Trimethyl Phosphate, also known as triethyl phosphate, is an important organic phosphorus compound with the chemical formula C₃H₉O₄P or (CH₃O)₃P=O. At room temperature, it typically appears as a colorless transparent liquid with a faint odor and good water solubility and mutual solubility with organic solvents. Its main chemical functions are as a methylating reagent, a flame retardant, and a polar non-polar solvent in chemical reactions. In the field of organic synthesis, it can provide methyl groups to target molecules and is often used to prepare drug and pesticide intermediates; as a flame retardant, it acts on plastic and resin materials through the gas-phase flame retardation mechanism; and in lithium-ion battery electrolytes, it is studied as an efficient flame retardant additive to enhance battery safety. Despite its wide range of applications, it should be noted that it has certain toxicity and may have an impact on the nervous system. Therefore, strict ventilation and protective measures are required during industrial production and laboratory operations.

|
Chemical Formula |
C3H9O4P |
|
Exact Mass |
140 |
|
Molecular Weight |
140 |
|
m/z |
140 (100.0%), 141 (3.2%) |
|
Elemental Analysis |
C, 25.72; H, 6.48; O, 45.69; P, 22.11 |


Trimethyl Phosphate,which is an important organic compound. It has unique chemical and physical properties, which make it widely used in multiple fields. The following is a detailed description of its purpose:
Application In The Fields Of Medicine And Pesticides
Triethyl phosphate is commonly used as a solvent in pharmaceutical and pesticide production. In the process of drug synthesis, many reactions need to be carried out in specific solvents to ensure the smooth progress of the reaction and the purity of the product. Triethyl phosphate has good solubility and can dissolve various organic compounds, so it is widely used in drug synthesis reactions. For example, in the intermediate synthesis process of some drugs, trimethyl phosphate can be used as a solvent to promote contact and reaction between reactants, thereby improving reaction efficiency.In pesticide production, trimethyl phosphate can also be used as a solvent to dissolve the active ingredients and other additives of pesticides. It can help pesticides disperse and dissolve better, improve the stability and effectiveness of pesticide use. For example, in the production of some insecticides and herbicides, trimethyl phosphate can be used as a solvent to evenly disperse pesticide components in the formulation, thereby improving the control effect of pesticides.

As an extractant

In addition to serving as a solvent, trimethyl phosphate can also be used as an extractant for separating and purifying target compounds from mixtures. In the fields of medicine and pesticides, it is often necessary to extract specific active ingredients from complex mixtures. Triethyl phosphate has good selectivity and extraction ability, which can selectively extract target compounds while reducing the content of impurities.For example, in the process of drug extraction, trimethyl phosphate can be used to extract components with medicinal value from plant extracts. It can form a complex with the target compound, thereby achieving the separation of the target compound from other components. In pesticide production, trimethyl phosphate can also be used to extract the active ingredients of pesticides from reaction products, improving product purity and quality.
Application In The Field Of Organic Synthesis
In organic synthesis reactions, the choice of solvent has a significant impact on the progress of the reaction and the generation of products. As an organic solvent, trimethyl phosphate has good solubility and stability, and can dissolve various organic compounds, providing a suitable reaction environment for organic synthesis reactions.For example, in some organic synthesis reactions such as esterification and condensation, trimethyl phosphate can be used as a solvent to promote contact and reaction between reactants. It can reduce the viscosity of reactants, increase their diffusion rate, and thus accelerate the reaction rate. Meanwhile, trimethyl phosphate can also stabilize the reaction system, reduce the occurrence of side reactions, and improve the purity and yield of the product.

As a catalyst

Triethyl phosphate can also serve as a catalyst in certain organic synthesis reactions. It can promote the progress of the reaction, reduce the activation energy of the reaction, and increase the reaction rate. For example, in some polymerization reactions, trimethyl phosphate can serve as a catalyst to promote monomer polymerization and generate high molecular weight polymers.In addition, trimethyl phosphate can also be used in combination with other catalysts to improve catalytic performance. For example, in some oxidation reactions, trimethyl phosphate can be used in combination with transition metal catalysts to promote the oxidation reaction of organic compounds and generate corresponding oxidation products.
Triethyl phosphate itself contains ester groups, so it can be used as an esterification agent in some esterification reactions. It can undergo esterification reaction with alcohol compounds to generate corresponding ester compounds. For example, trimethyl phosphate can react with alcohol compounds such as methanol and ethanol to produce ester compounds such as dimethyl phosphate and diethyl phosphate.These ester compounds have a wide range of applications in organic synthesis, pharmaceuticals, pesticides, and other fields. For example, dimethyl phosphate can be used as a solvent, plasticizer, etc; Diethyl phosphate can be used as a fuel additive, lubricant additive, etc.

Application In The Field Of Analytical Chemistry
Triethyl phosphate can be used as a reagent for the determination of zirconium. Accurately determining the content of metal elements in analytical chemistry is of great significance for fields such as materials science and environmental monitoring. Triethyl phosphate can form stable complexes with zirconium ions, and the content of zirconium can be indirectly determined by measuring the properties of the complexes.For example, in some ore analysis, the complexation reaction between trimethyl phosphate and zirconium ions can be used to determine the zirconium content in the ore using methods such as spectrophotometry and atomic absorption spectroscopy. This method has the advantages of high sensitivity and good selectivity, and can accurately determine the content of zirconium.

As a solvent and extractant

In analytical chemistry, trimethyl phosphate can also be used as a solvent and extractant for sample pretreatment and separation. For example, in the analysis of some environmental samples, the samples may contain multiple metal ions and organic compounds that need to be separated and enriched. Triethyl phosphate can be used as an extractant to selectively extract target metal ions or organic compounds, thereby achieving sample separation and enrichment.Meanwhile, trimethyl phosphate can also serve as a solvent to dissolve some compounds that are difficult to dissolve in water, providing a suitable solution environment for subsequent analysis and determination. For example, in the analysis of some organic pollutants, trimethyl phosphate can be used as a solvent to dissolve the organic pollutants, and then analyzed and determined using methods such as gas chromatography and liquid chromatography.
Triethyl phosphate can also be used as a gas chromatography stationary phase. Gas chromatography is a commonly used analytical separation technique, widely used in fields such as chemistry, biology, and environment. Fixed liquid is one of the core components of gas chromatography, which can selectively adsorb and desorb components in the sample, thereby achieving component separation.Triethyl phosphate, as a gas chromatography stationary phase, has good separation performance and stability. It can separate various organic compounds, such as alcohols, aldehydes, ketones, etc. For example, in the analysis of some volatile organic compounds, trimethyl phosphate can be used as a gas chromatography stationary phase to achieve accurate separation and quantitative analysis of volatile organic compounds.

Applications In Other Fields
With the increasing application of lithium-ion batteries in fields such as electric vehicles and energy storage, the safety of batteries has become a focus of attention. Triethyl phosphate can be used as a flame retardant additive for lithium-ion batteries to improve their safety. When the battery experiences abnormal conditions such as overheating or short circuit, trimethyl phosphate can decompose to produce non combustible gases, suppress the spread of flames, and thus reduce the risk of battery fire and explosion. Meanwhile, trimethyl phosphate can also improve the electrochemical performance of batteries, increase their cycle life and charge discharge efficiency.

As a solvent for paints, coatings, and plastics&As an additive for lubricating oil and fire retardant

Triethyl phosphate can be used as a solvent for paints, coatings, and plastics to improve their flowability and processability. In the production of paints and coatings, trimethyl phosphate can better disperse and dissolve pigments and resins, improving the quality and performance of paints and coatings. In the production of plastics, trimethyl phosphate can be used as a solvent to promote the molding and processing of plastics. For example, in the production of some polyvinyl chloride (PVC) plastics, trimethyl phosphate can be used as a solvent for plasticizers to better mix with PVC resin, improving the flexibility and processability of the plastic.
Triethyl phosphate can also be used as an additive in lubricants and fire retardants. In lubricating oil, trimethyl phosphate can improve the anti-oxidation and anti-wear properties of lubricating oil, and extend the service life of lubricating oil. It can work synergistically with other additives in lubricating oil to form a protective film, reducing wear and friction of mechanical parts. In fire retardants, trimethyl phosphate can play a flame retardant role. It can decompose to produce substances such as phosphoric acid, promote the formation of a carbon layer, and prevent the spread of flames. Meanwhile, trimethyl phosphate can also reduce the viscosity of the fireproof agent, improve the spraying performance and fireproof effect of the fireproof agent.

The synthetic method of trimethyl phosphate:
1. Phosphorus oxychloride reacts with methanol in the presence of potassium carbonate to generate it. Simultaneously react to generate dimethyl phosphate potassium salt, then react with dimethyl sulfate to generate it. The crude product of it is washed with water, decolorized, dehydrated and distilled under reduced pressure to obtain the finished product. Raw material consumption quota: phosphorus oxychloride 1094kg/t, methanol 686kg/t.
2. Add methanol and potassium carbonate to the reaction pot, cool to 5°C, start adding phosphorus oxychloride dropwise, keep the temperature below 30°C, after 2h dropwise, stir for 0.5h, and control the pH value at 7-8; Then add dimethyl sulfate, recycle methanol for 3 hours, then cool the material in the pot to below 20°C, add carbon tetrachloride to filter, wash the filter cake with a small amount of carbon tetrachloride, combine the lotion and filtrate, and recover tetrachloride carbonized, and distilled under reduced pressure to obtain the crude product. Add distilled water and activated carbon to the crude product, add anhydrous potassium carbonate for dehydration after filtration, and finally obtain the product by distillation under reduced pressure.
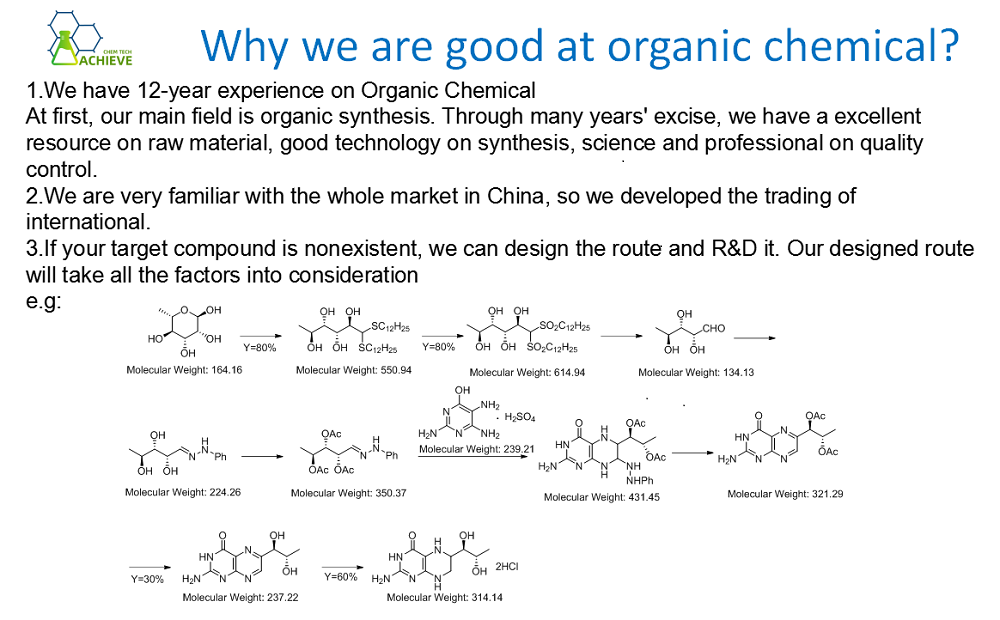
The production of it is mainly divided into two types: one is the reaction of trimethyl phosphate obtained from formaldehyde through chloroform, and the other is the reaction of it obtained from chloral through chloroform, wherein formaldehyde The temperature of the reaction with chloral is required to be above 100 degrees Celsius, while the reaction containing Australian needs to be heated to 150 degrees Celsius. In addition, since there is a decomposition reaction in the reaction, it is necessary to add a stabilizer to prevent the decomposition of the reactant
At present, the research on the production process of it mainly focuses on the research on the reaction temperature and stable production. An improved process is to use low-temperature reaction to reduce the reaction temperature to 50-60 degrees Celsius, which can greatly reduce energy consumption and increase the yield of it. Another improved process is to use photocatalyst, which can effectively inhibit the decomposition reaction in the reaction, thereby greatly increasing the yield of it. In addition, microwave technology or ionizing radiation technology can also be used to increase the reaction temperature, thereby effectively increasing the yield of it. In a word, the production process of product has always been the focus of research. The corresponding improvement technology is also constantly developing. Combining traditional reaction technology with new technology can effectively improve the production efficiency of trimethyl phosphate, thus ensuring the market demand.

