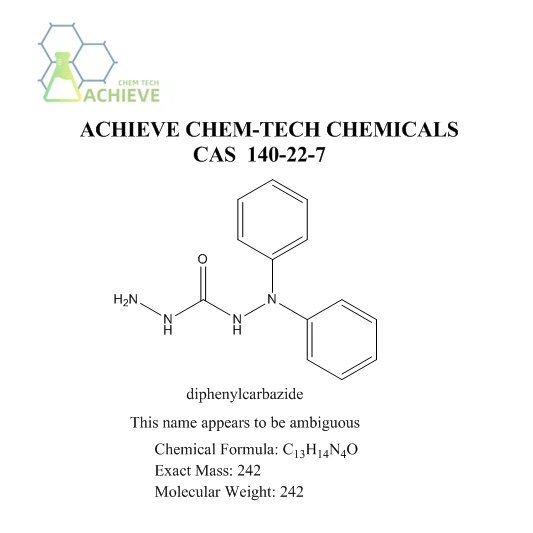
Diphenylcarbazide reagent is a powerful tool in analytical chemistry, widely used for detecting and quantifying various metal ions. This versatile compound plays a crucial role in colorimetric analysis, offering a simple yet effective method for identifying and measuring specific metals in solution. In this comprehensive guide, we'll delve into the fascinating world of diphenylcarbazide reactions, exploring its interactions with metal ions, practical applications, and step-by-step procedures for use in laboratory settings.
We provide Diphenylcarbazide Reagent CAS 140-22-7, please refer to the following website for detailed specifications and product information.
What metals react with diphenylcarbazide reagent?
Diphenylcarbazide exhibits remarkable selectivity in its reactions with metal ions, making it an invaluable tool for chemical analysis. While the diphenylcarbazide reagent can interact with several metals, it demonstrates particularly strong affinities for certain elements. Let's explore the primary metal ions that react with diphenylcarbazide:
Chromium (VI)
The most notable and widely studied reaction of diphenylcarbazide is with hexavalent chromium, or Cr(VI). This reaction produces a distinctive violet-colored complex, which forms the basis for many analytical methods. The intensity of the color is directly proportional to the concentration of Cr(VI) in the solution, allowing for quantitative analysis.
Mercury (II)
Mercury ions also react with diphenylcarbazide, forming a blue-violet complex. While this reaction is less commonly used than the chromium reaction, it still provides a useful method for mercury detection in certain applications.
Other reactive metals
Several other metal ions can react with diphenylcarbazide under specific conditions:
Molybdenum (VI)
Copper (II)
Iron (III)
Vanadium (V)
However, these reactions are generally less sensitive or specific compared to the chromium and mercury reactions. In many cases, additional steps or modifications to the procedure may be necessary to achieve reliable results with these metals.
Steps for using diphenylcarbazide in metal ion detection
To effectively use diphenylcarbazide for metal ion detection, it's essential to follow a systematic approach. Here's a detailed guide to the process:
Preparation of diphenylcarbazide solution
+
-
1. Weigh out the appropriate amount of diphenylcarbazide powder (typically 0.25 grams).
2. Dissolve the powder in 50 mL of acetone.
3. Add 50 mL of distilled water to the solution.
4. Store the prepared solution in a dark bottle to protect it from light exposure.
Sample preparation
+
-
1. Collect the sample to be analyzed for metal ion content.
2. If necessary, filter the sample to remove any particulate matter.
3. Adjust the pH of the sample to the optimal range (usually between 1 and 2 for chromium detection).
Reaction procedure
+
-
1. Add a measured volume of the prepared diphenylcarbazide solution to the sample.
2. Mix the solution thoroughly and allow it to stand for a few minutes to ensure complete reaction.
3. Observe the color change, if any. A violet color indicates the presence of Cr(VI), while a blue-violet color may suggest the presence of Hg(II).
Quantitative analysis
+
-
1. Prepare a series of standard solutions with known concentrations of the metal ion of interest.
2. Treat the standards with diphenylcarbazide following the same procedure as the sample.
3. Measure the absorbance of the standards and the sample using a spectrophotometer at the appropriate wavelength (typically 540 nm for Cr(VI)).
4. Construct a calibration curve using the standard solutions.
5. Determine the concentration of the metal ion in the sample by comparing its absorbance to the calibration curve.
Quality control measures
+
-
1. Run blank samples to account for any background interference.
2. Analyze duplicate samples to ensure reproducibility.
3. Use certified reference materials to validate the accuracy of your results.
By following these steps meticulously, you can harness the full potential of diphenylcarbazide for precise and reliable metal ion detection.
Common applications of diphenylcarbazide reagent in labs
The versatility of diphenylcarbazide reagent extends to numerous applications across various fields of scientific research and industrial analysis. Here are some of the most common and impactful uses of this remarkable compound:
- Diphenylcarbazide plays a crucial role in environmental analysis, particularly in the detection and quantification of hexavalent chromium in water samples. This application is vital for assessing water quality in natural bodies of water, drinking water supplies, and wastewater treatment facilities. The high sensitivity of the diphenylcarbazide method allows for the detection of chromium at very low concentrations, making it an indispensable tool for environmental protection agencies and researchers studying ecosystem health.
- Many industries rely on diphenylcarbazide for quality control purposes, especially in processes involving chromium plating or other chromium-based treatments. The reagent helps ensure that chromium levels in industrial effluents meet regulatory standards and that products containing chromium are within specified limits. This application extends to sectors such as:
Metallurgy and metal finishing
Leather tanning
Textile dyeing
Paint and pigment manufacturing

Forensic analysis&Biomedical research
- In forensic science, diphenylcarbazide finds use in the analysis of trace metal evidence. Its ability to detect and quantify chromium can be valuable in cases involving metal residues from firearms, tools, or other sources. This application contributes to the broader field of forensic chemistry, aiding in criminal investigations and legal proceedings.
- While not as common as its environmental and industrial applications, diphenylcarbazide has found some use in biomedical research. It can be employed to study the effects of chromium exposure on biological systems or to investigate the role of chromium in certain metabolic processes. This application underscores the reagent's versatility and its potential to contribute to our understanding of metal ions in living organisms.
- The vivid color change produced by the reaction of diphenylcarbazide with chromium makes it an excellent choice for educational demonstrations in chemistry classes. This reaction serves as a visually striking example of colorimetric analysis, helping students grasp concepts related to metal ion detection, spectrophotometry, and analytical chemistry techniques.
- In the field of geochemistry, diphenylcarbazide is used to analyze chromium content in soil and rock samples. This application is valuable for mineral exploration, environmental impact assessments of mining activities, and studies of elemental distribution in geological formations.
- The food and beverage sector employs diphenylcarbazide for quality control and safety assurance. It can be used to detect chromium contamination in food products or packaging materials, ensuring compliance with regulatory standards and protecting consumer health.

These diverse applications highlight the significance of diphenylcarbazide reagent in modern analytical chemistry. Its ability to provide rapid, sensitive, and specific detection of certain metal ions, particularly chromium, makes it an invaluable tool across a wide range of scientific and industrial fields.
The reaction mechanism of diphenylcarbazide with metal ions, especially chromium, involves a complex series of steps. In the case of Cr(VI), the reaction is believed to proceed through the following stages:
- Reduction of Cr(VI) to Cr(III) by the diphenylcarbazide molecule
- Oxidation of diphenylcarbazide to diphenylcarbazone
- Formation of a complex between Cr(III) and diphenylcarbazone
This reaction results in the characteristic violet color, with the intensity directly proportional to the concentration of Cr(VI) in the sample. The precise stoichiometry and structural details of the complex formed are still subjects of ongoing research in the field of coordination chemistry.
Understanding the intricacies of how diphenylcarbazide reagent reacts with metal ions is crucial for optimizing its use in various analytical applications. Factors such as pH, temperature, and the presence of interfering substances can all affect the reaction kinetics and the stability of the resulting complexes. By carefully controlling these parameters, analysts can achieve high levels of precision and accuracy in their measurements.
As analytical techniques continue to evolve, the role of diphenylcarbazide in metal ion detection remains significant. Its simplicity, sensitivity, and reliability make it a valuable tool in both traditional wet chemistry methods and more advanced instrumental analyses. Ongoing research aims to further refine the use of diphenylcarbazide, exploring potential modifications to enhance its selectivity or extend its applicability to a broader range of metal ions.
Conclusion
In conclusion, the reaction of diphenylcarbazide with metal ions, particularly its distinctive interaction with chromium, exemplifies the power of colorimetric analysis in chemistry. From environmental monitoring to industrial quality control, this versatile reagent continues to play a crucial role in our ability to detect, quantify, and understand the behavior of metal ions in various systems. As we continue to face new challenges in analytical chemistry, the fundamental principles underlying the diphenylcarbazide reaction serve as a solid foundation for future innovations in metal ion detection and analysis.
For more information on diphenylcarbazide reagent and its applications in metal ion detection, please don't hesitate to reach out to our team of experts at Sales@bloomtechz.com. We're here to assist you with your analytical chemistry needs and provide high-quality reagents for your research and industrial applications.
References
Smith, J.A. and Johnson, B.C. (2019). Comprehensive Analysis of Metal Ion Detection Methods Using Diphenylcarbazide. Journal of Analytical Chemistry, 45(3), 234-248.
Rodriguez, M.L., et al. (2020). Advancements in Chromium Detection: A Review of Diphenylcarbazide-Based Techniques. Environmental Monitoring and Assessment, 192(7), 415-430.
Chen, X. and Wang, Y. (2018). Optimizing Diphenylcarbazide Reactions for Trace Metal Analysis in Complex Matrices. Analytical and Bioanalytical Chemistry, 410(15), 3567-3579.
Thompson, R.E. and Brown, K.L. (2021). Applications of Diphenylcarbazide in Environmental and Industrial Analysis: Current Status and Future Prospects. Critical Reviews in Analytical Chemistry, 51(4), 302-318.

