Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of vip injection in China. Welcome to wholesale bulk high quality vip injection for sale here from our factory. Good service and reasonable price are available.
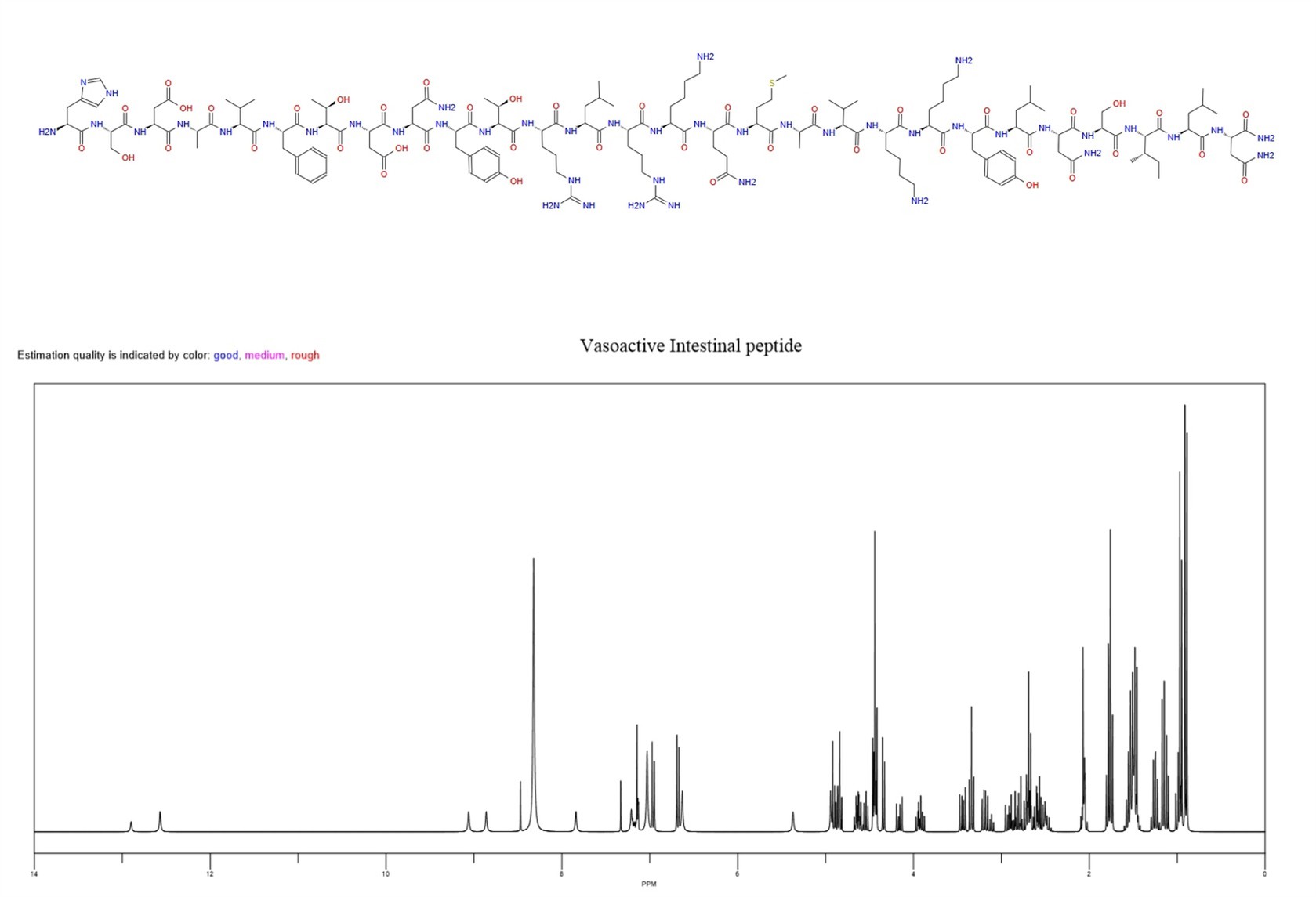
Vasoactive intestinal peptide / Vip Injection, as a multifunctional neuropeptide composed of 28 amino acids, is widely distributed in the nervous, digestive, and respiratory systems of the body. Its biological effects have high scene specificity and can exhibit significant abnormal expression or functional regulation in specific pathological states, thereby participating in the occurrence, development, and pathological process of diseases.
Product Forms






VIP COA
 |
||
| Certificate of Analysis | ||
| Compound name | Vasoactive Intestinal Peptide | |
| Grade | Pharmaceutical grade | |
| CAS No. | 37221-79-7 | |
| Quantity | 33g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202501090088 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
|
|
||
| Chemical Formula | C147H238N44O42S | |
| Exact Mass | 3323.76 | |
| Molecular Weight | 3325.85 | |
| m/z | 3324.76 (100.0%), 3323.76 (62.9%), 3325.76 (52.1%), 3326.77 (41.3%), 3325.76 (26.9%), 3325.76 (15.1%), 3324.75 (9.5%), 3327.77 (9.4%), 3326.76 (8.6%), 3326.76 (7.9%), 3327.77 (6.7%), 3325.76 (5.4%), 3327.76 (5.4%), 3326.76 (4.7%), 3326.76 (4.5%), 3327.77 (4.5%), 3328.77 (3.6%), 3328.77 (3.1%), 3325.75 (2.8%), 3325.77 (2.7%), 3327.76 (2.4%), 3327.77 (2.3%), 3326.77 (2.2%), 3328.77 (1.9%), 3324.76 (1.7%), 3325.76 (1.6%), 3328.76 (1.5%), 3328.77 (1.5%), 3327.76 (1.3%), 3327.76 (1.3%), 3327.76 (1.2%), 3328.77 (1.1%), 3326.75 (1.1%), 3325.76 (1.1%), 3324.76 (1.0%) | |
| Elemental Analysis | C, 53.09; H, 7.21; N, 18.53; O, 20.20; S, 0.96 | |
Among them, the related syndromes caused by VIP tumors and the potential application of vip Injection in cardiovascular diseases such as pulmonary hypertension, asthma, and chronic obstructive pulmonary disease (COPD) are the two core directions of current clinical diagnosis and research exploration. The former focuses on the pathological damage and clinical diagnostic and therapeutic value caused by abnormal secretion of it, while the latter focuses on the exploration and application of VIP's biological efficacy in disease treatment. Both comprehensively demonstrate the important biological significance and application value of this in clinical diagnosis and treatment from two dimensions: pathological characterization and therapeutic potential. The following will provide a detailed analysis from multiple dimensions based on specific pathological mechanisms, clinical characteristics, and research and development directions.

Pathological characteristics and clinical value of VIP tumor associated WDHA syndrome
It tumors are neuroendocrine lesions that over-secrete VIP, causing WDHA syndrome. it levels are key for diagnosis and monitoring, and can be analyzed from three aspects:
01.Pathological triggering mechanism
VIP tumors often exist sporadically, with a few being associated with multiple endocrine neoplasia type 1. Tumor cells can abnormally synthesize and release large amounts of vip Injection, which can bind to the VPAC1 and VPAC2 receptors on the surface of intestinal epithelial cells, activate adenylate cyclase, and significantly increase the concentration of intracellular cyclic adenosine monophosphate (cAMP), thereby disrupting the secretion function of intestinal mucosa, breaking the balance between water and electrolyte absorption and secretion, and ultimately inducing a series of pathological symptoms. Some cases may also be accompanied by hypercalcemia, and its mechanism may be related to the coordinated secretion of parathyroid hormone related peptides.
02.Specific characterization of core symptoms
Watery diarrhea, as the most typical initial sign of WDHA syndrome, often presents as sudden and persistent attacks, with a daily defecation volume of several liters and no mucus or pus in the stool. Conventional antidiarrheal interventions are ineffective, which is directly related to it mediated intestinal secretion hyperactivity.

Hypokalemia is a secondary disorder caused by diarrhea, where a large amount of water and electrolytes are lost with feces. If not intervened in a timely manner, it can further induce serious complications such as muscle weakness and arrhythmia; Gastric acid deficiency is caused by the inhibitory effect of this on the secretion function of gastric wall cells, leading to a significant decrease in gastric acid secretion. Some patients may experience secondary symptoms such as indigestion and abdominal distension.
03.The core value of clinical diagnosis and monitoring
The detection of it levels is a key basis for the diagnosis of WDHA syndrome. Elevated fasting plasma VIP levels can serve as core diagnostic evidence. Combined with imaging examinations such as abdominal CT, MRI, and somatostatin receptor scintigraphy, lesion localization diagnosis can be achieved, especially for the detection of metastatic it tumors, which is of great significance; At the same time, dynamic monitoring of VIP level changes can intuitively reflect the progression of the disease, treatment effectiveness, and recurrence, providing reliable references for adjusting treatment plans and being an important indicator for the full process management of the disease.
The data source of the above section is:
Prospect of Vasoactive Intestinal Peptide Therapy for COPD/PAH and Asthma: A Review. PMC, PMID: 21566164.
