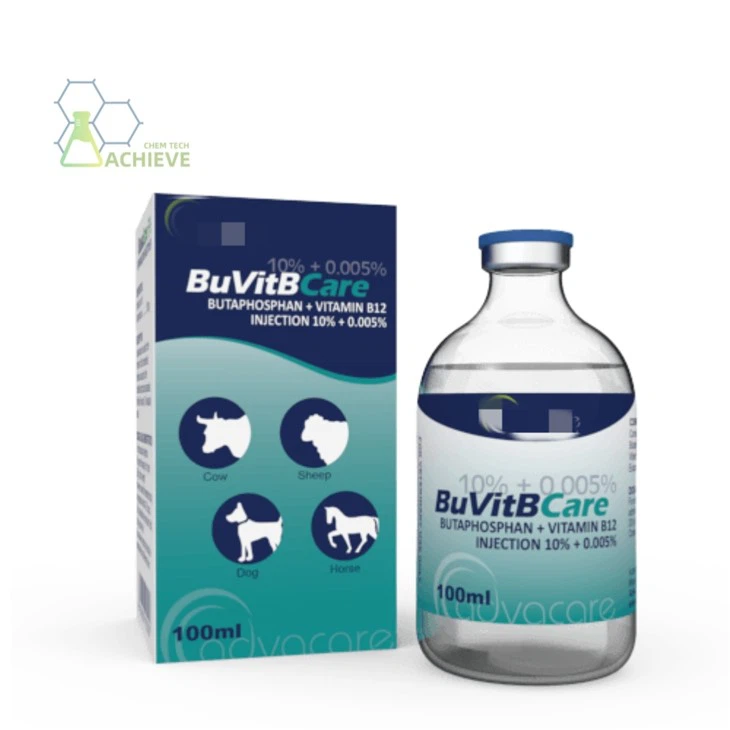
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of butafosfan injection in China. Welcome to wholesale bulk high quality butafosfan injection for sale here from our factory. Good service and reasonable price are available.
Butafosfan Injection is an organic phosphorus compound supplement widely used in veterinary clinical practice. Its core component is a compound of dimethylamino phosphate and vitamin B12. It mainly regulates the metabolic function of the liver, promotes energy metabolism and anabolic processes within the body, and effectively improves the weak state of the organism caused by metabolic disorders or nutritional imbalance. This preparation is commonly used to assist in the treatment of production stress, loss of appetite, and chronic wasting diseases in economic animals such as cattle and pigs. It helps restore physical strength and improve production performance. Its injectable formulation ensures rapid absorption and efficient utilization of the drug. As a supportive therapy, it must be combined with the main treatment targeting the cause and used under the guidance of veterinarians to ensure animal welfare and food safety. It is an important auxiliary treatment method in modern intensive livestock farming.

It is a water-soluble sterilization preparation made by mixing butafos and vitamin B12. Butaphosphat is an effective organic phosphorus supplement, and vitamin B12 can improve the metabolism of protein, carbohydrates, and fats, as well as enhance the synthesis of red blood cells. The combination of the two can improve energy utilization efficiency and regulate the imbalance of trace elements.
Formulation specifications and packaging
The commonly used clinical preparation is 10% injectin (100ml containing 10.0g of Butaphosphat), supplemented with vitamin B12 (0.00725g), sodium hydroxide (pH regulator), methyl paraben (0.1g, preservative) and other ingredients. The packaging adopts the standard configuration of 25kg fiber cardboard drum lined with plastic bag, and small size packaging can be customized according to customer needs. This formulation design balances drug stability and clinical convenience.
Our product




Additional information of chemical compound:
| Product Name | Butafosfan Powder | Butafosfan Injection |
| Product Type | Powder | Injection |
| Product Purity | ≥99% | ≥99% |
| Product Specifications | Customizable | Customizable |
| Product Package | Customizable | Customizable |
Our Product




Butafosfan+. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Butafosfan | |
|
CAS No. |
17316-67-5 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|
|
|
||


