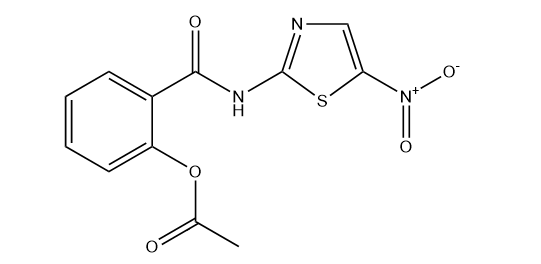
Nitazoxanide tablets, The chemical name is 2- [(5-nitro-1,3-thiazol-2-yl) carbamoyl] phenol acetate, which belongs to the derivatives of nitrothiazide amide. Its molecular formula is C12H9N3O5S, with a molecular weight of 307.28 g/mol. From a chemical structure perspective, it contains nitro groups, thiazole rings, benzamide groups, and acetate side chains. This complex molecular structure endows it with unique pharmacological activity. The nitro and thiazole rings in the molecule give it electron acceptor properties, while the benzamide group provides hydrogen bond donor ability. These structural features together determine its interaction mode with the target, thereby affecting the efficacy and mechanism of action of the drug.
Appearance and dosage form
Nitazoxanide raw materials usually appear as white to off white crystalline powders, which are characterized by the visible light absorption properties of the conjugated system in their molecular structure. During the tablet preparation process, the appearance of the final product can be adjusted by adding appropriate excipients. For example, adding light blocking agents such as titanium dioxide can make the tablets appear pure white, while adding FD&C dyes can prepare colored tablets. Its powder form is usually regular crystals, which helps to improve the bulk density and flowability of drugs, thereby optimizing the formulation process. The main dosage forms on the market are tablets (500mg) and dry suspensions (60ml, 100mg/5ml), which are suitable for patients of different ages. Tablets are easy for adults and older children to take, while dry suspensions are more suitable for patients with swallowing difficulties such as infants and young children.
Our product




Additional information of chemical compound:
| Product Name | Nitazoxanide Tablets | Nitazoxanide Powder |
| Product Type | Tablets | Powder |
| Product Purity | HPLC≥99.0% | ≥99% |
| Product Specifications | Customizable | Customizable |
| Product Package | Customizable | Customizable |
Our Product



Nitazoxanide+. COA
 |
||
Certificate of Analysis |
||
|
Compound name |
Nitazoxanide | |
|
CAS No. |
55981-09-4 | |
|
Grade |
Pharmaceutical grade | |
|
Quantity |
Customized | |
|
Packaging standard |
Customized | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
|
Lot No. |
20250109001 |
|
|
MFG |
Jan 12th 2025 |
|
|
EXP |
Jan 8th 2029 |
|
|
Structure |
|
|
| TEST STANDARD | GB/T24768-2009 Industry. Stnndard | |
|
Item |
Enterprise standard |
Analysis result |
|
Appearance |
White or almost white powder |
Conformed |
|
Water content |
≤4.5% |
0.30% |
| Loss on drying |
≤1.0% |
0.15% |
|
Heavy Metals |
Pb≤0.5ppm |
N.D. |
|
As≤0.5ppm |
N.D. | |
|
Hg≤0.5ppm |
N.D. | |
|
Cd≤0.5ppm |
N.D. | |
|
Purity (HPLC) |
≥99.0% |
99.5% |
|
Single impurity |
<0.8% |
0.48% |
|
Residue on ignition |
<0.20% |
0.064% |
|
Total microbial count |
≤750cfu/g |
80 |
|
E. Coli |
≤2MPN/g |
N.D. |
|
Salmonella |
N.D. | N.D. |
|
Ethanol (by GC) |
≤5000ppm |
400ppm |
|
Storage |
Store in a sealed, dark and dry place at-20 degrees |
|

