Marbofloxacin injection is a broad-spectrum antibiotic used in animals, belonging to the fluoroquinolon class of antibiotics. Its main component is mapofloxacin, which inhibits the activity of bacteril DNA gyrase, thereby preventing the replication, transcription, and protein synthesis of bacteril DNA, achieving bactericidal effect. The drug is widely used in veterinary clinic, mainly for the treatment of a variety of animal infectious diseses caused by sensitive bacteri.

(1) Storage conditions
Mabofloxaci injection should be stored in a light shielded and sealed manner to ensure the stability and effectiveness of the drug. The temperature should be controlled below 25 ℃. During storage, drugs should be protected from moisture, heat, or direct light to ensure their effectiveness and stability. The shelf life of drugs varies under different storage conditions, and they should be stored strictly according to the prescribed conditions to ensure drug quality.
(2) Packaging information
Mabofloxaci injection usually comes in different packaging specifications, such as 50ml: 5g, 100ml: 10g, 250ml: 25g, etc., calculated according to C17H19FN4O4. The packaging should indicate the name, specifications, production batch number, expiration date, and other information of the veterinary drug for easy identification and management.
Our product




Additional information of chemical compound:

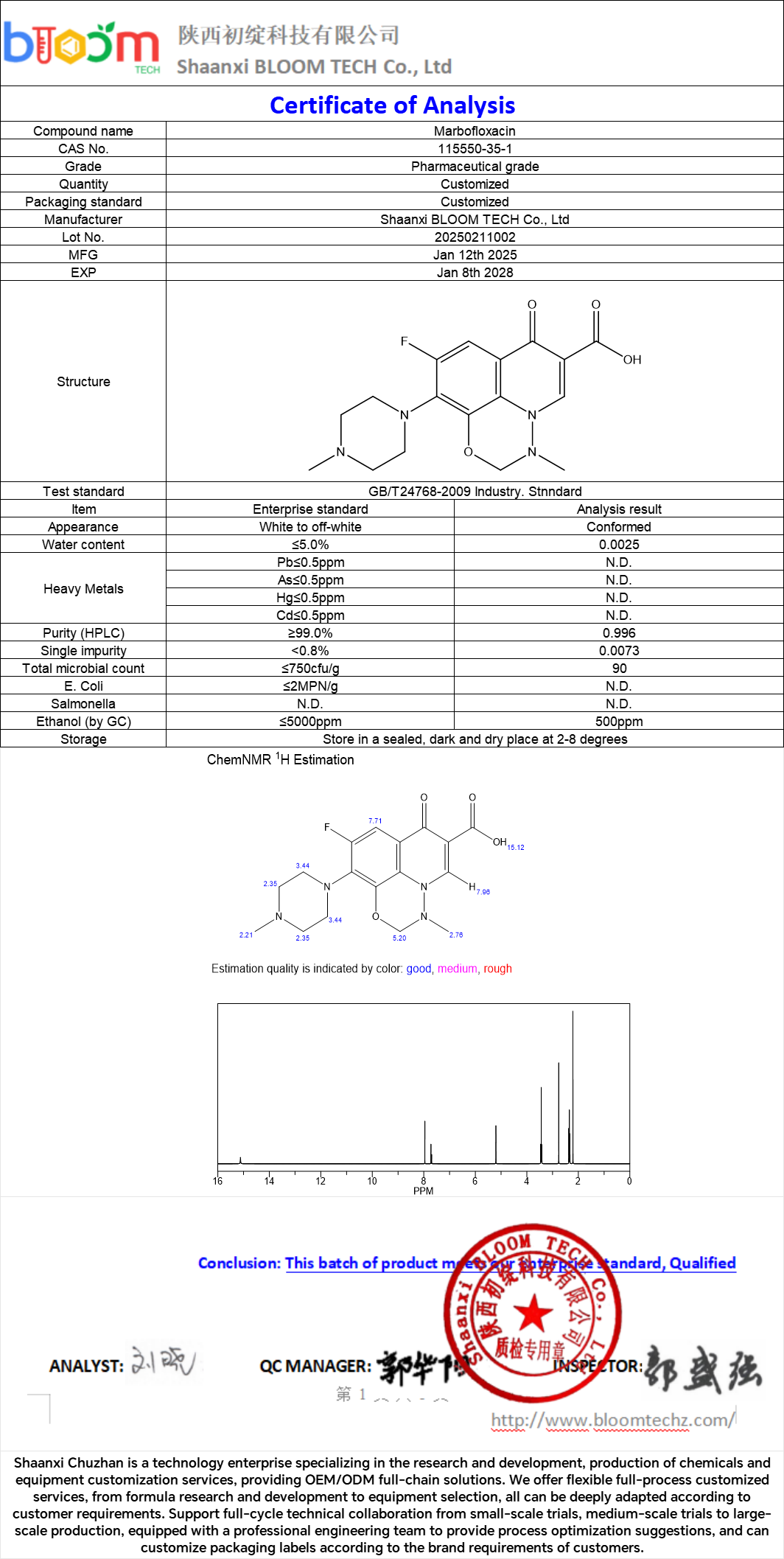
Our Product




Marbofloxacin+. COA


Indications, usage, and dosage
(1) Indications
Marbofloxacin injection is mainly used to treat a variety of animal infectious dieases caused by sensitiv bacteria, including but not limited to:
Sow: Uterine inflammation mastitis galactorrhea syndrome.
Cattle: respiratory contagions and lactation mastitis.
Other animals: Respiratory, digestive, urogenital, and skin infetions caused by bacteria sensitiv to mapofloxacin, such as deep and superficial skin contagionns and urethral infetions in dogs, skin and soft tissu infetions in cats, acute upper respiratory tract contagions, etc. It is also effective in treating bovine and sheep mastitis and porcine mastitis uterine inflammation amenorrhea syndrome.
(2) Usage and dosage
Intramuscular injection of 2mg per 1kg body weight (i.e. 1ml per 50kg body weight) once a day for 3 consecutive days in sows with uterine inflammation mastitis amenorrhea syndrome.
Cow respiratory tract contagion: intramuscular injection, once dose, 8mg per 1kg body weight (i.e. 2ml per 25kg body weight), single injection. The same part should not exceed 20ml for use.
Mastitis during lactation in cows: Subcutaneous injection, one dose, 2mg per 1kg body weight (i.e. 1ml per 50kg body weight), once a day, for 3 consecutive days.


Clinical application and case sharing
(1) Clinical application
Marbofloxaci has a wide range of clinical applications in veterinary medicine. It can not only be used to treat various infectious dieases mentioned above, but also can be used for other contagions caused by sensitiv bacteri according to specific conditions. Its broad-spectrm antibacteril activity and good pharmacokinetic properties make Mabofloxaci injection one of the important antibiotics in veterinary clinical practice.
(2) Case sharing
The following is a successful case of using Marbofloxacin Injection to treat bovine respiratory contagions:
Background of the case: A cow in a certain breeding farm experienced symptoms such as cough, runny nose, and difficulty breathing. After veterinary diagnosis, it was diagnosed as a respiratory contagion, suspected to be caused by sensitiv bacteri.
Treatment plan: Administer Marbofloxaci therapy, intramuscular injection, one dose, 8mg per 1kg body weight (i.e. 2ml per 25kg body weight), single injection.
Therapeutic effect: After treatmet, the symptoms of the cow gradually improved, including coughing, runny nose, and difficulty breathing. A few days later, the cow's breathing returned to normal, its mental state was good, and its appetite gradually recovered. After re examination, the respiratory contagion of the cattle has been effectively controlled.


Pharmacological effects and antibacterial spectrum
(1) Pharmacology
Mabofloxaci mainly exerts strong antibacteral effects by inhibiting the activity of bacteral DNA gyrase A subunit, interfering with the replication, transcription, and protein synthesis processes of bacteral DNA. This mechanism of action enables mapofloxacin to have a rapid bactericidal effect on various bacteri, and sensitiv bacteria can cause death within 20-30 minutes after exposure.
(2) Antibacterial spectrum
Mabofloxaci has broad-spectum antibacteral activity and is effective against various Gram positive and Gram negative bacteri.
Gram positive bacteria: especially have strong antibacteral activity against Staphylococcus, with MIC values of 0.2 μ g/ml and 0.23 μ g/ml for Staphylococcus aureus and Staphylococcus aureus, respectively, which are superior to ofloxacin and comparable to ciprofloxacin and enrofloxacin. In addition, it also has a certain antibacteral effect on streptococcus, but the MIC ₅₀ value is relatively high, at 3.8 μ g/ml.
Gram negative bacteria: The MIC of most Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis and other Escherichia coli is 0.08-0.28 μ g/ml; The MIC for Pasteurella multocida is 0.04 μ g/ml; The MIC for Pseudomonas aeruginosa is 0.94 μ g/ml. At the same time, some pathogenic bacteri in cattle, such as hemolytic Pasteurella, Pasteurella multocida, and Haemophilus influenzae, also have high activity.
In addition, some pathogenic bacteri such as Staphylococcus aureus, which have developed partial resistance to erythromycin, lincomycin, chloramphenicol, doxycycline, and sulfonamide drugs, are still sensitiv to ciprofloxacin. Therefore, this drug can be used as the first choice for treating canine pyoderma caused by this bacterium.

Pharmacokinetic properties
Animals injected with marbofloxacin Injection have rapid and complete absorption. For example, after intramuscular injection of 2.0mg per kilogram of body weight in pigs, the peak concentration of the drug can be reached within 2 hours, with a bioavailability of 80.2%; A single intramuscular injection of 8mg/kg of mapofloxacin in cattle can reach its peak blood concentration in about 1 hour, with a Cmax of approximately 8 μ g/ml; After subcutaneous injection of 2mg/kg mapofloxacin, the peak blood concentration can be reached within about 1 hour, with a Cmax of about 1.7 μ g/ml. Mabofloxaci is widely distributed in the body with a large apparent distribution volume, such as 1.75L · kg ⁻¹ in pigs. It is widely distributed in tissus such as the liver, kidneys, skin, lungs, and bladder, and the drug concentration in most tissus is higher than that in blood. Except for the central nervous system, the drug concentration in all tested tissus is higher than that in plasma.
Part of the marboxoxacin is metabolized and converted into inactive metabolites (N-demethylated MBF and N-oxo MBF) in the liver. Its main excretion pathway is through the kidneys, with a certain proportion excreted in urine, such as dogs excreting 30% -45% of the prototype drug in urine. The elimination half-life of mapofloxacin varies among different animals and administration methods. The half-life of intramuscular injection of this product in pigs is 20.09 hours; The elimination half-life of intramuscular injection in cattle is 9.5 hours, and the elimination half-life of subcutaneous injection is 5.6 hours; The elimination half-life of oral and subcutaneous injection in dogs can reach 14 hours and 13 hours, while that of calves (intramuscular injection), broiler chickens (intramuscular injection), sows (oral administration), and cats (oral administration) are 4.33, 5.26, 5.74, and 10 hours, respectively. Mabofloxaci is mainly excreted in its original form through urine and feces, and this longer half-life period allows the effective blood concentration to be maintained for a longer time after administration, making it suitable for once a day administration.
Lactation and Pregnancy: After oral administration of Mabofloxaci to sows, the t ₁/₂ β (10.09h) of pregnant pigs is significantly longer than that of lactating pigs (5.74h), and the drug clearance rate in lactating pigs is also higher (3.27ml/(min · kg · bw)). Therefore, when using Mabofloxaci for treatmet during lactation, the dosag should be increased.
Diease status: Thomas (1994) conducted a pharmacokinetic study on the intramuscular injection of mapofloxacin in healthy and hemolytic Pasteurella infected ruminant calves, and found that mapofloxacin had high bioavailability in both, but the absorption and elimination rates of the drug in dieased cows were lower than those in healthy cows.
Renal dysfunction: In the state of canine kidney injury, the pharmacokinetic parameters of mapofloxacin do not change significantly, indicating that mild renal dysfunction in clinical application does not require adjustment of the dosing regimen of mapofloxacin. However, when using fluoroquinolons in human medicine, if renal dysfunction occurs, the dosing interval should be extended or the dosag should be reduced to avoid drug accumulation and side effects.

Rest period and residue limit

Rest period for medication
The rest period of barbofloxacin Injection varies among different animals. The rest period for pigs is 4 days; The withdrawal period for intramuscular injection in cattle is 3 days, and the weaning period is 72 hours; The medication withdrawal period for subcutaneous injection is 6 days, and the milk withdrawal period is 36 hours. During the rest period, it is prohibited to slaughter animals or consume their related products (such as milk) to ensure that drug residues in animal based foods meet safety standards.
Residue limit
Referring to the European Union's EMA standards for the daily allowable intake (ADI) and maximum residue limit (MRL) of marboxloxacin in bovine adipose tissu, the reporting unit has established a maximum residue limit (MRL) of 50 μ g/kg for marboxloxacin in bovine fat. At the same time, there are relevant standards for the detection of residual mapofloxacin in bovine adipose tissu (trial), which use liquid chromatography tandem mass spectrometry (LC-MS) for qualitative and quantitative determination of mapofloxacin residues in bovine adipose tissu to ensure the safety of animal food.

FAQ
1. What unique effects does Marbofloxacin have on bacterial resistance?
Its broad-spectrum bactericidal activity belongs to the third-generation fluoroquinolone drugs. It mainly targets the bacterial DNA gyrase. It may still be effective for some bacterial strains that have developed resistance to early quinolones. However, due to the similar mechanism of action, long-term and widespread use may also exacerbate cross-resistance.
2. What are the key differences in the pharmacokinetics of Marbofloxacin in different animal species?
Its distribution volume is large and its metabolic rate in the animal body is low. It is mostly excreted by the kidneys in its original form. The key difference lies in the elimination half-life: dogs have approximately 12-14 hours, cats about 8-10 hours, while cows may be as short as 4-6 hours. This directly determines the species-specificity of the dosing interval.
3. What rare but serious risk should be particularly noted in the clinical application of this injection?
Apart from common allergic reactions or gastrointestinal issues, it is necessary to be vigilant about possible joint cartilage damage (arthritis) in young animals or those with rapid growth after using the drug. This is especially true when using high doses or over an extended period, which is a typical toxicity of fluoroquinolone drugs.
4. Has the issue of environmental residue been given sufficient attention?
As a persistent antibacterial drug, when it enters the environment through animal excrement, it may disrupt the soil microbial community and promote the spread of drug-resistant genes in the environment. Therefore, in intensive farming, it is necessary to strictly follow the withdrawal period and manage the waste properly.
Hot Tags: marbofloxacin injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale








