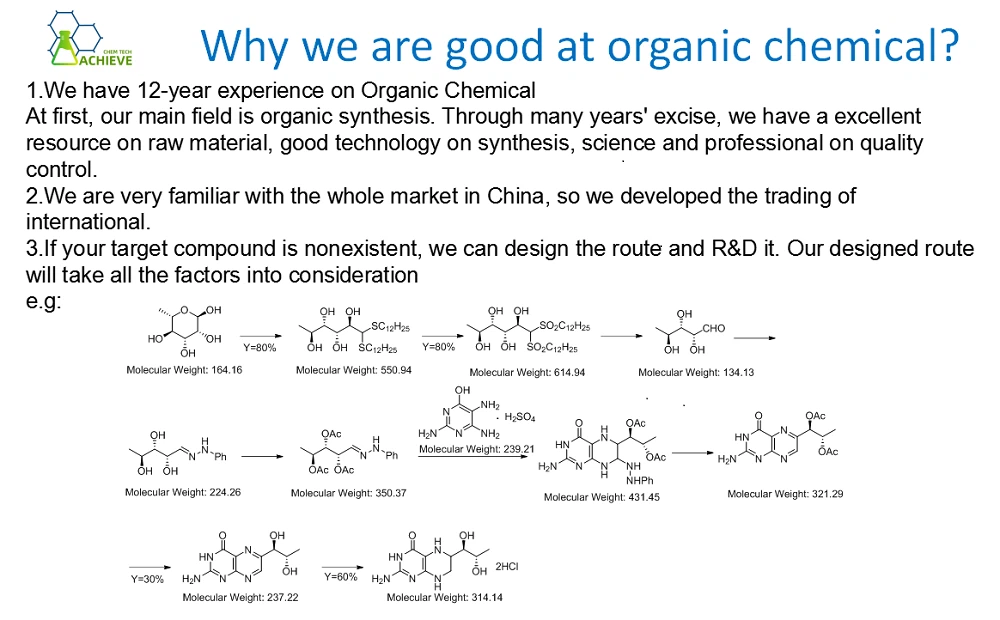
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of propylene carbonate cas 108-32-7 in China. Welcome to wholesale bulk high quality propylene carbonate cas 108-32-7 for sale here from our factory. Good service and reasonable price are available.
Propylene carbonate is a colorless, odorless, flammable liquid with the molecular formula C4H6O3, CAS C4H6O3. Can be miscible with ether, acetone, benzene, chloroform, vinyl acetate, etc., soluble in water and carbon tetrachloride, has strong absorption ability for carbon dioxide, and is stable. In industry, it is produced by adding epichlorohydrin and carbon dioxide under a certain pressure and distilling under reduced pressure.
Can be used as an oil solvent, spinning solvent, olefin, aromatic hydrocarbon extractant, carbon dioxide absorbent, water-soluble dye, and pigment dispersant. In the electronics industry, it can be used as an excellent medium for high-energy batteries and capacitors, and in the polymer industry, it can be used as a solvent and plasticizer for polymers.

|
Chemical Formula |
C4H6O3 |
|
Exact Mass |
102 |
|
Molecular Weight |
102 |
|
m/z |
102 (100.0%), 103 (4.3%) |
|
Elemental Analysis |
C, 47.06; H, 5.92; O, 47.01 |
|
|
|
Plasticizers used as adhesives and sealants. It can also be used as a curing accelerator for phenolic resins and a dispersant for water-soluble adhesive pigments and fillers. The chemical industry is the main raw material for synthesizing dimethyl carbonate and can also be used to remove carbon dioxide and hydrogen sulfide from natural gas and petroleum cracking gas· In addition, it can also be used in industrial fields such as textiles and printing and dyeing.

PC a five membered cyclic carbonate synthesized by cycloaddition of epichlorohydrin and carbon dioxide, has demonstrated extraordinary application value in various fields such as chemical engineering, energy, and materials due to its unique physical and chemical properties.
(1) Gas purification field
Carbon dioxide absorption:
Process advantages: Compared with the traditional alcohol amine method, the propylene carbonate method has the characteristics of high absorption load (0.55 m ³ CO ₂/m ³ solution) and low regeneration energy consumption (reduced by 30%).
Industrial case: Henan Junma Chemical's 300000 tons/year plant maintains a stable control of CO ₂ content in purified gas below 0.1%.
Natural gas desulfurization:
Collaborative absorption: It has selective absorption ability for H ₂ S and CO ₂, suitable for high sulfur natural gas purification.


Technical parameters: absorption temperature of 40 ℃, resolution temperature of 120 ℃, circulation volume of 1.5 L/m ³ natural gas.
(2) Industrial application of batteries
Lithium ion battery electrolyte:
Formula optimization: Adding 20% PC to the 1.2 mol/L LiPF ₆/EC+DEC system resulted in a low-temperature conductivity (-20 ℃) of 0.85 mS/cm for the electrolyte.
Performance improvement: After a certain power lithium battery company adopted this formula, the low-temperature capacity retention rate of the battery increased from 68% to 82%.
Lithium ion capacitors:
Electrolyte additive: Ethylene sulfite (ES) works synergistically with PC, and a 5% ES additive results in a 73.7% retention rate of the capacitor's 20C rate capacity.
(3) Polymer material processing
Plasticizer:


Epoxy resin modification: Adding 8% PC increased the shear strength from 18 MPa to 24 MPa, and a certain wind turbine blade enterprise extended the fatigue life by 30% after adopting it.
Polymerization reaction solvent:
Polycarbonate synthesis: When condensed with bisphenol A, PC acts as a solvent to reduce the reaction temperature by 15 ℃, and the molecular weight distribution index of the product decreases from 2.8 to 2.2.
(4) Synthesis of Fine Chemicals
Production of dimethyl carbonate (DMC):
Ester exchange process: PC reacts with methanol, with a DMC conversion rate of 92%. The purity of the product in a 50000 ton/year plant in a chemical industrial park remains stable at over 99.9%.
Pharmaceutical intermediates:
Synthesis of cephalosporin antibiotics: As an acylation reaction medium, it reduces reaction time by 40% and increases single batch yield by 12%.

Daily life applications

(1) Cleaning and degreasing
Low toxicity solvents:
VOC Advantage: Listed on the US EPA's Safer Chemicals List, with a vapor pressure of 0.023 mmHg, meeting VOC exemption standards.
Application case: Replacing dichloromethane for electronic component cleaning reduces surface tension by 30% and improves cleaning efficiency by 25%.
(2) Personal care and cosmetics
Safe alternatives:
Low irritation: Used as a substitute for N-methylpyrrolidone in cosmetic formulations, the skin irritation test score is reduced by 40%.
Film forming agent:
UV curable ink: Adding 10% PC increases the flexibility of the cured film by 50%, and the ASTM D522 bending test reaches 300 times without cracking.
(3) Home building materials
Wood adhesive:
Phenolic resin replacement: PC modified urea formaldehyde resin, with a bonding strength of 2.5 MPa and a 60% reduction in formaldehyde emissions.
Surface treatment agent:
Aluminum profile oxidation: Adding 3% PC improves the uniformity of oxide film thickness by 25%, and extends the salt spray test time from 120 hours to 180 hours.

Scientific research and cutting-edge exploration

(1) CO2 resource utilization
Chemical fixation technology:
Catalytic synthesis of PC: Titanate nanotube (TNT) catalyst, PO conversion rate>99.9%, PC selectivity 100%.
Mechanism study: The synergistic catalysis of hydroxyl and Lewis acid sites on the surface of TNT reduces the PO ring opening energy barrier.
(2) New energy materials
Solid electrolyte:
Polymer electrolyte: PC and PEO blend system, with an ionic conductivity of 1.2 × 10 ⁻⁵ S/cm (30 ℃), suitable for solid-state lithium batteries.
Sodium ion battery:
Electrolyte optimization: Adding fluorinated ethylene carbonate (FEC) to PC based electrolyte resulted in a sodium ion migration number of 0.82


(3) Environmentally friendly processes
Green catalytic system:
Biobased catalyst: Ionic liquid catalyzes the synthesis of PC from CO ₂ and epichlorohydrin, with a conversion rate of 85%, and can be recycled 10 times.
Degradable materials:
Poly (Propylene carbonate): PC co polymerized with CO ₂, with a material weight loss rate of 60% (after 3 months of soil burial), meeting the standards for biodegradable plastics.

Chemical properties and reaction mechanisms
(1) Molecular Structure and Physical Properties
Molecular Formula And Structure:
C4H6O3, a five membered cyclic structure containing one carbonate group and two methyl groups.
Physical State:
Colorless and transparent liquid, odorless, flammable, boiling point 242 ℃, melting point 48.8 ℃, density 1.205 g/cm ³.
Solubility:
It is miscible with polar solvents such as water, alcohol, ether, and benzene, and has strong absorption ability for gases such as carbon dioxide and hydrogen sulfide.
Stability:
Chemically stable, partially decomposed above 200 ℃, hydrolyzable under acid/alkali catalysis.
(2) Reactivity and Application Potential
Cycloaddition reaction:
Efficient synthesis of propylene carbonat from epichlorohydrin and carbon dioxide under the action of catalyst, with 100% atomic economy.
Solvent properties:
Its high dielectric constant (64.9) makes it an ideal component for lithium-ion battery electrolytes.
Functionalized derivatives:
Downstream products such as dimethyl carbonate and polycarbonate can be prepared through ester exchange, amine hydrolysis, and other reactions.

1. Synthesis based on 1,2-propanediol
Because the synthesis technology of 1,2-propanediol is relatively mature, and the product quality and output are relatively stable, there are many reports on the synthesis of it using propylene glycol as the main raw material.
Propylene glycol phosgene method:
The first industrial preparation of it was the synthesis reaction of 1,2-propanediol and phosgene.
Phosgene is a highly toxic substance, causing serious harm to people and the environment; In addition, the by-product hydrochloric acid is generated, which not only reduces the atomic economy of the process, but also increases the process investment cost due to the corrosion of hydrochloric acid on the equipment. Therefore, the use of this law has been prohibited.
Propylene glycol urea method
The synthesis of it from urea and 1,2-propanediol has been studied more in China. When propylene glycol reacts with urea to synthesize propylene carbonate, the first step is to generate amino carbonate, and the second step is to deamination and cyclization of amino carbonate to generate the target product, accompanied by the generation of by-product ammonia. The earlier reported patent for the preparation of it from urea and propylene glycol introduced mild reaction conditions and high yield of the target product. The catalyst introduced is organic tin, which has certain toxicity.
Using solid base catalyst can reduce the toxicity of the process. In the presence of solid alkali, such as zinc oxide, the reaction temperature is 100~200 ℃, nitrogen is introduced, and after a certain time of reaction, the yield of product calculated by urea can reach 99%. When the composite calcium oxide catalyst is used, under the condition of reduced pressure, the temperature is 150~160 ℃, the conversion of urea is 95%~98%, and the selectivity of it is 90%~98%. The catalyst can be recycled.
Using MgO calcined from basic magnesium carbonate as catalyst, it was synthesized from urea and propylene glycol. After 3 h reaction at 170 ℃, the PC yield was more than 90%. Inorganic lead and zinc compounds were used as heterogeneous catalysts. The yield of it was 98% in urea at 160 ℃ for 6 h; The reaction product and catalyst are easy to separate. Using Fe Zn oxide as catalyst, the yield of it was 78% after reaction at 170 ℃ for 2h. The main active component of the catalyst is ZnO, which is promoted by the joint action of ZnO and ZnFe2O4. The cost of product synthesized by propylene glycol urea method is relatively low, and it has certain advantages in process raw materials.
Propylene glycol carbon dioxide method
The reaction process uses carbon dioxide. Carbon dioxide is a greenhouse gas. As the concentration of carbon dioxide on the earth surface has increased due to human activities, it is a green synthetic idea to use carbon dioxide as a raw material to fix it into chemicals, and practical reports have been seen. At present, although the carbon dioxide used in most studies is not directly from emissions, its thinking is also considered green. The catalyst used in this method is alkali metal salt or alkaline earth metal salt, and the catalytic activity of potassium carbonate is high. In the homogeneous catalytic system, the yield of propylene carbonat can reach 12.6%.
In order to overcome the difficulties of product separation and catalyst recycling caused by homogeneous catalytic reaction, potassium carbonate was loaded on activated carbon for heterogeneous catalytic reaction. The results showed that the selectivity of products was improved. The solvent acetonitrile was used in the synthesis process, which reduced the green degree of the process. Tin organic compounds, such as Bu2SnO or Bu2Sn (OMe) 2, can also catalyze the reaction of 1,2-propanediol and carbon dioxide to produce propylene carbonat under supercritical conditions.
The addition of cosolvent or the presence of dehydrating agent is beneficial to the production and yield of the product. Water is generated during the reaction of 1,2-propanediol and carbon dioxide, which reduces the atom utilization rate in the reaction process, and the product will be hydrolyzed, so the yield of the product will be inhibited by water. This is a major problem to be solved in the process of industrialization.
Exchange method of propylene glycol and ester
It can be prepared by transesterification of 1,2-propanediol with diethyl carbonate or dimethyl carbonate.
The yield of it was 88% when alkali metal or alkaline earth metal was used as catalyst and reacted at 144 ℃ under normal pressure for 12 h. If dibutyltin dilaurate and trace strong base are used as catalyst for transesterification reaction, xylene reflux is used to control reaction temperature, and ethanol, the by-product, is continuously fractionated, the operation steps can be reduced. However, the raw materials used in this method are expensive, and the toxicity of organotin catalyst is relatively high, so it is not an ideal green process.
2. Synthesis based on propylene oxide
The addition cyclization of propylene oxide and carbon dioxide to produce is an exothermic and volume reducing reaction. Therefore, low temperature and high pressure conditions are conducive to the reaction. Because it is an addition reaction, the atomic economy of the process can reach 100 theoretically, but the actual situation is related to the catalytic system used.
The catalytic system mainly includes homogeneous catalytic system and heterogeneous catalytic system. In the homogeneous catalytic system, the complex catalyst can catalyze the reaction of propylene oxide and carbon dioxide to produce. Its disadvantage is that the catalyst concentration is relatively high and the reaction yield is relatively low. Quaternary ammonium salt, quaternary phosphine salt and alkali metal salt catalyst have high catalytic activity for the addition reaction of propylene oxide with carbon dioxide, and the conversion rate is relatively high.
A homogeneous metal ion complex catalyst, code name MC-3, catalyzes the reaction of propylene oxide with carbon dioxide under the conditions of reaction temperature of 135 ℃ and pressure of 3 MPa, and the yield of it is more than 94%. In addition, alkali metal salt catalyst can also catalyze the synthesis of propylene carbonate with the help of macrocyclic crown ether. Due to the strong toxicity of macrocyclic crown ether, the practical value of this synthesis method is reduced.
FAQ
Is propylene carbonate safe for skin?
Cosmetic products containing up to 20% Propylene Carbonate were essentially nonsensitizing and, at most, moderately irritating to human skin, nonphototoxic, and nonphotosensitizing. It is concluded that Propylene Carbonate is safe as a cosmetic ingredient in the present practices of use and concentration.
What is propylene carbonate used for?
Propylene Carbonate (CAS 108-32-7) is a **versatile, high-purity solvent** widely used in **coatings, adhesives, cosmetics, and chemical formulations**. Known for its **low volatility, high boiling point, and excellent solvency**, it is ideal for applications requiring **effective dissolving power and stability**.
Is propylene carbonate the same as acetone?
Propylene carbonate is used as a polar, aprotic solvent. It has a high molecular dipole moment (4.9 D), considerably higher than those of acetone (2.91 D) and ethyl acetate (1.78 D).
Is propylene carbonate safe for nails?
Further advantages of the propylene carbonate, propylene glycol and dimethyl isosorbide mixtures of the invention are that they are biodegradable, hypoallergenic and do not dry out or otherwise harm nails and cuticles.
Hot Tags: propylene carbonate cas 108-32-7, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, methyltetrahydrofuran