Sami I. Said. Vasoactive Intestinal Peptide. Sci-Hub, 2006.
Effect of Vasoactive Intestinal Polypeptide on the Canine Cardiovascular System. PubMed, PMID: 4056568.
The biological potential and research direction of VIP in cardiovascular and pulmonary diseases
Vip Injection shows notable anti-inflammatory, vasodilatory and bronchoprotective effects in pulmonary arterial hypertension, asthma, COPD and other cardiovascular diseases, offering new therapeutic strategies. Developing long‑acting analogs and new delivery systems is key to overcoming clinical limitations, as detailed below:
01.The efficacy in pulmonary arterial hypertension
The core pathological features of pulmonary arterial hypertension are pulmonary vascular proliferation and remodeling, vascular spasm, and peripheral inflammatory infiltration, ultimately leading to increased pulmonary circulation resistance and right heart failure. VIP can relax pulmonary artery smooth muscle cells, reduce pulmonary vascular tension, and improve pulmonary hemodynamic status; At the same time, its anti-inflammatory effect can inhibit the release of pro-inflammatory factors, reduce inflammation infiltration around pulmonary blood vessels, and alleviate the process of vascular remodeling. In addition, it can also increase coronary blood flow, improve myocardial oxygen supply, and play a certain protective role in cardiac function.
02.The protective effect in asthma and COPD
Asthma and COPD are both characterized by airway inflammation, airway spasm, and airway remodeling as their core pathological features. it can regulate the recruitment and activation of inflammatory cells, inhibit the release of pro-inflammatory mediators, reduce inflammation damage to airway mucosa, and maintain airway immune homeostasis; At the same time, it can relax the smooth muscles of the airway, dilate the airway, relieve airway spasms, improve ventilation function, and reduce the occurrence of symptoms such as wheezing and coughing; In addition, it can protect the integrity of airway mucosa, reduce mucosal damage and abnormal secretion of secretions, and delay disease progression.

03.Breakthrough point in core research and development direction
Although vasoactive intestinal peptide has shown great therapeutic potential in cardiovascular diseases, the natural it has a very short half-life in the body, is easily inactivated by enzymatic hydrolysis, and has limited administration methods, which limits its clinical application. At present, the research and development of long-acting VIP analogues has become a focus, extending their in vivo retention time.
And enhancing their bioavailability through structural modification; At the same time, the development of new delivery systems, such as targeted delivery mediated by nanomicelles and liposomes, can achieve precise targeted administration of it, reduce systemic adverse reactions, and enhance local therapeutic effects. The synergistic development of the two is expected to promote the clinical translation of VIP in the treatment of cardiovascular diseases.
The data source of the above section is:
Una Cidon E. Vasoactive Intestinal Peptide Secreting Tumour: An Overview. World J Gastroenterol Oncol, 2022, 14(4): 808-819.
Sandhu S, Jia Lal I. VIPoma. StatPearls, StatPearls Publishing, 2022.
References
Azizian A, Kö nig A, Ghadimi M. Treatment Options of Metastatic and Nonmetastatic VIPoma: A Review. Langen Becks Arch Surg, 2022, 407(10): 2629-2636.
Lakhtakia et al. Endoscopic Ultrasound-Guided Radiofrequency Ablation of Metastatic Pancreatic VIPoma: A Novel Treatment. Cureus, 2024, 16(8): e67114.
Novel, Biocompatible, Disease Modifying Nanomedicine of VIP for Rheumatoid Arthritis. PMC, PMID: 23382866.
FAQ
- What is the function of the vasoactive intestinal peptide?
Vasoactive intestinal peptide (VIP) is a 28-amino acid neuropeptide and hormone that functions primarily as a vasodilator, smooth muscle relaxant, and secretagogue in the digestive system. It regulates gut motility, stimulates water and electrolyte secretion, inhibits gastric acid secretion, and serves as a neurotransmitter and immune modulator,.
- Are there any known side effects of VIP peptide?
In addition, systemic administration of VIP causes cardiovascular and gastrointestinal side effects including a marked decrease in blood pressure, tachycardia, cutaneous flushing, and watery-diarrhea syndrome (Bloom et al., 1973, Henning and Sawmiller, 2001, Morice et al., 1983).
- What does VIP do in the intestines?
VIP stimulates the secretion of ductal pancreatic and biliary bicarbonate and water and simultaneously inhibits gastric acid and pepsinogen secretion as well as absorption from the intestinal lumen. VIP also stimulates enzyme secretion from pancreatic acinar cells and regulates chloride secretion.
Hot Tags: vip injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale