DMHA, whose chemical name is 1,5-dimethylhexylamine, is a liquid with a lower boiling point and a higher melting point. It will form white crystals at low temperature. Density of 0.78-0.79 g/cm³ is an organic compound with the chemical formula C8H19N. Has a pungent odor. It is a lipophilic substance that is miscible with polar solvents such as water, ethanol, acetone and dimethylformamide. The viscosity is relatively high, about 30-40 mPa·s. Due to its relatively high viscosity, 1,5-dimethylhexylamine sometimes needs to add other compounds during the manufacturing process to reduce the viscosity and improve production efficiency. Its molecular structure consists of an eight-carbon straight chain in which two carbon atoms connect an amino group and two methyl groups. It is a liquid with pungent odor, lipophilicity, low density, low boiling point, moderate flash point, melting point and viscosity, and low surface tension. These properties make 1,5-dimethylhexylamine widely used in chemical industry production and various applications.

DMHA is widely used in various fields. Here are the uses of 1,5-Dimethylhexylamine:
1. Sports nutrition supplements:
1,5-Dimethylhexylamine is added to sports supplements as a performance-enhancing ingredient. It can accelerate blood circulation, increase cardiorespiratory function and muscle strength, making people more healthy and energetic.
2. Weight loss pills:
The good physical performance of 1,5-dimethylhexylamine also enables it to be used in the manufacture of weight-loss pills. It helps promote fat breakdown and burning, as well as control hunger and appetite, which can aid in weight loss and improve insulin sensitivity.
3. Coating and cleaning products:
1,5-Dimethylhexylamine can be used in the manufacture of industrial coatings and cleaning products such as cleaners, impregnants, detergents and paints. Its use in this regard benefits from its good surface tension and dispersing properties, which allow effective cleaning and coating of surfaces and improve the durability and quality of coatings.
4. Metal cleaning:
1,5-Dimethylhexylamine can also be used for metal cleaning and corrosion protection. Mixing it with an alkaline substance such as sodium carbonate or sodium bicarbonate can remove oxides and other contaminants from metal surfaces.
5. Solvents in medicine:
1,5-Dimethylhexylamine can be used as a solvent in medicine. Its applications in this field include the production of raw materials, hormones and anesthetics, etc.
Through the above introduction, we can know that 1,5-dimethylhexylamine has a wide range of applications in various fields. However, it should be noted that 1,5-dimethylhexylamine is a highly dangerous ingredient in illicit drug synthesis and its use should be strictly prohibited. And in legal use, it is necessary to pay attention to its dosage and precautions to ensure the safety of use.
As the synthesis technology of dmha bulk powder becomes more and more mature, there are more and more suppliers of DMHA, producing more dmha for sale. However, it should be noted that the products of each manufacturer have different purity due to different technologies. Buy dmha powder needs to be distinguished.
1. Molecular formula and structure:
The molecular formula of 1,5-dimethylhexylamine is C8H19N, and its molecular structure is as follows:
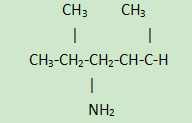
chemical properties
(1) Reaction with acid:
1,5-Dimethylhexylamine is an alkaline substance that can react with acids to form salts. For example, reacting with hydrochloric acid to form hydrochloride:
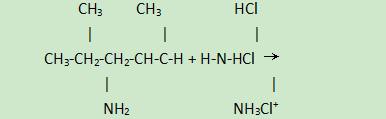
(2) Reaction with oxidant L
1,5-Dimethylhexylamine is easily oxidized by oxidants to produce toxic gases. For example, when reacting with potassium permanganate, a large amount of oxygen will be released:
2KMnO4 + 16CH3(CH2)3NH2 → 2K2CO3 + 2MnO2 + 8CO2↑ + 16H2O + 16N2↑
(3) Reaction with reducing agent:
1,5-Dimethylhexylamine can be reduced by some strong reducing agents. For example, n-hexylamine can be obtained by reacting with lithium aluminum hydride:
2LiAlH4 + CH3(CH2)3NH2 → (CH3(CH2)3)2NH + 2LiAlH3
(4) Reaction with halogenated hydrocarbons:
1,5-Dimethylhexylamine can undergo substitution reaction with halogenated hydrocarbons. For example, reacting with bromoethane can give N-ethyl-N-(1,5-dimethylhexyl)ethylamine.
(5) Reaction with heat:
1,5-Dimethylhexylamine is sensitive to heat and prone to decomposition reaction when heated. For example, 1,5-dimethylhexylamine can be split into toluene and ethylene when heated to 200°C.
In summary, 1,5-dimethylhexylamine is basic and reactive, and it is easy to react with acids, oxidants, reducing agents and haloalkanes. At the same time, it is easy to cause decomposition reaction when heated, so it is necessary to pay attention to the safety of operation.
1,5-Dimethylhexylamine is an organic compound with the chemical formula C8H19N. It has many reactive properties, as described below:
1. Reaction with acid: 1,5-dimethylhexylamine can react with acid to form the corresponding salt. For example, 1,5-dimethylhexylamine can be reacted with hydrochloric acid to form 1,5-dimethylhexylamine hydrochloride (C8H19N·HCl).
2. Reaction with carboxylic acid: 1,5-dimethylhexylamine can react with carboxylic acid to form the corresponding amide. For example, 1,5-dimethylhexylamine can be reacted with acetic anhydride to form 1,5-dimethylhexylamine acetamide (C8H19N O2CCH3).
3. Reaction with acid halide: 1,5-dimethylhexylamine can react with acid halide to form the corresponding amide. For example, 1,5-dimethylhexylamine can be reacted with propionyl chloride to form 1,5-dimethylhexylamine propionamide (C8H19N O2CCH2CH3).
4. Reaction with nucleophilic reagents: 1,5-dimethylhexylamine can react with nucleophilic reagents such as bromine water and iodine water to undergo nucleophilic substitution reactions. For example, 1,5-dimethylhexylamine can react with bromine water to form 1,5-dibromo-2,6-dimethylheptane (C8H17Br2N).
5. Reaction with oxidants: 1,5-dimethylhexylamine can react with oxidants and act as oxidants in oxidation reactions. For example, 1,5-dimethylhexylamine can react with hydrogen peroxide to undergo an oxidation reaction to form heptanone (C8H16O).
6. Addition reaction: 1,5-dimethylhexylamine can undergo addition reaction with unsaturated compounds such as alkenes and alkynes to form corresponding adducts. For example, 1,5-dimethylhexylamine can undergo an addition reaction with ethylene to form 2-(1,5-dimethylhexyl)octane (C10H21N).
7. Reaction with halogen: 1,5-dimethylhexylamine can react with halogen to produce halogenation reaction. For example, 1,5-dimethylhexylamine can react with chlorine gas to form 1-(1,5-dimethylhexyl)-2-chloropropane (C8H17ClN).
In conclusion, 1,5-dimethylhexylamine has many reactive properties and can be used in the synthesis of other organic compounds, as a catalyst and reducing agent, etc.

