Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of lithium tri tert butoxyaluminum hydride cas 17476-04-9 in China. Welcome to wholesale bulk high quality lithium tri tert butoxyaluminum hydride cas 17476-04-9 for sale here from our factory. Good service and reasonable price are available.
Lithium tri tert butoxyaluminum hydride (LTBA) is an organic metal compound. It is an important reducing agent with wide applications in organic synthesis. The appearance is usually a white to off white powder or solid, which may also be a colorless or light gray liquid (depending on the specific form and purity), soluble in certain organic solvents such as tetrahydrofuran (THF), and often used in solution form. It is a mild and highly selective reducing agent that can reduce ketones, aldehydes, and acyl chlorides at low temperatures (such as 0 ° C) without reducing fatty acid esters and nitriles. It can also reduce phenyl esters of fatty acids and cyclic acids, as well as tetrahydrofuran (ring opening), which is usually reacted under inert gas (such as argon) protection to avoid reacting with moisture and oxygen in the air. Mainly used for synthesizing organic molecules with biological activity, such as drug intermediates, participating in the construction of complex molecules, and improving synthesis efficiency. As a reducing agent, it also participates in various organic reactions.

Additional information of chemical compound:
|
Chemical Formula |
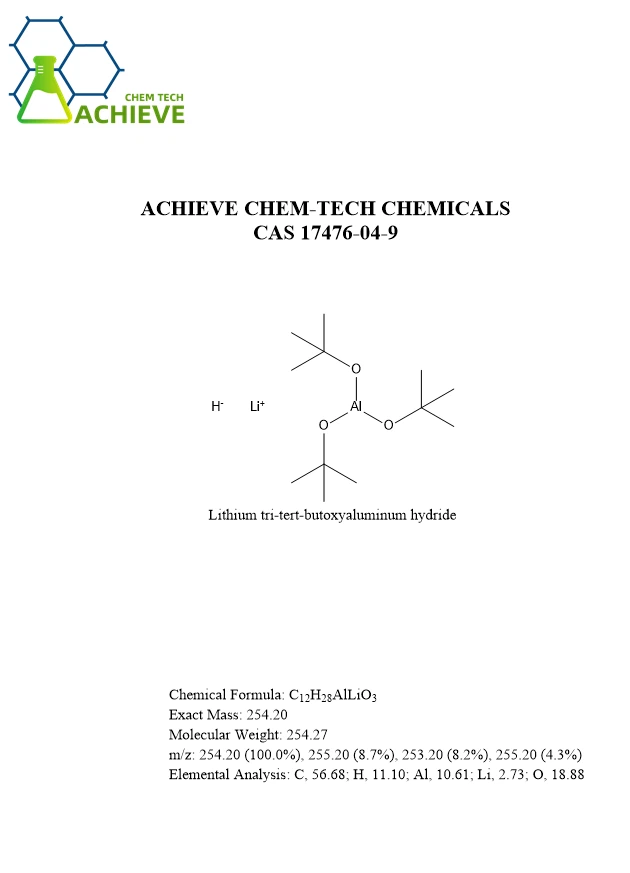
C12H28AlLiO3 |
|
Exact Mass |
254.20 |
|
Molecular Weight |
254.27 |
|
m/z |
254.20 (100.0%), 255.20 (8.7%), 253.20 (8.2%), 255.20 (4.3%) |
|
Elemental Analysis |
C, 56.68; H, 11.10; Al, 10.61; Li, 2.73; O, 18.88 |
|
Melting point |
300-319℃(dec.) (lit.) |
|
Boiling point |
66℃(THF) |
|
Density |
0.942 g/mL at 25℃ |
|
Storage Temperature |
2-8℃ |
 |
 |

Lithium tri tert butoxyaluminum hydride (LTBA), as a mild and highly selective organic metal reducing agent, has shown extensive application value in the field of organic synthesis. Its unique chemical properties enable it to efficiently reduce specific functional groups while avoiding interference with other sensitive groups, thus playing a key role in various fields such as medicine, fragrances, pesticides, dyes, and fine organic synthesis. The following is a detailed explanation of its purpose:
In pharmaceutical synthesis, LTBA is often used to construct the core skeleton of complex organic molecules, especially in the synthesis of steroid ketones, nucleoside prodrugs, and anticancer drug intermediates. Its high selective reduction ability can precisely control the reaction pathway, improve the purity and yield of the target product. LTBA has important applications in the reduction process of steroid ketones. For example, in the synthesis of nucleoside prodrugs for the treatment of hepatitis C virus (HCV), such as hydroxy-d-2 '- deoxy-2' - thiofluoro-2 '- chloro-C-methyl-3', 5 '- cyclophosphate, LTBA can selectively reduce specific functional groups in steroid ketones as a reducing agent, providing a key intermediate for subsequent functional group conversion. Its mild reaction conditions avoid the damage of other sensitive groups, ensuring the high efficiency and selectivity of synthesis. LTBA is one of the important raw materials for the production and treatment of cancer drug gemcitabine. In the synthesis pathway of gemcitabine, LTBA can efficiently reduce certain key intermediates and promote the generation of target products.

Application in the field of spice synthesis

In spice synthesis, LTBA is often used to construct organic molecules with specific aromas. Its high selective reducing ability can retain other sensitive groups in the molecule, ensuring that the aroma characteristics of the fragrance are not affected. LTBA performs well in synthesizing organic molecules with complex aromas. For example, when synthesizing certain fragrances with floral or fruity characteristics, LTBA can selectively reduce specific functional groups in the molecule while retaining other key functional groups, thereby ensuring the aroma characteristics of the fragrance. Its mild reaction conditions avoid the destruction of molecular structure and improve the quality of spices. LTBA is also commonly used in the synthesis of spice intermediates. These intermediates play a crucial role in subsequent spice synthesis. The high selectivity of LTBA makes the synthesis of intermediates more efficient and reliable, providing strong support for the final synthesis of spices.
In pesticide synthesis, LTBA is often used to synthesize organic molecules with specific biological activities. Its high selective reduction ability can ensure that the active groups of pesticide molecules are not damaged, while improving synthesis efficiency. LTBA has important applications in the synthesis of insecticides and herbicides. For example, when synthesizing certain organic molecules with high insecticidal or herbicidal activity, LTBA can selectively reduce specific functional groups in the molecule, retaining other key functional groups, thereby ensuring the activity of the pesticide. Its mild reaction conditions and high selectivity make the synthesis process more controllable and efficient. LTBA is also commonly used in the synthesis of pesticide intermediates. These intermediates play a crucial role in the subsequent synthesis of pesticides. The high selectivity of LTBA makes the synthesis of intermediates more efficient and reliable, providing strong support for the final synthesis of pesticides. Meanwhile, its mild reaction conditions also reduce the generation of by-products, improving the purity and quality of pesticides.

Application in the field of dye synthesis

In dye synthesis, LTBA is often used to synthesize organic molecules with specific colors and dyeing properties. Its high selective reduction ability can ensure that the chromophore groups of dye molecules are not damaged, while improving synthesis efficiency. LTBA has important applications in the synthesis of organic dyes. For example, when synthesizing certain dyes with bright colors and good dyeing performance, LTBA can selectively reduce specific functional groups in the molecule, retaining other key functional groups, thereby ensuring the color and dyeing performance of the dye. Its mild reaction conditions and high selectivity make the synthesis process more controllable and efficient. LTBA is also commonly used in the synthesis of dye intermediates. These intermediates play a crucial role in the subsequent synthesis of dyes. The high selectivity of LTBA makes the synthesis of intermediates more efficient and reliable, providing strong support for the final synthesis of dyes. Meanwhile, its mild reaction conditions also reduce the generation of by-products, improving the purity and quality of the dye.
Adverse reactions
Lithium Tri Tert Butoxyaluminum Hydride(LTBA) As an important organic metal reducing agent, it plays a crucial role in organic synthesis. However, its unique chemical properties also bring a series of potential safety risks and adverse reactions. The following are its adverse reactions:
Adverse reactions caused by physical and chemical properties
Intense reaction upon contact with water
LTBA is extremely sensitive to moisture and can react violently with water, releasing flammable hydrogen gas and potentially causing spontaneous combustion. This reaction not only leads to the failure of reagents, but may also cause fires or explosions, posing a serious threat to laboratory personnel and equipment.
Symptoms: During the reaction process, a large amount of heat and gas may be generated, causing the container to rupture or explode. Meanwhile, the released hydrogen gas may form an explosive mixture when mixed with air.
Response measures: When using LTBA, it is necessary to strictly avoid contact with water. All operations should be carried out under inert gas (such as argon) protection and using dry solvents and instruments.
Reacting with air
LTBA is prone to moisture absorption and hydrolysis in the air, leading to reagent deterioration. Hydrolyzed products may be corrosive and cause irritation to the skin and eyes.
Symptoms: Changes in reagent color, odor or precipitation.
Response measures: LTBA should be sealed and stored in a dry, cool place to avoid prolonged exposure to air. When retrieving reagents, they should be operated quickly and the exposure time of the reagents to the air should be minimized as much as possible.
HIGHLY FLAMMABLE
LTBA itself is highly flammable, with a low flash point, and is prone to causing fires under high temperature or open flame conditions.
Symptom: Bright flames and a large amount of smoke are produced when the reagent burns.
Response measures: When using LTBA, keep away from sources of fire and heat, and equip with corresponding fire-fighting equipment. Meanwhile, the experimental area should maintain good ventilation conditions to reduce the risk of fire.
Adverse reactions to human health
Skin contact
LTBA is corrosive to the skin and may cause symptoms such as skin burns, redness, and pain upon contact.
Symptoms: Red patches, blisters, ulcers, etc. appear on the skin contact area.
Response measures: If the skin accidentally comes into contact with LTBA, immediately rinse with plenty of water for at least 15 minutes and seek medical assistance. During the operation, appropriate protective clothing and gloves should be worn to avoid direct skin contact with the reagent.
Eye contact
LTBA has a strong irritant effect on the eyes, which may cause symptoms such as eye pain, tearing, redness, and swelling upon contact. In severe cases, it may even cause visual impairment.
Symptoms: Eye congestion, tearing, blurred vision, etc.
Response measures: If the eyes accidentally come into contact with LTBA, immediately rinse the eyes with plenty of water for at least 15 minutes and open the upper and lower eyelids to ensure thorough rinsing. Subsequently, immediate medical attention should be sought to avoid permanent damage to the eyes.
Inhale
If dust or smoke from LTBA is inhaled, it may cause irritation to the respiratory tract, leading to symptoms such as coughing and shortness of breath. Long term or excessive inhalation may also cause damage to the lungs.
Symptoms: cough, shortness of breath, chest tightness, etc.
Response measures: When using LTBA, it should be operated in a fume hood or well ventilated environment, and appropriate respiratory protective equipment should be worn. If you accidentally inhale dust or smoke from LTBA, you should immediately move to a place with fresh air and seek medical assistance.
Ingestion
If LTBA is ingested by mistake, it may cause serious irritation and damage to the digestive system, leading to symptoms such as nausea, vomiting, and abdominal pain. In severe cases, it may even endanger life.
Symptoms: Nausea, vomiting, abdominal pain, diarrhea, etc.
Response measures: If LTBA is accidentally ingested, rinse your mouth immediately and drink plenty of water to dilute the reagent. Subsequently, medical attention should be sought immediately, and the doctor should be informed of the name and amount of the reagents ingested, so that the doctor can take appropriate treatment measures.
Adverse reactions of environment and equipment
Pollution to the environment
If LTBA is discharged into the environment without proper treatment, it may cause pollution to water bodies and soil, affecting ecological balance.
Symptom: Elevated concentration of LTBA in water and soil, causing toxic effects on organisms.
Response measures: LTBA's waste should be properly disposed of in accordance with relevant regulations to avoid direct discharge into the environment. At the same time, cleaning and disinfection of the experimental area should be strengthened to reduce the impact of reagent residues on the environment.
Corrosion of equipment
LTBA may cause corrosion to experimental equipment, especially the parts in direct contact with reagents. This corrosion may cause equipment damage or performance degradation.
Symptoms: Corrosion marks and decreased sealing performance on the surface of the equipment.
Response measures: When using LTBA, corrosion-resistant materials should be selected to make experimental equipment, and the equipment should be regularly inspected and maintained. If corrosion is found on the equipment, the damaged parts should be replaced or repaired in a timely manner.
Adverse reactions during storage and transportation
Improper storage
If LTBA is stored improperly, such as at high temperatures, high humidity, or mixed with incompatible substances, it may cause reagent deterioration or safety accidents.
Symptoms: color change of reagents, odor, container rupture, etc.
Response measures: LTBA should be stored in a dry, cool, well ventilated place and kept away from sources of fire and heat. At the same time, incompatible substances such as acids and alcohols should be avoided during storage. During storage, the status of reagents and the integrity of containers should be checked regularly.
Transportation risk
LTBA may cause reagent leakage or safety accidents during transportation due to factors such as vibration, collision, or temperature changes.
Symptoms: reagent leakage, container rupture, fire or explosion, etc.
Response measures: When transporting Lithium Tri Tert Butoxyaluminum Hydride, a professional dangerous goods transportation company should be selected and the transportation vehicles should ensure compliance with relevant safety standards. At the same time, the reagents should be properly packaged and equipped with corresponding fire-fighting and emergency response equipment. During transportation, traffic rules and safety operating procedures should be strictly followed.
Hot Tags: lithium tri tert butoxyaluminum hydride cas 17476-04-9, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID, Cerium sulfate powder, CHICAGO SKY BLUE 6B, Consumable, Iridium III chloride