Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of hexafluoroisopropanol cas 920-66-1 in China. Welcome to wholesale bulk high quality hexafluoroisopropanol cas 920-66-1 for sale here from our factory. Good service and reasonable price are available.
Hexafluoroisopropanol (HFIP), CAS 920-66-1, molecular formula C3H2F6O, molecular weight 168.04, EINECS number 213-059-4. At room temperature, it is a colorless, transparent, non flammable liquid with an irritating odor. It has high polarity and can be miscible with water or most organic solvents in any proportion, but is insoluble in long-chain alkanes. In addition, it also has good thermal stability and ultraviolet light transmittance, capable of transmitting ultraviolet light with a wavelength less than 2000A and a low refractive index. In chemical laboratories, HFIP can be used as an analytical reagent and catalyst ligand. It can be used for the analysis of various polymers and as a regenerant for polymer reuse. In addition, HFIP can also be used as a ligand as a catalyst in organic synthesis reactions to improve reaction efficiency and selectivity.

|
Chemical Formula |
C3H2F6O |
|
Exact Mass |
168 |
|
Molecular Weight |
168 |
|
m/z |
168 (100.0%), 169 (3.2%) |
|
Elemental Analysis |
C, 21.44; H, 1.20; F, 67.84; O, 9.52 |
|
|
|

Hexafluoroisopropanol (HFIP) is an important fluorinated fine chemical with unique physical and chemical properties, such as high polarity, good solubility, thermal stability, and UV transmittance. These characteristics make HFIP widely used in multiple fields.

Solvent and cleaning agent field
1. Polymer dissolution and regeneration
HFIP, as an excellent solvent, can dissolve various polymers including polyester, polyamide, polyacrylonitrile, polyacetal, and hydrolyzed polyethylene ester. This characteristic makes it have important applications in the fields of plastic recycling and polymer regeneration. In the process of plastic recycling, HFIP can dissolve plastics mixed with pollutants and other metal ions, and then obtain pure plastics through liquid separation and solvent extraction. Due to the low boiling point of HFIP (58 ℃), the energy consumption of the extraction solvent is relatively low, making the process economically feasible.
2. Electronic industry cleaning agents
In the electroncs industry, HFIP is used as a solvent cleaner for cleaning electronc components and precision instruments. Its good solubility and volatility enable it to effectively remove dirt and grease from the surface of electronc components, while leaving no residue, thus ensuring the performance and reliability of electronc components. In addition, the low surface tension and low viscosity of HFIP also allow it to penetrate into tiny gaps, achieving more thorough cleaning.


3. Chemical reaction solvent
HFIP is also widely used in organic synthesis and analytical chemistry. It is soluble in various organic solvents such as alcohols, ethers, esters, and aromatic hydrocarbons, making it an ideal solvent for many chemical reactions. HFIP can provide good solubility and reaction environment in solution preparation, reaction catalysis, and chemical analysis, promote reaction progress, and improve product purity.
Medical field
1. Pharmaceutical intermediates
HFIP is an important intermediate for the synthesis of various fluorinated pharmaceuticals. For example, it can be used to synthesize the inhaled anesthetic sevoflurane. Sevoflurane is an advanced inhaled anesthetic with advantages such as no residue, rapid induction and recovery, and strong controllability of anesthesia depth. It is currently one of the most ideal inhaled anesthetics. HFIP can also be used to synthesize other fluorine-containing drugs, such as antidepressants, anticancer drugs, or anti-inflammatory drugs.


These drugs play an important role in clinical treatment, and HFIP, as a key intermediate, provides the possibility for the synthesis of these drugs.
2. Drug solvents and reaction media
In addition to serving as an intermediate, HFIP can also be used as a drug solvent and reaction medium. Its good solubility and chemical stability enable it to dissolve multiple drug components and promote drug synthesis reactions. In the process of drug formulation, HFIP can also be used to adjust the solubility and stability of drugs, thereby improving their efficacy and safety.
Pesticide field
1. Pesticide intermediates
HFIP is also an intermediate in the synthesis of agricultural chemicals. It can be used for synthesizing pesticides such as fluoroether pyrethroid insecticides. These pesticides have the advantages of high efficiency, low toxicity, and broad spectrum, and play an important role in agricultural production. HFIP, as a key intermediate, provides necessary raw material support for the synthesis of these pesticides.


2. Pesticide solvents and synergists
HFIP can also be used as a solvent and enhancer in pesticide formulations. Its good solubility and permeability can promote the spread and penetration of pesticides on plant surfaces, thereby improving the effectiveness of pesticide control. Meanwhile, HFIP can also synergistically interact with other pesticide components to enhance the overall efficacy of pesticides.
Surface active agents and emulsifiers field
1. Fluorinated surfactants
HFIP can be used to prepare fluorinated surfactants. These surfactants have excellent surface activity and wettability, which can reduce the surface tension of liquids and improve their wettability. Fluorinated surfactants can improve the wetting and stability of products such as coatings, inks, and lubricants, thereby enhancing their performance and quality.

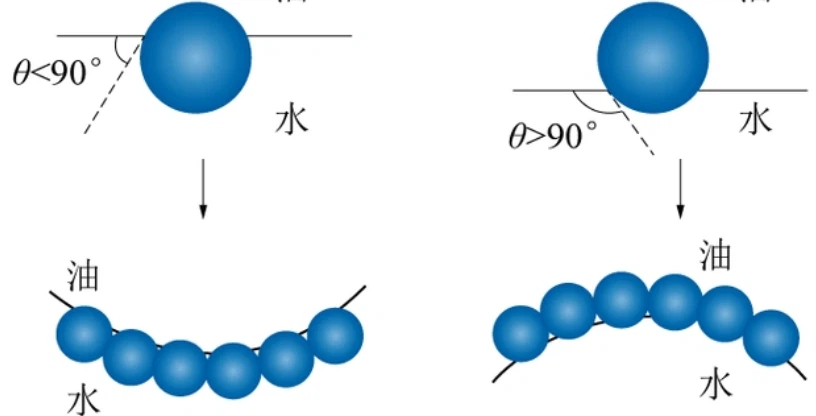
2. Fluorinated emulsifier
In addition to surfactants, HFIP can also be used to prepare fluorinated emulsifiers. These emulsifiers can form a stable emulsion film at the oil-water interface, allowing the oil-water phase to mix evenly and form a stable emulsion. Fluorinated emulsifiers play an important role in the fields of cosmetics, food, and medicine, providing guarantees for the stability and effectiveness of products.
In the field of catalysts and reaction media
1. Organic synthesis catalyst
HFIP has a catalytic effect in organic synthesis reactions. It can be used as a catalyst for esterification, etherification, olefin polymerization, and hydrogenation reactions. Its acidic and hydrogen bonding properties make it an effective catalyst for many organic reactions. For example, in region selective halogenated reactions, HFIP as a solvent can greatly shorten the reaction time and improve the yield of the product.


HFIP also exhibits excellent catalytic performance in organic synthesis reactions such as Friedel Crafts alkylation.
2. Special reaction medium
In addition to serving as a catalyst, HFIP can also be used as a special reaction medium. For example, in the rhodium (I) - catalyzed [4+2] intramolecular cycloaddition reaction of ether bound alkynyl dienes and the [5+2] cycloaddition reaction of alkynyl vinyl cyclopropane, HFIP as a fluorinated solvent can improve reaction efficiency and promote product generation. In addition, in liquid-phase peptide chemistry, HFIP is also used as a highly soluble solvent for the synthesis and analysis of peptides and peptide intermediates.
Polymer materials field
1. Polymer synthesis and modification
HFIP is also widely used in the field of polymer materils. It can be used as a solvent in the synthesis and modification process of polymer materils. For example, in the synthesis of polymer materils such as polyester and polyamide, HFIP can provide good solubility and reaction environment, promoting the growth of polymer chains and the progress of crosslinking reactions.


Meanwhile, HFIP can also be used in the modification process of polymer materils to improve their performance and quality by introducing fluorinated groups.
2. Polymer Material Analysis
In addition to synthesis and modification, HFIP can also be used in the analysis process of polymer materils. Its good solubility and chemical stability enable it to dissolve various polymer materils and conduct analysis and testing. For example, HFIP plays an important role in polymer molecular weight determination and thermal performance analysis.
Other fields
1. Antioxidant
HFIP has good antioxidant properties and can prevent oxygen from oxidizing certain compounds. Therefore, it is often used as an antioxidant to delay the oxidation and aging process of substances. HFIP, as an antioxidant, can protect the quality and stability of products in the fields of food, cosmetics, and medicine.
2. Chemical analysis reagents
In some chemical analyses, hexafluoroisopropanol can be used as a solvent and analytical medium for the sample. Its good solubility and chemical stability enable it to improve the accuracy and sensitivity of analysis.


For example, HFIP plays an important role in chemical analysis methods such as chromatography and mass spectrometry.
3. Lithium battery electrolyte additive
With the rapid development of new energy vehicles and energy storage technology, the demand for lithium battery electrolyte additives is also constantly increasing. HFIP can be used as a raw material in the production process of lithium battery electrolyte additive tris (hexafluoroisopropyl) phosphate ester. This additive can improve the performance and safety of lithium batteries, thereby meeting the needs of new energy vehicles and energy storage technology.

Synthesis of Hexafluoroisopropanol:
Hexafluoroacetone (HFA, hexafluoroacetone tone) is commonly used in catalytic hydrogenation and reduction synthesis, and can also be used in gas-phase or liquid-phase methods. HFA is gaseous at room temperature and reacts with excess hydrogen in a tubular reactor under the action of a catalyst. The gas-phase method has a fast reaction rate, but the reaction temperature is high, which can easily lead to side reactions. HFA combines with water to form hydrates containing different amounts of water. The liquid-phase catalytic hydrogenation reaction of HFA hydrate can be directly carried out at lower temperatures. The preparation of HFIP through catalytic reduction requires the use of expensive catalysts, and HFA usually contains small amounts of hydrogen fluoride, hydrogen chloride, and other impurities, which can easily cause catalyst poisoning, thereby increasing costs and reducing reaction selectivity.
45 kilograms of hexafluoroacetone hydrate (containing 1% catalyst) were fed into a 50 liter reactor for vacuum nitrogen and nitrogen hydrogen substitution.
Inject hydrogen gas to atmospheric pressure, heat to 100 ° C, stir at 30 revolutions per minute, inject hydrogen gas to 1 megapascal, and react at constant pressure until there is no pressure drop within 1 hour.
Reduce temperature and pressure, press out the material and filter it.
Using liquid-phase catalytic hydrogenation method to reduce HFA and prepare HFIP.
The results show that:
Under the reaction conditions of 1% catalyst dosage, 100 ° C, and 1.0 MPa, the conversion rate of HFA was 98.87% and the reduction selectivity was 97.87% when the reaction time was 22 hours.
Meanwhile, increasing the temperature can effectively improve reaction efficiency, shorten reaction time, and optimize the reaction temperature to 100 ° C.
The pH change of the reaction solution has no significant effect on the reaction.
After 48 hours of pretreatment, the catalyst can achieve a conversion rate of 98.98% and a reduction selectivity of 98.90% within 5 hours under the same reaction conditions.
FAQ
What is hexafluoroisopropanol used for?
It has also found use in biochemistry to solubilize peptides and to monomerize β-sheet protein aggregates. Because of its acidity (pKa 9.3), it can be used as acid in volatile buffers for ion pair HPLC - mass spectrometry of nucleic acids. HFIP has corrosive vapours that quickly cause eye and lung damage.
Is hexafluoroisopropanol toxic?
Toxicity. Hexafluoro-2-propanol has very low acute toxicity, hence its use as a precursor to anesthetics. Although it has low acute toxicity, it is a strong irritant to skin and eyes. Animal experiments show possible adverse effects on fertility, placing HFIP as a reproductive toxicity category 2 material.
Hot Tags: hexafluoroisopropanol cas 920-66-1, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale









