergoes a condensation reaction with 1- (3-chloro-5- (trifluoromethyl) phenyl) -2,2,2-trifluoroethyl-1-one under the action of a base to prepare (Z) -4- (3- (3-chloro-5- (trifluoromethyl) phenyl) -4,4,4-trifluorobutyl-2-enoyl) - N - (2-oxo-2- (2,2,2-trifluoroethyl) amino) ethyl) -1-naphthalimide. Finally, the compound was reacted with hydroxylamine sulfate to prepare Aflana.
(2) A synthetic route starting from nitromethane intermediate and 1-chloro-3- (trifluoromethyl) -5- (3,3,3-trifluoropropy-1-en-2-yl) benzene compound
Dipole cycloaddition reaction: Nitromethane intermediate undergoes dipole cycloaddition reaction with 1-chloro-3- (trifluoromethyl) -5- (3,3,3-trifluoropropy-1-en-2-yl) benzene compound. The starting material nitromethane compound is prepared by coupling reaction of bromobenzene derivatives and nitromethane under the action of palladium catalyst and ligand.

Physical state and appearance characteristics:
Afoxolaner powder appears as white to off white crystalline powder under standard conditions, which makes it easy to mix with other excipients in formulation development. Its crystal morphology may be further characterized by X-ray diffraction (XRD) to determine crystal stability. In the production of formulations, the particle size distribution of powders needs to be strictly controlled to ensure the uniform dispersion of drugs in chewable tablets. For example, NexGard chewable tablets are made into light red to reddish brown circular or square tablets using a specific process to ensure drug stability and enhance palatability.
Solubility and solvent selectivity:
The water solubility of Aflana is extremely low, with a solubility of only 3.4 × 10 ⁻⁵ g/L at 25 ℃, which limits the development of its water-based formulations. However, it exhibits good solubility in organic solvents, especially in dimethyl sulfoxide (DMSO) where the solubility can reach ≥ 250 mg/mL (399.44 mM). This characteristic provides convenience for laboratory research, for example, in pharmacokinetic experiments, DMSO is often used as a solvent to prepare stock solutions. In addition, further research is needed on the solubility in solvents such as ethanol and acetone to optimize the formulation process.
Thermal stability and storage conditions:
The thermal stability study of Aflana shows that it can maintain chemical stability for a long time at room temperature, but degradation may occur under high temperature or light conditions. Therefore, it is recommended to store at -20 ℃ or 2-8 ℃ in the dark to prevent loss of active ingredients. In the stability test of the formulation, it is necessary to monitor the changes in its content at different temperatures (such as 40 ℃/75% RH accelerated test) to ensure the quality of the product within its shelf life. For example, the expiration date of Nicosin chewable tablets is 24 months, and its sealed packaging design can effectively isolate moisture and oxygen, extending drug stability.
PH and pKa values:
The acidity coefficient (pKa) of Aflana is 12.45 ± 0.46 (predicted value), indicating that it exists in anionic form under physiological pH conditions. This characteristic affects its absorption mechanism in the gastrointestinal tract, for example, in dogs, Aflana enters the bloodstream through passive diffusion and carrier mediated transport. Its high pKa value also explains its stability in acidic environments (such as gastric juice), but ionization may occur under alkaline conditions, and the use of pH regulators needs to be considered in formulation design.
Spectral characteristics and identification methods:
The InChIKey of Afrana is OXDDDHGGRFRLEE-UFFAOYSA-N, and the SMILES code is C1(C(NCC(=O)NCC(F)(F)F)=O)=C2C(C=CC=C2)=C(C2CC(C3=CC(C(F)(F)F)=CC(Cl)=C3)(C(F)(F)F)ON=2)C=C1. These unique identifiers can be used for database retrieval and structure validation. Its UV visible absorption spectrum has characteristic absorption peaks at specific wavelengths, which can be used for content determination. For example, high-performance liquid chromatography (HPLC) is commonly used to detect purity (≥ 98%) and ensure the quality of raw materials.
Physical properties related to the formulation:
In formulation development, the physical properties of Aflana directly affect process parameters. For example, its high melting point requires temperature and pressure control during the compression process to avoid crystal transformation. The flowability of its powder needs to be improved by adding flow aids (such as microcrystalline cellulose) to ensure that the weight difference of the tablets meets pharmacopoeia standards. In addition, studies on the compatibility of Afurana with excipients have shown that it does not interact with commonly used excipients such as starch and lactose, but should avoid contact with metal ions to prevent catalytic degradation.
The correlation between physical properties and pharmacological effects:
The physical properties of afoxolaner powder are closely related to its pharmacological effects. For example, its low water solubility may lead to slow release in the gastrointestinal tract, prolonging the duration of drug efficacy (such as the monthly efficacy of Nissin). Its crystal morphology affects the dissolution rate, which in turn affects the peak time of blood drug concentration (Tmax=2-4 hours). In addition, its binding affinity with GABA receptors and glutamate gated chloride ion channels may be influenced by molecular conformation, and conformational stability is related to its physical state.
Hot Tags: afoxolaner powder, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 4 Fluorotropacocaine, Articaine Hcl powder, epinephrine powder, Larocaine powder, sapropterin dihydrochloride powder, tetracaine hcl