Amid the wave of anti-aging research, NAD+ (nicotinamide adenine dinucleotide) and MOTS-C (mitochondrial-derived peptide) stand as two pivotal molecules, reshaping human understanding of life's aging process through their unique mechanisms. The former, acting as the "commander" of cellular energy metabolism, and the latter, serving as the "messenger" facilitating communication between mitochondria and the cell nucleus, demonstrate remarkable potential through synergistic effects in energy regulation, metabolic balance, and disease intervention.
NAD+ is an essential enzyme in the body that regulates various cell functions in the body. NAD+ stands for nicotinamide adenine dinucleotide and is present in all living cells in the human body.
It is necessary for many body functions and cellular processes, including metabolism, immune health, and DNA repair. Practitioners also use it to slow the signs of aging, boost energy levels, and improve mental health.

MOTS-c targets skeletal muscle and can enhance glucose metabolism. Therefore, MOTS-c plays an important role in the regulation of cardiovascular, diabetes, exercise and longevity. It is a new mitochondrial signaling mechanism and plays a role in regulating intracellular and intercellular metabolism.

Business Process
|
|
|
|
|
|
NAD+: The "Core Hub" of Cellular Energy

From Energy Powerhouse to DNA Guardian
NAD+ is one of the most critical coenzymes within cells, its molecular structure formed by the connection of nicotinamide and adenine nucleotides via a phosphodiester bond. In energy metabolism, NAD+ accepts hydrogen atoms during the tricarboxylic acid cycle of aerobic respiration to form NADH. It then transfers electrons to oxygen via the mitochondrial respiratory chain, ultimately generating ATP-the "energy currency" of cells. If this process is disrupted, cells face an energy crisis leading to functional decline.
The "Molecular Marker" of Aging
NAD+ levels plummet dramatically with age. Studies indicate that while humans possess approximately 6-8 grams of NAD+ at age 20, this drops to less than 2 grams by age 60. This decline directly causes mitochondrial dysfunction, accumulated DNA damage, and heightened inflammatory responses, ultimately triggering age-related diseases such as metabolic syndrome, neurodegenerative disorders, and cardiovascular diseases.