Application of livestock and poultry breeding
In pig farming, in response to postpartum weakness in sows and weaning stress in piglets, butafosfan injection of butafos can significantly increase feed intake (by 15% to 20%) and shorten the postpartum recovery period by 2-3 days. The practice of broiler farming has shown that the weight of breast and leg muscles has increased by 23.9% and 24.7% respectively, while reducing the feed to meat ratio by 0.15-0.2. In cow breeding, injection on the day of calving can reduce the incidence rate of subclinical ketosis by 35%, and the recovery time after gastric transposition can be shortened by 40%.
Companion Animal Medical
Clinical applications in dogs and cats have shown that this drug has significant therapeutic effects on postoperative rehabilitation, transportation stress, and elderly animal frailty syndrome. In specific cases, the recovery time of white blood cell count in surgical dogs after injection was shortened by 50%, and the incidence of stress-induced diarrhea was reduced by 60%. For competitive dogs, continuous use can increase exercise endurance by 15% to 20% and shorten muscle fatigue recovery time by 30%.


Special economic animals
In fur animal farming, butafos can improve fur quality, increase mink fur density by 12%, and increase fox fur grade by 0.5-1 levels. Aquaculture experiments have shown that after injectin, the spawning rate of Nile tilapia increases by 25%, the survival rate of juvenile fish increases by 18%, and the growth rate accelerates by 20%. These data validate its extensive application value in the field of special aquaculture.
Typical case analysis
Treatment of ketosis in dairy cows
Fifty perinatal cows from a large-scale ranch were treated with Butaphosphate Injectin (15ml/head, qd × 5d) in combination with Calcium Gluconate. The results showed that the blood ketone level decreased from 3.2mmol/L to 1.8mmol/L, the feed intake increased by 25%, and the recovery time of milk production was shortened by 4 days.
Weaning stress in piglets
300 weaned piglets at 21 days of age were randomly divided into a control group and an experimental group. The experimental group was injected with butafos (2ml/head, qd × 3d). Result: The diarrhea rate in the experimental group decreased by 18%, daily weight gain increased by 22%, and survival rate increased by 5%.
Racing fatigue recovery
8 endurance horses, immediately injected with Butaphosphat (20ml/horse, qd × 2d) after the race. Monitoring indicators show that creatine kinase (CK) levels decreased by 40%, lactate clearance rate increased by 35%, and the average performance of the next day's competition improved by 1.5 places.

Usage and dosage specifications
Principle of dose calculation
The basic dosage standard is: 10-25ml/time for horses and cows, 2.5-8ml/time for sheep, 2.5-10ml/time for pigs, 1-2.5ml/time for dogs, and 0.5-5ml/time for cats. In practical applications, individualized adjustments should be made based on animal weight, physiological status, and severity of the disease. For example, newborn piglets are calculated at 0.1ml/kg body weight, while adult cows are calculated at 0.05ml/kg body weight.
Treatment course setting standards
For acute metabolic disorders, butafosfan injection is recommended to use it continuously for 3-5 days, once a day; Chronic diseases require continuous treatment for 7-14 days, once every 2 days;
Preventive medication can start 24 hours before a stress event and last for 3 days. After the course of treatment, blood routine, liver and kidney function and other indicators should be monitored to evaluate the treatment effect.
Selection of administration route
Intravenous injectin is suitable for acute cases and can quickly reach the peak blood drug concentration; Intramuscular injectin is suitable for routine treatment, with an absorption half-life of approximately 1.2 hours; Subcutaneous injectio is suitable for chronic conditioning and can last for up to 24 hours. There are differences in bioavailability among different routes of administration: intravenous injectin is 100%, intramuscular injectin is about 85%, and subcutaneous injectin is about 75%.