|
||

Preparation process and quality control
In the preparation process of Nitazoxanide Tablets, the active pharmaceutical ingredient needs to be mixed with the excipients by an equal amount of addition method to ensure a content uniformity RSD of less than 2.0%. In the wet granulation process, a 5% ethanol solution of polyvinylpyrrolidone K30 is commonly used as a binder to produce particles with a bulk density of 0.55-0.65 g/cm ³. The compression parameters are usually set to a pressure of 15-20 kN and a rotational speed of 20-30 rpm, which can produce tablets with a hardness of 8-12 kp and a brittleness of less than 0.5%. The coating process uses gastric soluble film coating premix such as Obadai, which increases weight by 3-5% and can effectively improve appearance and control release. A reasonable formulation process can ensure the quality and stability of tablets, improve the efficacy and safety of drugs.
Quality control is a crucial step in ensuring the quality of Nitazoxanide. In the quality control of raw materials and formulations, strict testing is required for indicators such as drug content, purity, related substances, dissolution rate, etc. The content determination adopts methods such as high-performance liquid chromatography to ensure that the drug content meets the prescribed standards. Purity testing requires that the impurity content in the drug be within the allowable range, and the control of relevant substances is crucial to ensure the safety and effectiveness of the drug. The dissolution test evaluates the drug's dissolution performance under specified conditions, ensuring that the drug can be effectively released and absorbed in the body. Through strict quality control, the stable and reliable quality of Nitazoxanide can be ensured, providing assurance for the treatment of patients.
The contact angle measurement of the active pharmaceutical ingredient shows a contact angle of 98 ° in water, indicating its strong hydrophobicity. By adding 2% sodium dodecyl sulfate, the contact angle can be reduced to 65 °, significantly improving wettability. The regulation of surface properties is crucial for improving the dissolution of poorly soluble drugs. In the tablet disintegration time limit test, the optimized prescription can completely disintegrate within 15 minutes, meeting the requirements of the pharmacopoeia.

Indications
Nitazoxanide are mainly used to treat diarrhea caused by Cryptosporidium and Giardia, and can also be used to treat other intestinal parasitic infections, such as Bacillus subtilis and other sporozoites, amoebids, human roundworms, hookworms, flagellates, beef tapeworms, short membrane shell tapeworms, and liver flukes. In addition, it also has activity against certain anaerobic and microaerophilic bacteria, such as Helicobacter pylori. In terms of antiviral effects, it also has a certain therapeutic effect on rotavirus gastroenteritis and chronic hepatitis C. Its wide range of indications makes Nitazoxanide play an important role in clinical treatment, providing effective drug options for the treatment of various diseases.


Usage and dosage
The usage and dosage for adults and children are different. Generally speaking, the commonly used dosage for adults is 500 mg, twice a day. The dosage for children should be adjusted according to age and weight, and the specific usage and dosage should follow the doctor's advice. The correct usage and dosage are key to ensuring the efficacy and safety of drugs. Patients should strictly follow the doctor's instructions to take the medication and avoid increasing or decreasing the dosage or changing the medication method on their own.
Adverse reactions
Using Nitazoxanide Tablets may result in some adverse reactions, such as rash, nausea, vomiting, chest discomfort, tachycardia, chills, fever, headache, and other symptoms. These adverse reactions are usually mild and gradually disappear after discontinuation of the medication. However, if the adverse reactions are severe or long-lasting, patients should seek medical attention promptly so that doctors can take appropriate treatment measures. Understanding the adverse reactions of drugs can help patients detect and address potential issues in a timely manner during drug use, ensuring medication safety.


