Angiotensin Peptide(Hypertensin), molecular formula C62H89N17O14, CAS 1407-47-2, is a collective term for a series of bioactive peptide hormones that play a crucial role in regulating arterial pressure, fluid, and electrolyte homeostasis. It usually appears as a white or slightly yellowish amorphous powder. This powder has good hygroscopicity, so it may absorb moisture and clump in humid environments. The molecular weight is approximately 1046.2 Daltons (Da), which is due to its composition of 8 amino acid residues and the presence of two disulfide bonds.
The exact molecular weight may vary slightly due to the modification or degradation of amino acids.The specific types of hypertensin include Angiotensin I, Angiotensin II (Angiotensin II, abbreviated as Ang II), and Angiotensin III, whose physical properties vary due to different molecular structures. As an ionic compound, Ang II has a certain conductivity in solution. The magnitude of its conductivity depends on its dissociation degree and ion concentration in the solution. By measuring the conductivity, the degree of ionization and dissociation state of Ang II in solution can be understood.
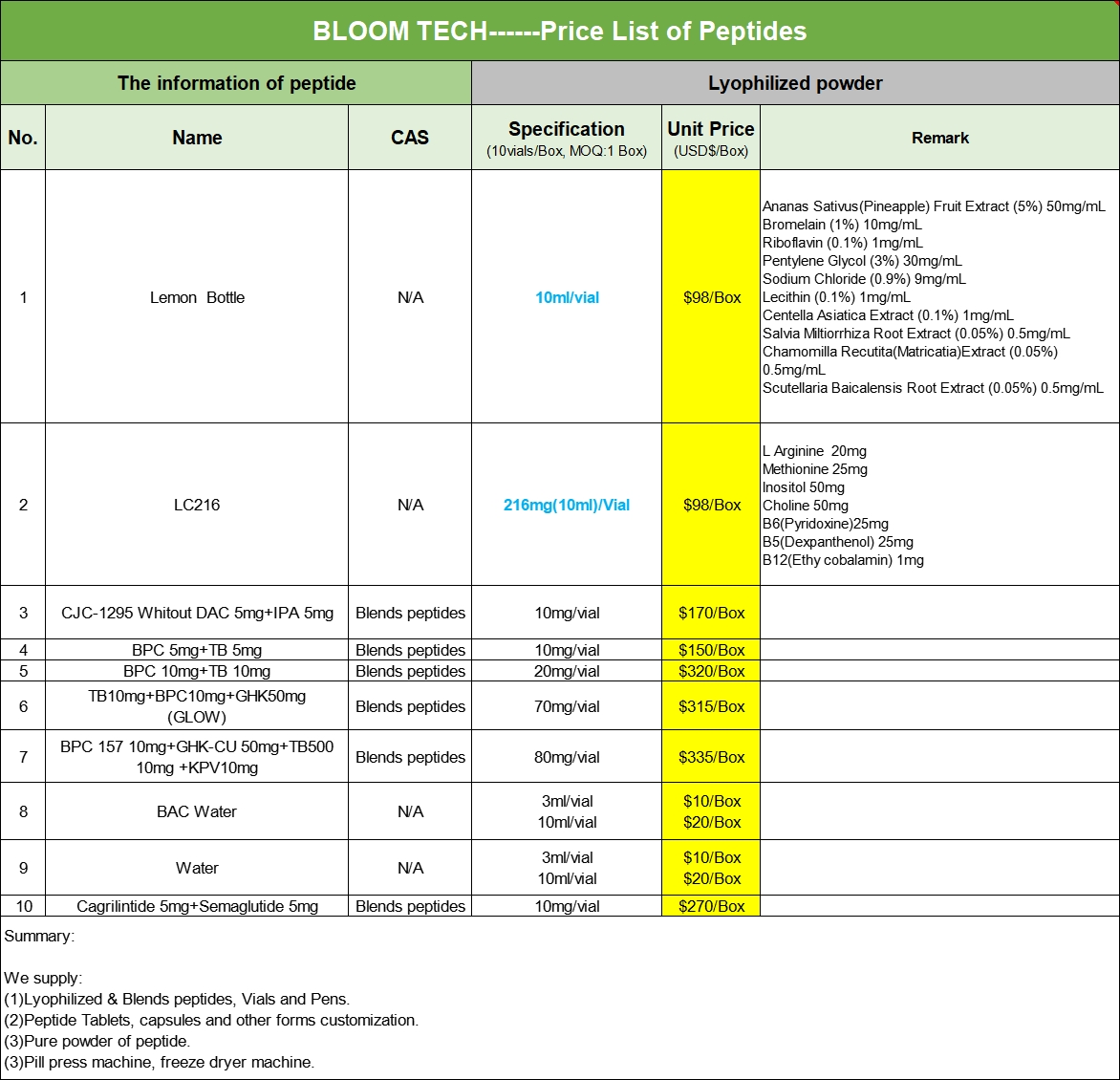
Our Products Form





Angiotensin COA



Angiotensin Peptide, also known as hypertensin, is a series of bioactive peptide hormones that play a crucial role in the cardiovascular system. Hypertensin mainly participates in physiological processes such as arterial pressure regulation, water salt balance, and vasculovascular remodeling.

1. Arterial pressure regulation
Hypertensin plays a crucial role in arterial pressure regulation, particularly in maintaining arterial pressure homeostasis. Ang II is the most active member of the hypertensin family, acting by activating the angiotnsin receptor (AT1R). Ang II can contract vascular smooth muscle, increase peripheral resistance, and thus increase arterial pressure. In addition, Ang II can also stimulate the release of aldosterone, leading to water and sodium retention, further raising hypertension.
2. Water salt balance
Hypertensin maintains water salt balance by regulating kidney function. Ang II can stimulate the reabsorption of sodium ions in the renal proximal tubules, reduce urinary sodium excretion, and thus prevent the occurrence of hyponatremia. Meanwhile, Ang II can also promote the release of antidiuretic hormone (ADH), reduce urine output, and further maintain fluid balance.

3. Cardiovascular remodeling
Hypertensin also plays an important role in the process of vasculovascular remodeling. Under the pathological conditions of vasculovascular diseases such as hypertension and atherosclerosis, angiotnsin can promote the proliferation and migration of vascular smooth muscle cells, leading to thickening of vascular walls and narrowing of lumens. In addition, Ang II can stimulate the proliferation and collagen synthesis of myocardial fibroblasts, promote myocardial fibrosis, and affect cardiac function.
4. Oxidative stress
Hypertensin is closely related to oxidative stress. Ang II can stimulate the production of reactive oxygen species (ROS), leading to an enhanced oxidative stress response. Oxidative stress not only aggravates vasculovascular injury, but also participates in the occurrence and development of many vasculovascular diseases, such as atherosclerosis, heart failure, etc.
5. Autophagy
In recent years, studies have found that angiotnsin is also involved in regulating autophagy. Autophagy is an intracellular degradation and recovery mechanism that is of great significance for maintaining cellular homeostasis. Ang II can inhibit autophagy by activating the mTOR signaling pathway, leading to intracellular protein and organ accumulation, thereby affecting the function and survival of vasculovascular cells.

Angiotensin peptide, especially Ang II, play an important role in the vasculovascular system. In addition to its known pressor effect and function in regulating water salt balance, Ang II also participates in the regulation of cellular autophagy. Autophagy is an intracellular degradation process that maintains cellular homeostasis and normal function by clearing damaged or excess organelles and proteins.

Basic concepts and mechanisms of autophagy
Autophagy is a highly conserved cellular process that involves encapsulating organelles, proteins, and other cellular components within a bilayer membrane structure to form autophagosomes, which then fuse with lysosomes for degradation. This process is crucial for maintaining cellular homeostasis, coping with external pressures, and promoting cell survival. The regulatory mechanism of autophagy is complex, involving multiple signaling pathways and molecular mechanisms, including PI3K/Akt/mTOR pathway, AMPK pathway, etc.
Molecular mechanism of Ang II regulating autophagy
Ang II activates NADPH oxidase and mitochondrial KATP channel through AT1 receptor
After binding to the AT1 receptor, Ang II can activate NADPH oxidase, leading to a significant production of reactive oxygen species (ROS). ROS, as an important signaling molecule, can activate various signaling pathways, including the autophagy pathway.

Meanwhile, Ang II can further promote ROS production by opening mitochondrial KATP channels. These ROS can activate the expression of autophagy related genes, promoting the formation and degradation of autophagosomes.
Ang II regulates autophagy through the Akt/mTOR signaling pathway
Ang II regulates autophagy through the Akt/mTOR signaling pathway
Akt is a serine/threonine kinase, also known as protein kinase B, which plays a critical role in various cellular processes. Activation of Akt can inhibit the activity of mTOR, which is a negative regulator of autophagy. When mTOR is inhibited, autophagy is activated.

However, Ang II can indirectly inhibit autophagy by activating Akt through AT1 receptors. This effect may have a double-edged sword effect in the vasculovascular system: on the one hand, inhibiting autophagy may contribute to cell survival; On the other hand, excessive inhibition may lead to cell damage and functional impairment.
Ang II regulates autophagy by affecting NOS activity
Nitric oxide (NO) is an important signaling molecule involved in various physiological and pathological processes. NO is catalyzed by nitric oxide synthase (NOS). Ang II can inhibit the activity of NOS, leading to a decrease in NO production. The reduction of NO may regulate autophagy by affecting autophagy related signaling pathways, such as the PI3K/Akt/mTOR pathway. In addition, NO may directly participate in the formation and degradation of autophagosomes, although research in this area is not yet in-depth.


The role of Ang II induced autophagy in atherosclerosis
Atherosclerosis is one of the main pathological bases of vasculovascular disease. A large number of studies have shown that Ang II plays an important role in the occurrence and development of atherosclerosis. Ang II induces the release of large amounts of reactive oxygen species, damages endothelial cells, induces inflammation, and promotes the migration, proliferation, and apoptosis of vascular smooth muscle cells, leading to structural and functional abnormalities in the vascular wall.
During this process, Ang II induced autophagy may have a dual effect. On the one hand, basic/physiological autophagy helps to clear damaged organelles and proteins, maintaining the homeostasis and normal function of vascular wall cells; On the other hand, excessive autophagy may lead to cell death and plaque instability, and aggravate the process of atherosclerosis.


The role of Ang II in regulating autophagy in myocardial ischemia-reperfusion injury
Myocardial ischemia-reperfusion injury is one of the common pathological processes in vasculovascular diseases. During myocardial ischemia, insufficient oxygen supply leads to cell damage; During reperfusion, due to the excessive generation of oxygen free radicals and mechanisms such as calcium overload, cell damage is further exacerbated.
During this process, Ang II inhibits autophagy by activating the Akt/mTOR signaling pathway, which may help alleviate myocardial ischemia-reperfusion injury. However, this effect may be time-dependent and dose-dependent. In the early stages of ischemia, inhibiting autophagy may contribute to cell survival; During the late stage of ischemia or reperfusion, excessive inhibition may lead to cell damage and functional impairment.


The role of Ang II in regulating autophagy in atrial remodeling
Atrial remodeling is one of the important pathological foundations of arrhythmia diseases such as atrial fibrillation. Research has shown that Ang II regulates autophagy and promotes increased collagen secretion by activating the ERK signaling pathway in atrial fibroblasts, leading to increased deposition of collagen in the cardiac interstitium and atrial remodeling.
During this process, Ang II induced autophagy may participate in the occurrence and development of atrial remodeling by affecting the synthesis and degradation of collagen. In addition, autophagy may also regulate the process of atrial remodeling by affecting the proliferation and apoptosis of atrial fibroblasts.


Angiotensin Peptide is a series of bioactive peptide hormones that play a crucial role in regulating arterial pressure, fluid, and electrolyte homeostasis. The laboratory synthesis of Angiotnsin mainly involves chemical synthesis techniques, including amino acid condensation, peptide chain elongation, and the use of necessary protective groups and activation reagents. The synthesis usually adopts the methods of solid-phase peptide synthesis (SPPS) or liquid-phase peptide synthesis (LPPS). These methods allow for the construction of peptide chains by gradually adding amino acids.
1. Solid phase peptide synthesis (SPPS)
Solid phase peptide synthesis is a commonly used method for peptide chain synthesis, which uses a solid phase support to fix the N-terminus of the peptide chain, and then extends the peptide chain by gradually adding amino acid residues.
The following is a simplified synthesis route for Angiotnsin II (Ang II):
Step 1: Fixation of starting amino acids
Connect the carboxyl end of the starting amino acid (such as L-valine) to the hydroxyl group on the solid support (such as polystyrene resin) through ester bonds.
Chemical equation:H2N-CH(CH3)-CH2-COOH+R-OH → H2N-CH(CH3)-CH2-COO-R+H2O
Step 2: condensation of amino acids
Protect the amino group of the first amino acid (such as with Boc protection), then add the second amino acid (such as L-isoleucine) and connect it through peptide bonds. This step usually requires the use of condensation agents (such as DCC, DIC, etc.) and catalysts (such as DMAP).
Chemical equation:Boc-H2N-CH(CH3)-CH2-COO-R+H2N-CH(CH3)-CH2-COOH → Boc-H2N-CH(CH3)-CH2-CONH-CH (CH3)-CH2-COO-R+H2O
Step 3: Remove protection and elution
Remove protective groups (such as Boc), and then elute the peptide chain from the solid-phase support. This step usually requires the use of acid or alkali.
Chemical equation:Boc-H2N-CH(CH3)-CH2-CONH-CH(CH3)-CH2-COO-R+HCl → H2N-CH(CH3)-CH2-CONH-CH(CH3 -CH2-COO-R+Boc Cl
Step 4: Subsequent amino acid condensation
Repeat the process of the second and third steps, gradually adding the remaining amino acids (such as L-tyrosine, L-proline, and L-phenylalanine) until complete Ang II is synthesized.
Step 5: Purification and identification of the final product
Remove impurities through appropriate purification methods such as HPLC, and identify the structure and purity of the final product through mass spectrometry, nuclear magnetic resonance, and other means.

2. Liquid phase peptide synthesis (LPPS)
Liquid phase peptide synthesis is a method of peptide chain synthesis carried out in solution. Compared with solid-phase peptide synthesis, liquid-phase peptide synthesis does not require solid-phase support, but gradually constructs peptide chains in solution. The following is a simplified synthesis route for Ang II:
Step 1: Activation of starting amino acids
Activating the carboxyl group of starting amino acids (such as L-valine) typically involves the use of activation reagents (such as NHS, EDC, etc.).
Chemical equation:H2N-CH(CH3)-CH2-COOH+NHS+EDC → H2N-CH(CH3)-CH2-CO-NHS+EDC · HCl
Step 2: condensation of amino acids
React the activated amino acid with the amino group of the second amino acid (such as L-isoleucine) to form a peptide bond. This step is usually carried out under mild conditions and does not require additional catalysts.
Chemical equation:H2N-CH(CH3)-CH2-CO-NHS+H2N-CH(CH3)-CH2-COOH → H2N-CH(CH3)-CH2-CONH-CH(CH3)-CH2-COOH+NHS
Step 3: Subsequent amino acid condensation
Repeat the process of the second step, gradually adding the remaining amino acids (such as L-tyrosine, L-proline, and L-phenylalanine) until the complete Ang II is synthesized.
Step 4: Purification and identification of the final product
Similar to solid-phase peptide synthesis, impurities are removed through appropriate purification methods, and the structure and purity of the final Angiotensin Peptide are identified through mass spectrometry, nuclear magnetic resonance, and other means.
Emerging Research and Future Directions
Current emerging researches mainly focus on three core directions. First is the in-depth exploration and application of the protective axis. The ACE2-Ang(1-7)-MasR protective axis has been proven to counteract the damage caused by the classical pathogenic ACE-Ang II-AT1R axis. Ang(1-7), Ang(1-9) and the novel endogenous peptide alamandine can effectively dilate blood vessels, inhibit inflammatory responses and alleviate organ fibrosis, showing prominent protective effects especially in heart failure and hypertensive nephropathy. Progress has also been made in the modification of related peptide analogs, which effectively solves the problems of short half-life and low bioavailability of natural peptides.
Second is the mechanistic research on tissue-specific RAS (tRAS). The autocrine and paracrine functions of local RAS in the heart, kidney, brain, adipose tissue and other organs have been gradually revealed. Its functional abnormality is closely associated with local organ lesions, providing precise targets for organ-specific protection. Third is technology-driven research innovation. With the aid of AI virtual screening and high-throughput molecular docking technology, high-activity ACE inhibitory peptides and ACE2 upregulatory peptides can be rapidly screened.

Meanwhile, it assists in designing stable and low-toxic peptide analogs, greatly improving the efficiency of drug research and development. In addition, studies have discovered its correlation with cross-border diseases such as obesity, Alzheimer's disease and pulmonary fibrosis, further expanding its application scope.
Future research will focus on four major directions: first, dual-pathway balanced regulation, which combines inhibition of the pathogenic axis and activation of the protective axis to achieve the dual effects of disease suppression and organ protection.
Second, optimization of delivery systems, developing nanocarriers and targeted preparations to enhance the stability and targeting ability of peptide drugs and reduce systemic side effects; third, personalized precision therapy, screening susceptible populations combined with genomics and proteomics, and accelerating efficacy evaluation via organoid technology; fourth, development of natural peptide resources, exploring bioactive peptides derived from plants and milk to provide safe nutritional intervention strategies for chronic diseases.
FAQ
What is it and its function?
+
-
It is a peptide hormone that causes vasoconstriction and an increase in blood pressure. It is part of the renin–angiotensin system, which regulates blood pressure. It also stimulates the release of aldosterone from the adrenal cortex to promote sodium retention by the kidneys.
Where is itproduced?
+
-
The liver creates and releases a protein called angiotensinogen. This is then broken up by renin, an enzyme produced in the kidney, to form angiotensin I.
What is the difference between renin and it?
+
-
Renin: Your kidneys mostly make the enzyme renin. It helps control your blood pressure. It also helps balance your sodium and potassium levels. Angiotensin II: This is a hormone.
Hot Tags: angiotensin peptide cas 1407-47-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 2 bromo N 4 chlorophenyl aniline, 3 6 4 4 5 5 tetramethyl 1 3 2 dioxaborolan 2 yl dibenzo b d furan 4 yl pyridine, 2 chlorospiro cyclopenta def phenanthrene 4 9 fluorene , propylene oxide market, 10 biphenyl 3 yl 2 bromo 9 9 dimethyl 9 10 dihydroacridine, 2 5 dibromo 1 1 4 1 terphenyl
