Pharmacokinetic characteristics
Absorption and distribution
30 minutes after intramuscular injection, the peak blood concentration of the drug is reached, with a bioavailability of 82% to 88%. The distribution volume (Vd) is 0.6~0.8L/kg, widely distributed in tissues such as liver, kidney, and muscle. The protein binding rate is about 25%, mainly binding to albumin.
Metabolism and excretion
Mainly metabolized by the CYP450 enzyme system in the liver, producing inactive metabolites. The half-life (t ₁/₂) is 6-8 hours, and the clearance rate (CL) is 0.15~0.2 L/kg/h. About 70% is excreted through the kidneys, 25% through bile, and 5% in its original form. Patients with renal insufficiency need to adjust their dosage.
Consideration for special populations
Metabolism slows down in elderly animals, with a half-life extended to 10-12 hours; The clearance rate of young animals increases, and the half-life is shortened to 4-6 hours. Pregnant animals can pass through the placental barrier, while lactating animals can secrete milk. It is necessary to weigh the pros and cons when using it.
Drug interactions
Collaborative efficiency combination
Combined with vitamin B ₁₂, it can enhance the production of red blood cells and increase hemoglobin concentration by 15% to 20%. Combined with energy supplements such as ATP and coenzyme Q ₁₀, muscle endurance can be improved by 20% to 25%. Combined use with electrolyte supplements can improve symptoms of metabolic acidosis.
Antagonism and Taboos
Avoid simultaneous use with calcium preparations as it may form insoluble calcium phosphate precipitates. Combined with tetracycline antibiotics, it can reduce their absorption rate by 30% to 40%. Be cautious when used in animals with severe liver and kidney dysfunction, as it may prolong the accumulation time of drugs.
Suggestions for monitoring indicators
During the combination therapy, blood routine, liver and kidney function, electrolytes and other indicators need to be monitored. It is recommended to conduct a comprehensive evaluation before, during (day 3), and after (day 7) treatment. Adjust the dosage or discontinue medication promptly when abnormalities occur.

Adverse reactions and treatment
1. Common adverse reactions
Pain at the injectin site (incidence rate of about 15%), transient diarrhea (8%), and temporary decrease in feed intake (5%). Most reactions are mild and resolve on their own within 24-48 hours.
2. Serious adverse reactions
Allergic reactions (incidence<0.1%), manifested as rash, difficulty breathing, and decreased blood pressure. Immediately discontinue medication and administer rescue measures such as adrenaline and glucocorticoids. Long term high-dose use may lead to phosphate deposition, and regular monitoring of blood phosphorus levels is necessary.
3. Risk prevention and control measures
Perform an allergy test before first use (subcutaneous injectin of 0.1ml diluent). Control the single dose not to exceed 120% of the recommended upper limit. Avoid repeated injectins in the same area. Stop medication immediately and record any adverse reactions.
Storage and Quality Control
1. Storage condition requirements
The ideal storage temperature is 2-8 ℃, and it should be stored away from light. After opening, butafosfan injection must be used up within 24 hours. During transportation, maintain a temperature of 5-25 ℃ and avoid severe vibrations.
2. Quality inspection indicators
The appearance should be a colorless and transparent liquid with a pH value of 6.5~7.5 and an osmotic pressure of 280~320mOsm/kg. The content determined by high-performance liquid chromatography should be between 95.0% and 105.0% of the labeled amount. Bacterial endotoxin limit<0.5EU/ml.
3. Validity period management
The unopened preparation has a shelf life of 36 months, which is shortened to 7 days after opening. Regularly inspect inventory drugs and follow the principle of "first in, first out". Near expiry drugs (with a remaining shelf life of less than 6 months) should be stored separately and given priority for use.
Market and Regulatory Information
1. Registration status both domestically and internationally
Approval number from the Chinese Ministry of Agriculture: Veterinary Medicine (202X) XXXXXX. EU EMA registration number: EMEA/V/C/00XXXX. The US FDA has not yet approved it, but it allows its use within the framework of investigational new drugs (IND).
2. Price System Analysis
The price of raw materials is about 1000~1500 yuan/kg, and the price of formulations varies depending on the specifications: 100ml packaging costs about 80~120 yuan/bottle, and 500ml packaging costs about 350~500 yuan/bottle. Price fluctuations are influenced by factors such as raw material costs, production processes, and market demand.
3. Future Development Trends
The main development directions are the research and development of new long-acting formulations (extending the action time to 72 hours), the development of targeted delivery systems (improving tissue specificity), and the optimization of combination therapy regimens (combined with probiotics and herbal preparations). It is expected that the market size will grow at an annual growth rate of 8% to 10% in the next 5 years.
Hot Tags: butafosfan injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale