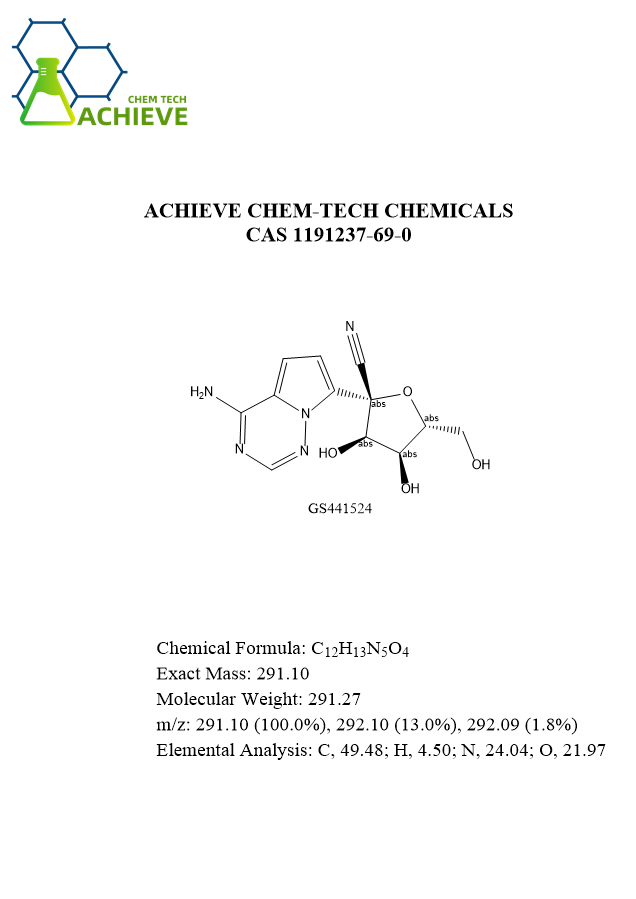
GS-441524 fip is a specific drug for feline infectious peritonitis. This disease is a highly lethal feline disease caused by mutations in feline enterocoronavirus (FCOV). The characteristics of this disease are fluid accumulation in the body cavity, granulomatous vasculitis, and organ damage. Clinical manifestations include weight loss, loss of appetite, fever, jaundice, and ascites. Due to the lack of effective treatment methods, the prognosis of traditional treatment plans is extremely poor, and most sick cats die or are euthanized within a few months after diagnosis. It has made revolutionary breakthroughs in treatment, significantly improving the survival rate of diseased cats by inhibiting the activity of viral RNA dependent RNA polymerase.

It is mainly transmitted through the fecal oral route. During the infection process, FCOV replicates within intestinal epithelial cells, and some viruses may mutate to acquire the ability to cross species infect monocytes and macrophages, leading to FIP. According to pathological manifestations, FIP can be divided into two types: exudative (wet) and non exudative (dry). Wet FIP is characterized by fluid accumulation in the body cavity, while dry FIP presents with granulomatous vasculitis and organ damage. In a multi cat environment, the infection rate of FCoV can be as high as 90%, but the incidence rate of FIP is usually less than 10%. Kittens, immunocompromised cats, and genetically susceptible cats (such as purebred cats) have a higher risk of developing the disease.
At the same time, our company not only provides GS-441524 pure powder, but also offers injections and tablets. If needed, please feel free to contact us.
|
|
Client Feedbacks
 |
 |
 |
About Shipping

Successfully delivery all over the world:
https://www.bloomtechz.com/news/the-shipping-information-of-100-tablets-qty-gs-85323689.html

GS-441524 powder COA


GS441524 fip has shown good efficacy in the treatment of viral infections. However, the preparation process of GS441524 is complex and requires strict control over each step to ensure the quality and stability of the injection. The following is the preparation process:
Raw materials and equipment for preparation
Raw material selection
GS441524 raw materials should be selected from high-purity and stable products to ensure the quality of the injection. The source of raw materials should be reliable, preferably purchased from formal channels, and avoid using materials of unknown origin.

Solvent selection
The choice of solvent is crucial for the solubility and stability of GS441524. Common solvents include 5% ethanol, 30% propylene glycol, 45% PEG400, and 20% water (pH 1.5, containing HCl). The selection of these solvents is based on the solubility, stability, and biocompatibility of GS441524 injection.

Equipment preparation
Electronic balance: used for precise weighing of GS441524 raw materials and solvents.
Magnetic stirrer: used for stirring solutions to ensure that the raw materials are fully dissolved.
Ultrasonic cleaner: used for ultrasonic treatment to improve the solubility and stability of drugs.
Aseptic filter: used to filter solutions and ensure the sterility of injections.
Injection bottles and sealing machines: used for packaging and encapsulating injections.

Preparation steps
| Raw material preparation | Accurately weigh the required amount of GS441524 raw material according to the preparation requirements. Select suitable solvents such as 5% ethanol, 30% propylene glycol, 45% PEG400, and 20% water (pH 1.5, containing HCl) for diluting GS441524. |
| Dilution and ultrasonic treatment | Place the weighed GS441524 raw material in a sterile container, add an appropriate amount of solvent, and stir until completely dissolved. Then place the dissolved solution in an ultrasonic water bath for 5-20 minutes of ultrasonic treatment to improve the solubility and stability of the drug. |
| Aseptic filtration | Select a suitable sterile filter based on the characteristics of the solution and sterile requirements, such as a 0.22 μ m microporous membrane. Filter the solution after ultrasonic treatment through a sterile filter to remove impurities and microorganisms from the solution, ensuring the sterility of the injection. |
| Packaging and packaging | Select sterile 50ml glass injection bottles as packaging containers to ensure the sealing and sterility of the injection bottles. The filtered solution can be divided into injection bottles according to the specified volume (such as 10ml or 15ml). Use appropriate sealing materials (such as rubber stoppers and aluminum caps) to encapsulate the injection bottle, ensuring the sealing and stability of the injection. |
| Storage conditions | Store the packaged GS441524 injection in a refrigerated environment to prevent drug degradation. It determines the expiration date of the injection based on the stability of the drug and experimental data, and clearly indicates it on the label. |
Possible problems and solutions during the preparation process

Solubility issue
It has low solubility in certain solvents, which may result in uneven drug concentration. Suitable solvent combinations can be selected, such as a mixed solvent of 5% ethanol, 30% propylene glycol, 45% PEG400, and 20% water, to enhance the solubility of the drug. During the dissolution process, methods such as ultrasonic treatment or heating are used to promote the dissolution of the drug.
Aseptic issues
During the preparation process, if the aseptic operation is not strict, it may lead to microbial contamination of the injection, affecting the safety and efficacy of the drug. During the preparation process, strict adherence to sterile operating procedures and the use of sterile equipment and solvents can be ensured. Then perform aseptic testing on the prepared injection to ensure that it meets the aseptic requirements.


Stability issues
The stability of GS-441524 may vary under different conditions, such as light exposure, temperature, pH value, and other factors that may affect the stability of the drug. During the preparation process, the pH value of the solution can be strictly controlled, usually adjusted to around 1.5, and an appropriate amount of hydrochloric acid (HCl) can be added to stabilize the pH value. Store the prepared injection under dark and constant temperature refrigeration conditions to extend its shelf life. Add an appropriate amount of stabilizers, such as antioxidants, metal chelating agents, etc., to the injection to improve the stability of the drug.
Pollution issues during packaging and packaging processes
Improper handling during packaging and packaging may result in contamination of the injection by microorganisms or foreign objects. Strict adherence to sterile operating procedures and the use of sterile equipment and containers can be ensured during the packaging and packaging process. Strict quality testing is conducted on packaged and encapsulated injections to ensure their sterility and stability.

What are the advantages and disadvantages of GS-441524 injections, tablets, and powders
GS-441524 FIP is currently available in various dosage forms on the market, including injections, tablets, and powders. There are differences in preparation process, stability, administration method, bioavailability, and safety among different dosage forms. The following will provide a detailed analysis of these three dosage forms:
Comparison of key parameters for different dosage forms
| Parameter | Injection | Tablet | Powder |
| Method of administration | Subcutaneous/intravenous injection | Take orally | Subcutaneous injection after dissolution |
| Bioavailability | High (close to 100%) | Low (10% -20%) | Depends on the dissolution process |
| Onset | Quick (within a few hours) | Slow (several hours to several days) | Depends on dissolution and injection speed |
| Storage conditions | Refrigeration (2-8 ℃) | Avoid light at room temperature | Avoid light at room temperature |
| Stability | Tall | Medium | Depends on packaging and storage conditions |
| Safety | Injection related risks (pain, ulceration) | Gastrointestinal irritation | Prepare pollution risk |
| Scope of application | Critical condition, unable to take orally | Long term treatment and good compliance of affected cats | Economically limited, requiring flexible adjustment of dosage |
Prevention and prognosis
Preventive Measure
Reduce stress: Avoid moving, changing food, introducing new cats and other sources of stress.
Boosting immunity: Regularly supplement with lactoferrin (100mg/day) and probiotics (such as Saccharomyces boulardii).
Disinfection and isolation: FCOV can be transmitted through feces, and it is necessary to clean the litter box daily and disinfect it with a chlorine containing disinfectant (such as 84 disinfectant diluted 1:100).
Prognosis
If treated promptly, the 3-month survival rate of wet FIP can reach 60% -70%;
The prognosis of dry FIP is poor, with a 3-month survival rate of less than 40%;
The prognosis of brain/eye type FIP is the worst, with a 3-month survival rate of only 10% -20%.
The Future of FIP Treatment: Ongoing Research and Innovations
The success of antiviral therapies in treating FIP has opened the door to further research and innovations. Ongoing clinical trials are exploring new drugs, combination therapies, and optimized treatment protocols to improve survival rates and minimize side effects.
Vaccine Development
While vaccines for FIP have had limited effectiveness due to the complexity of FCoV mutation and immune response, research continues to refine vaccine strategies. The development of mRNA vaccines, which have shown promise in other infectious diseases like COVID-19, offers hope for a future FIP vaccine.
Supportive Care
In addition to antiviral therapy, supportive care plays a crucial role in managing FIP. This includes nutritional support, pain management, and treatment of secondary infections. A holistic approach to care ensures that cats receive the best possible chance of survival and recovery.
The landscape of FIP treatment has undergone a remarkable transformation in recent years. From a once-fatal disease to a manageable condition, the success stories of FIP survivors like Bart and Nutter Butter serve as a testament to the power of scientific innovation and perseverance. With ongoing research and a growing arsenal of antiviral therapies, the future looks bright for cats diagnosed with FIP. While challenges remain, the progress made thus far offers hope and healing to countless feline lives around the world.
Frequently Asked Questions
What does GS-441524 treat?
+
-
Feline infectious peritonitis (FIP) is a severe viral disease with a very high fatality rate. GS-441524 is an adenosine analogue that acts as an antiviral and has shown promise in FIP treatment.
How quickly does GS-441524 work?
+
-
This medication should take effect quickly and improvement in clinical signs may be seen in as few as 2 to 5 days with obvious improvement in 1–2 weeks.
Has any cat ever survived FIP?
+
-
Meet the cats who have overcome FIP, Feline Infectious Peritonitis. These cats and their caregivers have incredible stories of survival after facing a once-incurable disease. Over 15,000 cats have already been treated.
Hot Tags: gs-441524 fip, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 5 chloro 2 3 difluoroterephthalonitrile, 3H Indole 3 hexanoic acid 2 3 dimethyl ethyl ester, DERMORPHIN, 1-bromo-5-chloronaphthalene, CAS 4786 80 5, CAS 2130009 90 2