Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of hexafluorobisphenol a cas 1478-61-1 in China. Welcome to wholesale bulk high quality hexafluorobisphenol a cas 1478-61-1 for sale here from our factory. Good service and reasonable price are available.
Hexafluorobisphenol A (HFBA) is an important fluorinated aromatic compound. Its chemical structure is similar to that of bisphenol A, but the hydrogen atoms on the benzene ring are replaced by six fluorine atoms, resulting in a highly fluorinated characteristic. This compound is usually a white crystalline solid and has excellent thermal stability and chemical inertness, which is mainly attributed to the strong electronegativity of the fluorine atoms and the high bond energy of the carbon-fluorine bond. HFBA is a key monomer for synthesizing high-performance polymers, especially for preparing special engineering plastics such as polyimides, polyesters, and epoxy resins. These materials can still maintain excellent mechanical strength and insulation properties under high temperatures, strong corrosive environments, or extreme conditions. Additionally, it is also applied in the fields of coatings, adhesives, and electronic packaging materials to enhance the product's weather resistance, hydrophobicity, and flame retardancy. Although HFBA plays a significant role in industry, its environmental persistence and potential bioaccumulation have also raised concerns. Strict environmental protection regulations must be followed during production and handling. In summary, HFBA, through the unique properties of the fluorine atoms in its molecule, has driven the development of advanced materials science and has become an indispensable functional chemical in the high-tech industry.

|
|
|
|
Chemical Formula |
C15H10F6O2 |
|
Exact Mass |
336 |
|
Molecular Weight |
336 |
|
m/z |
336 (100.0%), 337 (16.2%), 338 (1.2%) |
|
Elemental Analysis |
C, 53.58; H, 3.00; F, 33.90; O, 9.52 |

Hexafluorobisphenol A, also known as Bisphenol AF, is a type of bisphenol compound containing a hexafluororing. It has various unique physical and chemical properties and is widely used in multiple industrial fields.
1. Fluororubber vulcanization accelerator:
It is an important vulcanization accelerator (crosslinking agent) for fluororubber, with over 70% of fluororubber using the bisphenol AF vulcanization system. This vulcanization system can endow fluororubber with excellent physical properties and chemical stability, enabling it to maintain good performance in harsh environments such as high temperature, high pressure, and strong corrosion.
2. Polymer material synthesis:
It can be used as a monomer to synthesize various fluorinated polymer materials, such as fluorinated polyamide, fluorinated polyester, fluorinated polyarylether, fluorinated polyether ketone, fluorinated polycarbonate, fluorinated epoxy resin, fluorinated polyurethane, etc. These polymer materials have excellent heat resistance, corrosion resistance, wear resistance, and insulation properties, and are widely used in fields such as microelectronics, fuel cells, optics, and space technology.
3. Gas separation membranes and proton exchange membranes:
It can also be used to manufacture new materials such as gas separation membranes and proton exchange membranes. These materials have broad application prospects in fields such as energy, environmental protection, and chemical engineering, such as hydrogen separation and proton conduction in fuel cells.
4. Dielectric coatings and fiber optic sheaths:
Their insulation properties and chemical stability make them suitable for manufacturing materials such as dielectric coatings and fiber optic sheaths. These materials play an important role in fields such as electronics and communication, protecting electronic devices and optical fibers from external environmental influences.
5. Adhesive:
It can also be used as a raw material for adhesives to manufacture various high-strength and high weather resistant adhesives. These adhesives have wide application value in fields such as aerospace, automotive manufacturing, and construction.

Hexafluorobisphenol A can be used as a monomer to synthesize fluorinated polyimides, fluorinated polyesters, fluorinated polyarylethers, fluorinated polyether ketones, fluorinated polycarbonates, fluorinated epoxy resins, fluorinated polyurethanes, and other fluorinated polymers. It is widely used in gas separation membranes, proton exchange membranes, dielectric coatings, fiber optic sheaths, photovoltaic cell substrates, adhesives, and other fields such as microelectronics, fuel cells, optics, and space technology. Bisphenol AF modified heterocyclic special polymers can achieve higher thermal stability, oxidation resistance, transparency, low dielectric constant, and water absorption. Bisphenol AF can also be used as an intermediate in organic synthesis.
In a pressure vessel, heating hexafluoroacetone and phenol for catalytic condensation reaction produces bisphenol AF, which is the traditional synthesis method of bisphenol AF. The HF used in the early stages of this method is both a solvent and a catalyst, with a reaction temperature of around 100 ℃ and a controlled pressure of 0.8-1.0 MPa. It requires special equipment such as pressure vessels and refrigeration devices, which have strict requirements for process technology and equipment, and cause serious acid pollution, making it unsuitable for large-scale industrial production. HF is highly toxic, corrosive, and dangerous, making it difficult to recycle and reuse.
Boron trifluoride, methylsulfonic acid, or trifluoromethanesulfonic acid can also be used as reaction solvents or catalysts. Boron trifluoride is a colorless gas with a suffocating and irritating odor, moderate toxicity, strong corrosiveness, and can corrode glass even when cold. It is thermally stable and undergoes explosive decomposition when in contact with water, producing boric acid and hydrogen fluoride.
Methyl sulfonic acid is a colorless and transparent liquid with strong acidity and high catalytic activity. It has attracted attention as a homogeneous catalyst, but has weak ability to oxidize organic compounds, low toxicity, and less corrosiveness than mineral acids. It is easy to separate from reaction mixtures and can be reused. Methanesulfonic acid has a low cost of use, can reduce the occurrence of side reactions, and can lower product color, making it an environmentally friendly catalyst. Trifluoromethane sulfonic acid does not free fluoride ions and has the function of a halogen-free liquid organic superacid. Hexafluoroacetone is a highly toxic gas that causes inconvenience in operation.
Hexafluoroacetone trihydrate has much lower toxicity, and stable trihydrate supplied to the market is usually selected. Concentrated acid can dehydrate the hydrate and convert it into hexafluoroacetone. The reaction equation is as follows:
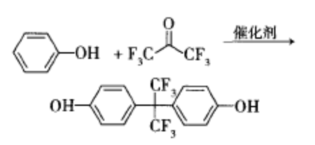
In a pressure vessel, using HF as the solvent and catalyst, hexafluoroepichlorohydrin is isomerized into hexafluoroacetone, which reacts directly with phenol without purification to obtain bisphenol AF. The toxicity of the raw material hexafluoroepichlorohydrin in this method is relatively low, but it uses HF as a solvent and catalyst, requires reaction in an autoclave, and needs to be equipped with a refrigeration device. The operation is cumbersome and has certain risks, and good sealing measures need to be taken. This method is similar to the catalytic condensation of phenol and hexafluoroacetone, but has a higher industrial cost and is not conducive to large-scale production.
The Lewis acid catalyst for hexafluoropropane isomerization can be selected from A1C13, Cr2O3, A12O3, A1F3, etc. Cr2O3 or mixed catalysts mainly composed of Cr2O3 are particularly effective, with high activity, good selectivity, and long catalyst life. Using anhydrous HF to treat Cr O as a catalyst and running continuously for 1000 hours, hexafluoroepichlorohydrin can be almost quantitatively converted to hexafluoroacetone. The reaction equation is as follows:
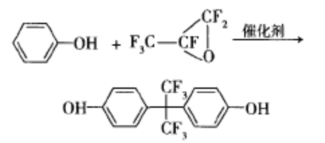
In a 250 mL autoclave, Rammell, Peter Paul, and others shaken 100 g of hexafluoroepichlorohydrin and 100 g of HF at 100 ℃ for 24 hours to obtain hexafluoroacetone, with a conversion rate and selectivity of 98.9%. In the presence of phenol, bisphenol AF is obtained through reaction.
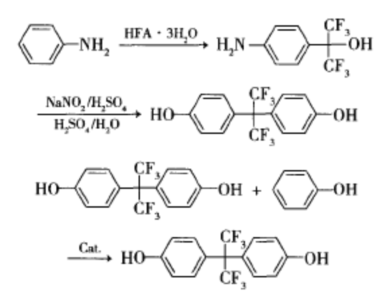
The 4-step synthesis method uses hexafluoroacetone trihydrate and aniline as raw materials, and synthesizes bisphenol AF through condensation, diazotization, hydrolysis, and Friedel Crafts alkylation in 4 steps at atmospheric pressure. A new method for the synthesis of bisphenol AF has been established by optimizing the synthesis conditions of intermediates.
This synthesis process is environmentally friendly, with mild reaction conditions, good selectivity, and high yield, avoiding the need for industrial high-pressure equipment and refrigeration devices. The reaction intermediates 4-amino-phenylhexafluoroisopropanol and 4-hydroxyhexafluoroisopropanol are widely used in many fields such as medicine and synthetic materials. This method has easy access to raw materials, low toxicity, and is easy to operate. Efforts have been made in raw material matching, catalyst research and development, and product separation and purification to reduce costs. Multiple products can be co produced, making it more competitive in the market. The reaction equation is as follows:
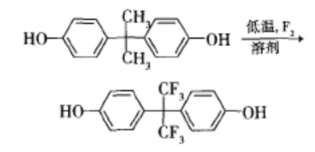
Low temperature fluorination of bisphenol A in an appropriate solvent yields bisphenol AF. The preparation process is simple, with low energy consumption, minimal pollution, high efficiency, and has good market prospects. This method provides a new approach for the preparation of bisphenol AF. The reaction equation is as follows:
The refining of reaction intermediates and bisphenol AF is a key technology in the production of bisphenol AF. Common refining measures include dissolution, filtration, distillation, extraction, decolorization, crystallization, and recrystallization. Dissolve in sodium hydroxide solution in the reaction product of phenol and hexafluoroacetone, filter, impurities containing iron hydroxide and tar.
Dilute the filtrate with water, then treat it with hydrochloric acid solution, and control the pH to 7-9 at 25 ℃ to obtain refined bisphenol AF with a purity of 99.8% and an absorbance difference AABS of 0.025 between 555-700 nm. Heat bisphenol AF and water, then cool the solution to obtain precipitated bisphenol AF; Or heat bisphenol AF and water to a temperature above 90 ℃, separate the liquid phase from the molten bisphenol AF phase, and cool the liquid phase to obtain precipitated bisphenol AF.

Hexafluorobisphenol A has the following characteristics and applications:
High temperature stability:
It has excellent high temperature stability and can maintain its chemical stability and physical properties for a long time in high temperature environments, suitable for applications in high temperature processes and environments.
Chemical inertness:
It has high chemical inertness, is resistant to many chemicals, is not easily corroded and dissolved, and can be used as a storage container and pipeline material for chemicals, solvents, etc.
Wear resistance:
It has high wear resistance and can be used to manufacture wear-resistant and friction resistant components and lubricating materials, such as bearings, seals, etc.
Insulation performance:
It has good insulation performance and can be used to manufacture electrical insulation materials, such as wires and cables, insulators, etc.
Biocompatibility:
It has good biocompatibility and can be used as a raw material for medical devices and biomedical materials, such as artificial joints, implants, etc.
Environmental friendliness:
It has good environmental performance, is not easy to degrade, and can be used as a raw material for environmentally friendly materials, such as filters, isolation membranes, etc.
Bisphenol AF, also known as hexafluorobisphenol A, 2,2-bis (4-hydroxyphenyl) hexafluoropropane, 2,2-bis (4-hydroxyphenyl) hexafluoropropane, etc., is abbreviated as BPAF. It is slightly soluble in carbon tetrachloride, difficult to dissolve in water, and easily soluble in organic solvents such as ethanol, acetone, ether, toluene, and strong alkaline solutions. Bisphenol AF can decompose and burn when heated to a temperature of 510 ℃.
Bisphenol AF dissolves in dilute alkali to form corresponding metal salts, which can undergo nitration reaction, nitrosation reaction, Friedel Crafts alkyl substitution reaction, halogenation reaction, carboxylation reaction, esterification reaction. It can be condensed with acetone to form polyphenols, condensed with excess formaldehyde in alkaline medium to form phenolic esters, and condensed with fatty alcohols, epoxy polymers, epichlorohydrin, etc. to form corresponding ethers. Bisphenol AF is widely used, but there are relatively few reports on its synthesis due to safety, environmental, cost, and technical confidentiality issues. The synthesis methods of bisphenol AF include catalytic condensation of phenol and hexafluoroacetone, catalytic condensation of phenol and hexafluoroepichlorohydrin, condensation, diazotization, hydrolysis, alkylation of aniline and hexafluoroacetone, and low-temperature fluorination of bisphenol A.
There is still great potential to optimize the production process of bisphenol AF. Improving the existing synthesis process, selecting suitable solvents and catalysts, shortening reaction time, controlling system side reactions, reducing the amount of by-products generated, simplifying operations, refining products and recycling by-products can save production materials, reduce production costs, protect the ecological environment, improve the quality and yield of bisphenol AF, and have a positive impact on enhancing product competitiveness, creating huge economic and social benefits. With the research, development, and large-scale application of fluororubber and fluorinated polymers, bisphenol AF is playing an increasingly important role, and the demand is constantly increasing. It is still necessary to improve the research and development of the synthesis process of bisphenol AF, product refining, and by-product recovery and treatment.

The development prospects of hexafluorobisphenol A (HFBPA or BPAF) are influenced by various factors, including market demand, technological progress, policy environment, etc. The following is a detailed analysis of its development prospects:
Technological progress and industrial upgrading
Production process optimization
With the advancement of technology and the upgrading of industries, the production process of HFBPA will be continuously optimized and improved. Enhance market competitiveness through measures such as improving production efficiency, reducing production costs, and improving product quality.
New catalyst and reaction conditions
The development of new catalysts and reaction conditions will help improve the yield and selectivity of HFBPA, reduce production costs and environmental impact. This will provide strong support for the widespread application of HFBPA.
Extension of industrial chain and coordinated development
The collaborative development between upstream and downstream enterprises in the HFBPA industry chain will help improve the competitiveness of the entire industry chain. By strengthening cooperation in raw material supply, product development, marketing, and other aspects, resource sharing and complementary advantages can be achieved to promote the rapid development of the HFBPA industry.
Development prospects and outlook
The market size continues to expand
With the rapid development of downstream industries such as fluororubber and semiconductors, and the continuous expansion of emerging application fields, the market size of HFBPA will continue to expand. It is expected that the market demand for HFBPA will maintain a stable growth trend in the coming years.
Technological innovation and industrial upgrading
Technological innovation and industrial upgrading are important driving forces for the development of the HFBPA industry. Enterprises need to strengthen their technological research and innovation capabilities, promote the optimization and improvement of the production process of hexafluorobisphenol A, as well as the research and application of new catalysts and reaction conditions.
Environmental Protection and Sustainable Development
Environmental protection and sustainable development will become important directions for the development of the HFBPA industry. Enterprises need to strengthen the construction and management of environmental protection facilities to achieve green production; At the same time, actively exploring sustainable development models to promote the sustainable development of the HFBPA industry.
International Development and Cooperation
With the deepening of globalization, the HFBPA industry will pay more attention to international development and cooperation. Enterprises need to strengthen their connections and cooperation with the international market, expand international market channels and product application areas; Actively participate in the formulation and implementation of international standards and rules, and enhance international competitiveness.
Hot Tags: hexafluorobisphenol a cas 1478-61-1, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 3 bromobenzo b naphtho 2 3 d thiophene, 1 3 5 dimethylphenyl 6 1 methylethyl isoquinoline, 2 chlorospiro fluorene 9 9 xanthene 7 carbonitrile, CAS 1115639 92 3, CAS 553663 65 3, 2 4 chlorophenyl 4 dibenzo b d furan 3 yl 6 phenyl 1 3 5 triazine