trimethyl phosphate (chemical formula: (CH3O) 3PO), as the simplest trialkylphosphate, plays an important role in organic chemistry and industrial applications. Since its discovery in the 19th century, this colorless and transparent liquid compound has received continuous attention due to its unique chemical properties and extensive application value. Triethyl phosphate is not only a model molecule for studying the structure and properties of organic phosphorus compounds, but also plays an irreplaceable role in flame retardant materials, solvents, extractants, and organic synthesis intermediates.
In 1811, French chemist Thenard first reported the formation of esters through the reaction of phosphoric acid with ethanol, which is considered the beginning of organophosphate chemistry. In the following decades, scientists discovered various alkyl phosphate esters, but clear records of trimethyl phosphate did not appear until the mid-19th century.
In 1847, German chemist August Wilhelm von Hofmann first isolated and described trimethyl phosphate while studying the reaction between methanol and phosphorus pentoxide. In his paper published in the Journal of the German Chemical Society, Hofmann detailed the physical properties of this new compound, including its unique solubility and volatility.
In the second half of the 19th century, with the development of organic structure theory, many chemists devoted themselves to elucidating the structure of trimethyl phosphate. In 1873, Russian chemist Alexander Mikhailovich Zaitsev confirmed through systematic chemical degradation experiments that the compound discovered by Hofmann was indeed a fully esterified product formed by three methyl groups and phosphoric acid. The synthesis methods during this period mainly relied on the direct reaction of methanol with phosphorus pentoxide or phosphoryl chloride, with low yields and many by-products.
At the beginning of the 20th century, with the advancement of physical and chemical analysis methods, the structural research of trimethyl phosphate entered a new stage. In 1905, British chemist Thomas Martin Lowry first determined the molecular weight of trimethyl phosphate using the freezing point method, and the results were highly consistent with theoretical values, providing key evidence for confirming its molecular formula. In the 1920s, the application of X-ray diffraction technology enabled scientists to study the molecular configuration of trimethyl phosphate more intuitively.
In 1935, American chemist Linus Pauling discovered in his research that the phosphorus oxygen bond in trimethyl phosphate molecules had partial double bond properties, which was of great significance for understanding the electronic structure of phosphate compounds. Pauling's research shows that the phosphorus atom in trimethyl phosphate adopts sp3 hybridization, forming sigma bonds with three methoxy groups and d π - p π bonds with oxygen atoms. This electronic structure explains the relative stability of trimethyl phosphate.
During World War II, the military application of infrared spectroscopy and Raman spectroscopy technology promoted the study of vibrational spectra of trimethyl phosphate. In 1943, American chemist Richard C. Lord first reported the complete infrared spectrum of trimethyl phosphate, providing important reference for subsequent structural analysis. The emergence of nuclear magnetic resonance technology in the 1950s enabled scientists to study the molecular structure and conformational changes of trimethyl phosphate more accurately.
The industrial production method of trimethyl phosphate has undergone multiple technological innovations. Early industrial production (1920-1940) mainly used the reaction route of methanol and phosphorus oxychloride (POCl3), which was optimized by German chemist Gerhard Schrader in 1927. The reaction conditions were slow dropwise addition of methanol at low temperature (0-5 ℃), and the yield could reach 75%. However, this method generates a large amount of corrosive hydrogen chloride by-products and requires high equipment requirements.
In the 1950s, Monsanto, an American company, developed a direct esterification process between methanol and phosphorus pentoxide. The reaction was carried out under mild conditions (60-80 ℃), using inert solvents to reduce side reactions, and the yield increased to over 85%. The improved version of this process is still one of the main methods of industrial production today. In the 1960s, Japanese scientists developed a gas-phase catalytic esterification method, which reacted methanol with phosphoric acid in the gas phase in the presence of alumina catalyst, achieving continuous production.
In the 21st century, the concept of green chemistry has promoted the innovation of the synthesis method of trimethyl phosphate. In 2008, the Chinese Academy of Sciences developed an ionic liquid catalytic system to make the reaction conditions more moderate (at room temperature), and the catalyst can be recycled. In 2015, BASF, a German company, reported a new synthesis process based on supercritical methanol, which significantly improved reaction efficiency while reducing energy consumption and waste generation.
FAQ
1. What are the main applications of triethyl phosphate?
Its main applications include: serving as a methylating reagent and reaction solvent in organic synthesis; functioning as an efficient flame retardant, used in plastics, resins and lithium-ion battery electrolytes; and being used as an extractant or stabilizer in certain fields.
2. What are its physical characteristics?
At room temperature, it is a colorless and transparent liquid with a faint odor. It can be miscible with water and most common organic solvents. Its boiling point is relatively high (about 197°C) and its chemical properties are relatively stable.
3. What should be noted when using triethyl phosphate?
Due to its potential toxicity to the nervous system, strict protective measures must be taken during operation (such as fume hoods, gloves, and goggles), and inhalation of vapors or skin contact should be avoided. When storing, it should be sealed and kept away from heat sources and oxidants.
Hot Tags: trimethyl phosphate cas 512-56-1, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 3 methyl 4 piperidone, 2 6 Pyridinedicarboxylic acid, 3 Nitrobenzonitrile, 2 4 Quinolinediol, 3 Phenyltoluene, squaric acid treatment