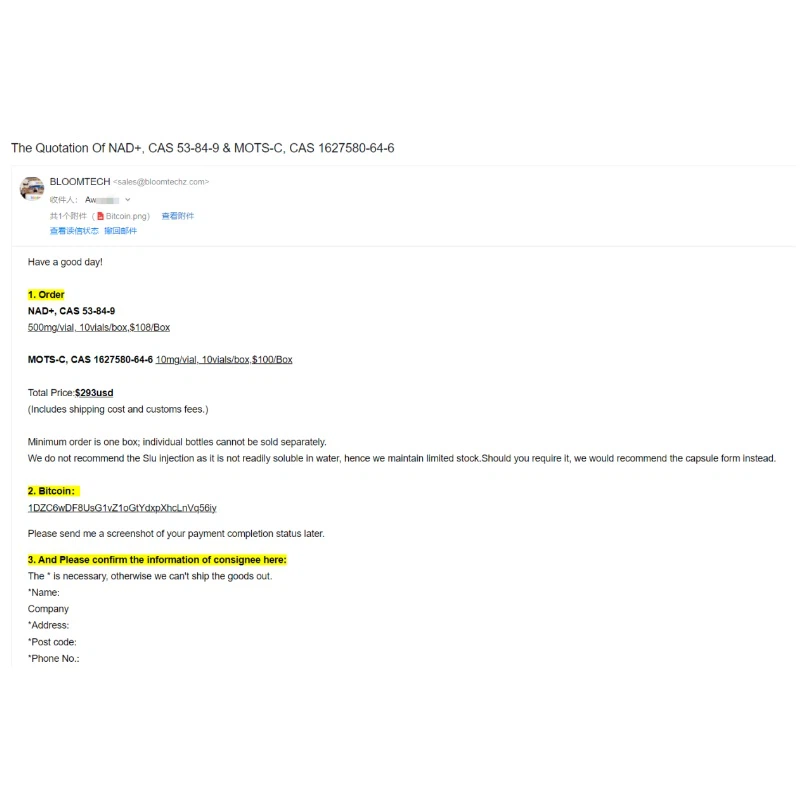
The "Golden Pathway" for Clinical Application
Currently, NAD+ levels are primarily elevated through three approaches:
Precursor Supplementation: Precursor molecules like NMN and NR (nicotinamide riboside) can be directly converted into NAD+ by cells. Among these, NMN has become the mainstream supplement due to its high bioavailability and excellent safety profile.
Inhibiting Consumption: The CD38 enzyme is the primary consumer of NAD+, and its activity significantly increases with age. CD38 inhibitors like quercetin and apigenin can reduce this inefficient NAD+ depletion.
MOTS-C: The "Anti-Aging Code" of Mitochondria
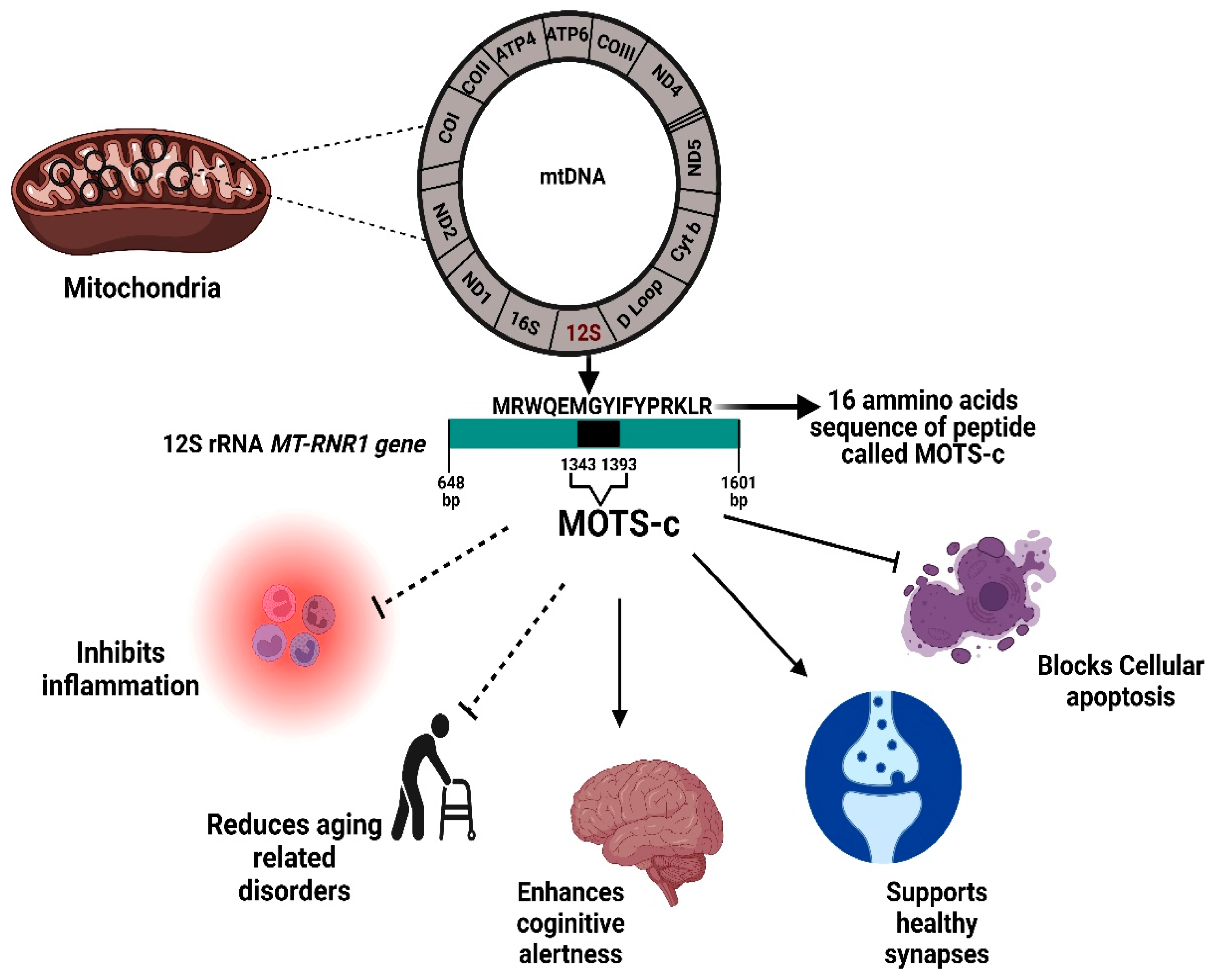
The "Interlocutor" Between Mitochondria and the Nucleus
MOTS-c is a 16-amino acid peptide encoded by mitochondrial DNA, with the sequence MRWQEMGYIFYPRKLR. As a mitochondrial signaling peptide, MOTS-c traverses the cell membrane to directly regulate gene expression in the nucleus. Its core mechanism involves activating the AMPK (Adenosine Monophosphate-Activated Protein Kinase) pathway, enhancing insulin sensitivity, and improving glucose and lipid metabolism.
The "Natural Regulator" of Metabolic Diseases
MOTS-c exhibits multifaceted effects in metabolic regulation:
Glucose Metabolism: By inducing accumulation of the AMP analog AICAR, it activates AMPK, promotes skeletal muscle glucose uptake, and lowers blood glucose levels. Animal studies show MOTS-c injections reduce blood glucose by 40% in diabetic mice.
Lipid Metabolism: MOTS-c inhibits fatty acid synthesis, promotes fatty acid β-oxidation, and reduces fat accumulation. In obese mouse models, 8 weeks of MOTS-c treatment resulted in 15% weight loss and a 22% reduction in fat content.


