Bremelanotide (link:https://www.bloomtechz.com/synthetic-chemical/peptide/bremelanotide-powder-cas-189691-06-3.html) is a synthetic polypeptide hormone whose structure is similar to leptin, tyrosine and skin melanin that naturally exist in the human body. As a new type of drug, Bremelanotide has a regulating function and is often used in the treatment of erectile dysfunction and low libido. The use scenarios in daily life are becoming more and more extensive. With people's in-depth research on him, people have discovered many kinds of his synthesis methods, which will be analyzed one by one below.

1. Synthesized from L-tyrosine:
In the original synthesis method, the structure of Bremelanotide was obtained by deforming L-tyrosine. First, L-tyrosine is converted to dipeptidyl-L-tyrosine (Boc-Tyr(OCO)2-OH) in the presence of hydrofluoric acid, where Boc represents succinimidomethyl (t -butoxycarbonyl) group. Next, add benzyl alcohol (PhCH2OH), 1-(3-diethylaminopropoxy)-2-(2-methazolyl)ethane (EDC), and N-hydroxybutanediol in tetrahydrofuran (THF) Reagents such as imide (HOBt) can combine dipeptidyl-L-tyrosine with pseudopeptide alanine (Phe-Aib-Aib-Lys(Ac)-Gly-Lys(Ac)-Aib-Leu- NH2) was synthesized into Bremelanotide.
(1) Conversion of L-tyrosine to dipeptidyl-L-tyrosine:
First, dissolve L-tyrosine in water and add appropriate amount of hydrofluoric acid. Typically, the concentration of L-tyrosine is 1M and the amount of hydrofluoric acid is 5-10%. The mixture was allowed to stand at 0-4°C for 1 hour and then quenched.
Next, the obtained L-tyrosine-hydrofluoric acid complex was added dropwise to tetrahydrofuran. At the same time, an appropriate amount of reaction reagents such as benzyl alcohol, EDC and HOBt were added to tetrahydrofuran. After standing for 2 hours under stirring, dipeptidyl-L-tyrosine (Boc-Tyr(OCO)2-OH) can be obtained.
(2) Reaction of dipeptidyl-L-tyrosine with pseudopeptide alanine:
Dissolve the obtained dipeptidyl-L-tyrosine in N,N-dimethylformamide (DMF) and add the pseudopeptide alanine. Reaction conditions include temperature, reaction time, and concentration of reactants, etc. These factors will affect the yield and purity of the product. At a concentration of 225 g/L, the ratio of Boc-Tyr(OCO)2-OH to pseudopeptide alanine was 1:30.
After stirring and standing in -N,N-dimethylformamide for 24 hours, the reaction solution was poured into ice water and precipitated with acidic aldehyde solution. The result is the final product of Bremelanotide.
The synthesis method mainly has the following advantages: high product purity and simple production process. But this method also has some disadvantages, such as time-consuming, high cost and so on.
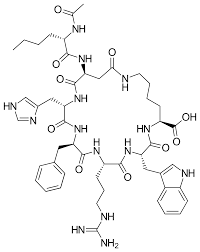
2. Solid phase synthesis:
Solid-phase synthesis is currently one of the most commonly used synthetic methods for Bremelanotide, and it is also a major synthetic method in the field of peptide synthesis. In solid-phase methods, polypeptide chains are built by successive additions of amino acid units. First, the first amino acid unit is modified into a sterically restrictive and non-degradable N-protecting group (such as Fmoc), which is then attached to the porous medium. Next, by introducing acrylic acid protecting group, acylation reaction and other steps, each amino acid unit is added on the porous medium, and a complete polypeptide chain is gradually constructed. Finally, in the steps of removing N-protecting group, denitrification, etc., a Bremelanotide polypeptide chain with a specific sequence can be prepared.
The solid-phase synthesis method mainly has the following advantages: high efficiency and high utilization rate of raw materials. But this synthetic method also has some disadvantages, such as multiple drying and washing steps are required in the production process, and these steps will lead to a decline in the quality of the product.
3. Liquid phase synthesis:
Liquid phase synthesis is another synthesis method of Bremelanotide, which is carried out through a liquid reaction system. The main steps of the synthesis method include raw material screening, drug molecule design, synthesis route selection, reaction, purification and the like. Compared with solid-phase synthesis, liquid-phase synthesis method can obtain Bremelanotide with higher purity and biological activity. In the liquid phase synthesis method, the commonly used reaction reagents are dihydroxyethyl mercaptan (EDT), ethoxycarbonylating agent (EEDQ), thioacetyl chloride, etc.
The liquid phase synthesis method mainly has the following advantages: simple operation and high product purity. However, this synthesis method also has some disadvantages, such as longer synthesis period and higher cost.
4. Physicochemical synthesis:
In the physicochemical synthesis method, Bremelanotide is synthesized by physical and chemical methods. This method mainly has two different approaches: one is to realize the synthesis of Bremelanotide through repeated operation of solid-liquid phase transformation; the other is to promote the self-assembly and polymerization of polypeptide chains through physical and mechanical compression, thereby synthesizing Bremelanotide .

Reaction mechanism:
The synthesis of Bremelanotide is mainly by converting L-tyrosine into L-phenolic acylate, then synthesizing the L-phenolic acylate and pseudopeptide, and finally obtaining the product. The mechanism of the reaction is as follows:
(1) Conversion of L-tyrosine into L-phenol acylate:
L-tyrosine reacts with sodium azide in THF solution to generate L-phenol acylate. Sodium azide will convert the hydroxyl group in tyrosine to an azide group, and then acidic treatment will convert the azide group to a phenolic acylate.
(2) L-phenol acylate is converted into acid chloride:
L-phenolic acylate reacts with acetyl chloride in THF to produce acid chloride. Five times the amount of TBHS is to absorb the generated HCl. Acid chlorides are reaction intermediates that are required for subsequent reactions.
(3) Synthesis of acid chloride and pseudopeptide:
The intermediate acid chloride reacts with the pseudopeptide in DMF to generate the polypeptide chain of Bremelanotide. What happens in this step is an anhydride condensation reaction, in which HOBt is the catalyst.
(4) Purification and analysis of the product:
The final product also needs to be purified and analyzed to ensure the purity and quality of the compound.
The physicochemical synthesis method mainly has the following advantages: simple operation and short preparation period. However, this synthetic method also has some disadvantages, such as low product purity and insufficient product diversity.
In conclusion, the synthesis methods of Bremelanotide are diverse and flexible, and the selection depends on many factors, such as reaction conditions, synthesis cost and reaction time, etc. Regardless of the method used, stable reaction conditions and high-purity products are the keys to the successful synthesis of Bremelanotide.