Storage conditions and stability
Storage conditions
Nitazoxanide need to be stored under refrigeration conditions of 2-8 ° C, which is mainly due to their sensitivity to light and humidity. During storage, drugs should be avoided from being exposed to strong light, while maintaining the sealing of the packaging to prevent moisture from affecting the drugs. Proper storage conditions can ensure the quality and stability of drugs, and extend their shelf life.
Stability
In the accelerated stability test, after being placed at 40 ° C/75% RH for 6 months, the content of relevant substances increased by 1.2% and decreased by 3.8%. The light test showed that under 4500 lx illumination for 10 days, the drug degradation rate reached 5.6%. The degradation products mainly include deacetylnitroaniline and nitro reduction products, which need to be controlled within the limit of 0.5% total impurities and 0.2% individual impurities by HPLC method. Understanding the stability characteristics of drugs can help develop reasonable storage and use plans, avoiding drug failure or adverse reactions due to improper storage.
Pharmacological effects and mechanisms
Antibacterial and antigen worm effects
Nitazoxanide is a broad-spectrum antibacterial and antiparasitic drug. In vitro studies have shown that it can treat infections caused by worms and protozoa such as dysentery amoeba, Giardia, Trichomonas vaginalis, as well as bacteria such as Campylobacter jejuni, Clostridium difficile, Clostridium perfringens, and Helicobacter pylori. Although its mechanism of action is not fully understood, it is believed to be related to the enzyme dependent electron transfer reaction that inhibits pyruvate and iron redox protein oxidoreductase, the latter of which is crucial for anaerobic energy metabolism. By interfering with the energy metabolism processes of parasites and bacteria, Nitazoxanide can inhibit their growth and reproduction, thereby achieving the goal of treating infections.
Antiviral effect
In recent years, studies have found that Nitazoxanide also has inhibitory effects on certain viruses. For example, it is effective against rotavirus gastroenteritis and chronic hepatitis C. In vitro experiments have confirmed that the metabolite of Nitazoxanide, tizoxanide, has an inhibitory effect on HCV replication. A multicenter double-blind randomized controlled trial investigated the efficacy of Nitazoxanide in the treatment of chronic hepatitis C, with a treatment regimen of Nitazoxanide 500 mg po bid or placebo for 24 weeks. As a result, at the end of the treatment, the HCV RNA seroconversion rates in the Nitazoxanide group and placebo group were 30.4% (7/23) and 0% (0/24), respectively; After 24 weeks of follow-up after the course of treatment, the continuous negative conversion rate of HCV RNA in the Nitazoxanide group was 17.4% (4/23). This indicates that Nitazoxanide has certain potential in antiviral therapy, providing a new option for the treatment of related diseases.
Other functions
Nitazoxanide tablets can also regulate autophagy and inhibit mTORC1 signaling, demonstrating effective inhibition of HBV replication. It induces a decrease in the production of several HBV proteins (HBsAg and HBeAg, core antigens) in 2.2.15 cells, but does not affect the transcription of HBV RNA. These mechanisms of action further expand the application scope of Nitazoxanide, making it possible to play an important role in the treatment of various diseases. However, the current research on these mechanisms of action is not deep enough, and further studies are needed to reveal their specific modes of action and potential clinical application value.
Clinical Applications
:max_bytes(150000):strip_icc()/GettyImages-88689474-5942c30a3df78c537b1588e8.jpg?size=x0)
Treatment of Protozoal Infections
Nitazoxanide tablets are most commonly used for the treatment of protozoal infections, particularly those caused by Giardia lamblia and Cryptosporidium parvum. Giardiasis, caused by Giardia lamblia, is a common intestinal infection characterized by diarrhea, abdominal cramps, and bloating. Nitazoxanide has been shown to be highly effective in treating giardiasis, with cure rates exceeding 90% in clinical trials.
Cryptosporidiosis, caused by Cryptosporidium parvum, is another intestinal infection that can be severe, particularly in immunocompromised individuals such as those with HIV/AIDS. Nitazoxanide has been approved by the U.S.
Treatment of Helminthic Infections
In addition to its antiprotozoal activity, nitazoxanide tablets have also demonstrated efficacy against certain helminthic infections, such as fascioliasis and taeniasis. Fascioliasis, caused by the liver flukes Fasciola hepatica and Fasciola gigantica, is a zoonotic infection that can cause liver damage and other complications. Nitazoxanide has been shown to be effective in treating fascioliasis, with cure rates comparable to those of traditional anthelmintic agents.
Taeniasis, caused by tapeworms of the genus Taenia, is another helminthic infection that can be treated with nitazoxanide. Although less commonly used than other anthelmintic agents such as praziquantel, nitazoxanide has shown promise in treating taeniasis, particularly in cases where other treatments are contraindicated or ineffective.

Frequently Asked Questions
How fast does nitazoxanide work?
+
-
Nitazoxanide should start working against the diarrhea-causing organisms soon after your first dose. However, you might not feel the effects of the medication right away. In clinical studies, researchers checked in with people who took nitazoxanide (Alinia) 7 to 10 days after the start of their treatment.
Who should not take nitazoxanide?
+
-
Kidney disease. Liver disease. An unusual or allergic reaction to nitazoxanide, other medications, foods, dyes, or preservatives. Pregnant or trying to get pregnant.
Does nitazoxanide cause hair loss?
+
-
Nitazoxanide may cause side effects like nausea, vomiting, stomach pain, fever, hair loss, dizziness, giddiness, and headache.
What are the side effects of nitazoxanide?
+
-
Overview: Nitazoxanide is used to treat diarrhea caused by certain parasites. It kills them by interfering with their ability to grow and use energy. Common side effects include stomach pain, headache, change in urine color, and nausea.
Hot Tags: nitazoxanide tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CAS 58164 04 8, CAS 1300115 09 6, raloxifene powder, 1H Benzimidazole 1 4 bromophenyl 2 ethyl , CAS 26885 42 7, 2 1 1 biphenyl 4 yl 4 6 dichloropyrimidine