A "Potential Intervention Target" for Age-Related Diseases
MOTS-c levels decline significantly with age, and its deficiency is closely associated with aging phenotypes such as insulin resistance, cognitive decline, and muscle atrophy. Restoring MOTS-c levels reverses these phenotypes:
Neuroprotection: MOTS-c crosses the blood-brain barrier, inhibits neuronal apoptosis, and improves cognitive function in Alzheimer's disease mice.
Muscle Regeneration: MOTS-c activates the mTOR pathway to promote muscle stem cell proliferation, alleviating age-related muscle atrophy.
Longevity Extension: In nematode models, MOTS-c overexpression extends lifespan by 20%; in mouse studies, MOTS-c treatment increases male lifespan by over 10%.
Synergistic Anti-Aging
Complementarity in Metabolic Pathways
NAD+ and MOTS-C form a complementary network in metabolic regulation:
NAD+ regulates mitochondrial biogenesis via Sirtuin proteins, while MOTS-C directly acts on mitochondria to enhance their function;
NAD+ serves as a substrate for PARP enzymes to repair DNA damage, while MOTS-C reduces DNA damage triggers by suppressing inflammatory responses;
MOTS-C elevates NAD+ levels, further activating Sirtuin proteins to create a positive feedback loop.
Synergistic Effects in Disease Intervention
In treating metabolic syndrome, combining NAD+ precursors with MOTS-c yields synergistic outcomes:
Diabetes: NMN elevates NAD+ levels to improve insulin secretion; MOTS-c enhances insulin sensitivity, jointly regulating blood glucose.
Non-alcoholic fatty liver disease: NAD+ promotes fatty acid oxidation while MOTS-c inhibits fat synthesis, jointly reducing hepatic lipid accumulation.
Cardiovascular disease: NAD+ repairs vascular endothelial damage and MOTS-c lowers atherosclerosis risk, collectively safeguarding cardiovascular health.
The Next Frontier in Anti-Aging Research
The combined application of NAD+ and MOTS-c has now entered preclinical research. For instance, in trials targeting age-related muscle atrophy, mice in the combined therapy group exhibited a 30% increase in muscle strength and an 18% extension in lifespan compared to monotherapy groups. Looking ahead, advancements in gene editing technologies (such as CRISPR-Cas9) may enable more precise anti-aging interventions by regulating NAD+ synthase (e.g., NAMPT) or MOTS-c gene expression.
Peptides are no longer just biological building blocks-they are the keystones of a healthier, more sustainable future. From curing cancer and reversing aging to powering solar cells and biodegradable plastics, these tiny molecules are rewriting the rules of science and industry.
The next decade will see peptides go mainstream, with oral drugs, personalized anti-aging serums, and AI-designed therapies becoming part of daily life. Yet, to harness their full potential, we must overcome technical barriers, ensure equitable access, and navigate ethical minefields.
One thing is certain: the peptide revolution is here, and it's only just beginning. As research accelerates, we may soon live in a world where chronic diseases are obsolete, aging is optional, and sustainability is built into every product we use.
The future of health, energy, and materials science isn't in pills or plastics-it's in the elegant dance of amino acids. Welcome to the peptide century.