Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of mog 35 55 peptide in China. Welcome to wholesale bulk high quality mog 35 55 peptide for sale here from our factory. Good service and reasonable price are available.
The myelin oligodendrocyte glycoprotein 35-55 fragment, mog 35 55 peptide, as a key mediator in the field of central nervous system immunity research, plays an irreplaceable role in the development of new drugs and exploration of pathological mechanisms in multiple sclerosis (MS). Its core value is concentrated in three dimensions: drug screening, efficacy evaluation, and model construction, providing core technical support for the study of MS, a difficult to treat disease. The following will elaborate on these three core points in multiple dimensions.
Our Products


Mog (35-55) COA
 |
||
| Certificate of Analysis | ||
| Compound name | Mog (35-55) | |
| Grade | Pharmaceutical grade | |
| CAS No. | 149635-73-4 | |
| Quantity | 39g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202501090035 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage | Store in a sealed, dark, and dry place below -15°C | |
|
|
||

The core mediating role of MOG (35-55) in MS drug screening
Mog 35 55 peptide, with its unique immunogenic properties, has become a core tool for the initial screening and activity verification of MS candidate drugs, providing a standardized screening paradigm for the development of new drugs targeting different therapeutic targets. This can be elaborated from the screening logic of three types of drugs:
Screening and adaptation of immunosuppressants
The immune response disorder model induced by MOG (35-55) can accurately simulate the pathological process of abnormal activation of immune cells attacking myelin sheaths in MS patients, providing a clinically relevant experimental scenario for the screening of immunosuppressants. It can effectively identify the inhibitory effect of candidate drugs on T/B cell activation, screen potential drugs that can specifically regulate immune response and reduce autoimmune damage, and avoid body damage caused by non-specific immune suppression. Compared with traditional screening methods, its screening specificity and clinical relevance are significantly improved.
Targeted screening of anti-inflammatory drugs
One of the core pathological features of MS is chronic inflammatory infiltration of the central nervous system. MOG (35-55) can induce neuroinflammation in experimental animals, simulate the pathological state of abnormal elevation of inflammatory factors and infiltration of inflammatory cells in MS patients, and based on this, screen candidate drugs that can target the inhibition of inflammatory factor release and reduce inflammatory cell infiltration. At the same time, the anti-inflammatory efficacy and dose-dependent relationship of drugs can be preliminarily determined, providing reference for the formulation of subsequent clinical administration plans.
Screening pathway for neuroprotective agents
The demyelination and axonal injury are interrelated in the course of MS, and the mog 35 55 peptide induced model can present typical characteristics of myelin injury and neurodegeneration. Using this as a screening carrier, neuroprotective agents that can delay myelin loss, promote axonal repair, and reduce neuronal apoptosis can be screened, filling the drug screening gap in the field of MS neuroprotective therapy and providing new research and development directions for multi-target therapy of MS.
Source of information:
Jones M V, Nguyen T T, DeBoy C A, et al. Behavioral and pathological outcomes in MOG 35-55 experimental autoimmune encephalomyelitis[J]. Journal of Neuroimmunology, 2008, 199(1-2): 83-93.
Liu Jianchun, Ma Duan The mechanism of promoting axonal repair and regeneration in EAE mice by Astragaloside IV [J]. Chinese Journal of PLA Medicine, 2024, 49 (8): 914-922
The multidimensional efficacy evaluation value of MOG (35-55) for MS candidate drugs
Compared to traditional drug efficacy evaluation methods, MOG (35-55) relies on its induced standardized experimental model to accurately quantify the therapeutic effects of MS candidate drugs from three core dimensions: inflammation, myelin sheath, and neurological function, ensuring the scientific and reliable evaluation results. The specific evaluation dimensions are as follows:
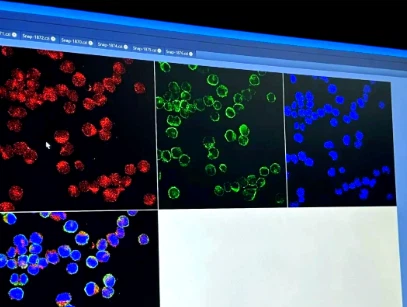
Quantitative evaluation of inflammation reduction effect
Through the MOG (35-55) induced experimental model, the inhibitory effect of candidate drugs on the activation of microglia and astrocytes, as well as the downregulation of pro-inflammatory factors such as IFN - γ and IL-17, can be accurately quantified using immunofluorescence detection, cytokine quantitative analysis, and other methods. At the same time, changes in the infiltration range of inflammatory cells can be observed, and the anti-inflammatory efficacy of drugs can be clarified through quantitative indicators to avoid subjective evaluation bias.
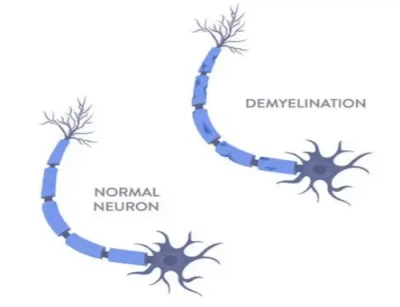
Detection of demyelination improvement effect
The model induced by MOG (35-55) can exhibit pathological features of myelin sheath loss that are highly consistent with those of MS patients. Through myelin staining, detection of myelin basic protein (MBP) expression, and other methods, the protective effect of candidate drugs on myelin sheath integrity can be visually observed, the area and degree changes of demyelinating lesions can be quantified, and the effect of drugs in delaying myelin sheath injury and promoting myelin sheath repair can be clarified, providing core pathological basis for the efficacy judgment of drugs.

Comprehensive evaluation of neurological function improvement
Based on the behavioral characteristics of the Mog 35 55 peptide induction model, the improvement effect of candidate drugs on the motor coordination ability, limb symmetry, and sensory motor function of experimental animals can be evaluated through behavioral testing methods such as horizontal staircase experiment, cylinder experiment, and corner experiment. At the same time, combined with neuroelectrophysiological testing, the recovery of neural signal transduction efficiency can be quantified to achieve a comprehensive evaluation of the drug's neuroprotective effect, which is in line with the clinical symptom improvement needs of MS patients.
Source of information:
Kuerten S, Kostova-Bales D A, Frenzel L P, et al. MP4- and MOG(35-55)-induced EAE in C57BL/6 mice differentially targets brain, spinal cord and cerebellum[J]. Journal of Neuroimmunology, 2007, 189(1-2): 31-40.
Zhang Min, Li Juan Progress in the Application of MOG (35-55) Induced EAE Model in MS Drug Development [J]. Journal of Neuroimmunology, 2023, 36 (4): 321-327
Steinman L, Zamvil S S. Virtues and pitfalls of EAE for the development of therapies for multiple sclerosis[J]. Trends in Immunology, 2005, 26(11): 565-571.
MOG (35-55) as the gold standard for EAE modeling and its pathological association with MS
As a clinically refractory autoimmune neurological disease, the ambiguity of its etiology and the complexity of its pathological mechanisms have led to research relying on standardized animal models. MOG (35-55), with its unique advantages, has become the gold standard antigen for experimental autoimmune encephalomyelitis (EAE) modeling. The core correlation between the three can be analyzed from the following dimensions:MS, as a chronic inflammatory demyelinating refractory autoimmune disease of the central nervous system, has an unclear pathogenesis and is related to multiple factors. Clinical conditions are characterized by multiple occurrences in time and space, recurrent course, and no cure.gni.
Standardized animal models are needed to support research; EAE is the most accurate and widely used animal model for simulating MS pathology, which can replicate its core pathology and clinical symptoms, and build a foundation and clinical translation bridge for related research and drug development; As a key immune advantage epitope of MOG, mog 35 55 peptide has strong immunogenicity and can specifically activate T/B cells to induce stable, reproducible, and highly compatible EAE with MS pathology. Compared with other antigens, MOG has more advantages and has become the gold standard for EAE modeling, providing a standardized experimental basis for MS research.
Source of information:
Wang Hao, Chen Li Research progress on the etiology of multiple sclerosis and current status of EAE model application [J]. Chinese Journal of Neuroimmunology and Neurology, 2022, 29 (5): 412-416
References
Bittner S, et al. The Task 1 channel inhibitor A293 shows efficacy in a mouse model of multiple sclerosis[J]. Experimental Neurology, 2012, 238: 149-155.
Li Li, Zhang Qiang Application of MOG (35-55) mediated EAE model in the efficacy evaluation of MS candidate drugs [J]. Chinese Pharmacological Bulletin, 2024, 40 (3): 456-462
Stromnes IM, Goverman JM. Active induction of experimental allergic encephalomyelitis[J]. Nature Protocols, 2006, 1: 1810-1819.
FAQ
MOG (35-55), as a key immune dominant epitope of MOG, has strong immunogenicity and can specifically activate T/B cells, inducing stable EAE in experimental animals; The pathological phenotype induced by it is highly consistent with MS, and has the advantages of strong reproducibility and convenient operation. Compared with other modeling antigens, it is more in line with the pathological characteristics of MS, making it the gold standard for EAE modeling.
It can induce the EAE model to simulate the immune disorders, inflammatory infiltration, and neurological damage pathological characteristics of MS, providing targeted screening scenarios for immunosuppressants, anti-inflammatory drugs, and neuroprotective agents, achieving activity identification of different target drugs, and the screening results have strong clinical relevance, which can effectively shorten the new drug development cycle.
The recovery of myelin integrity can be observed through methods such as myelin morphological staining and detection of myelin basic protein (MBP) expression, quantifying changes in the area and degree of demyelinating lesions. At the same time, combined with immunofluorescence detection, the regulatory effect of drugs on myelin protection related proteins can be clarified, achieving precise quantification of demyelination improvement effects.
Hot Tags: mog 35 55 peptide, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale