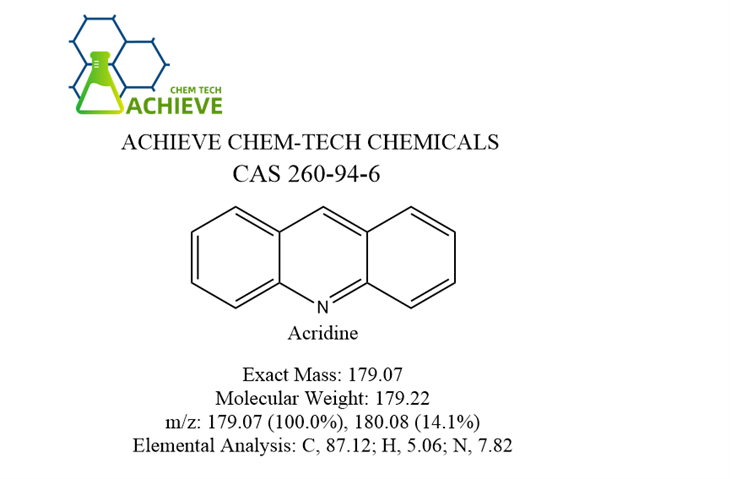
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of acridine cas 260-94-6 in China. Welcome to wholesale bulk high quality acridine cas 260-94-6 for sale here from our factory. Good service and reasonable price are available.
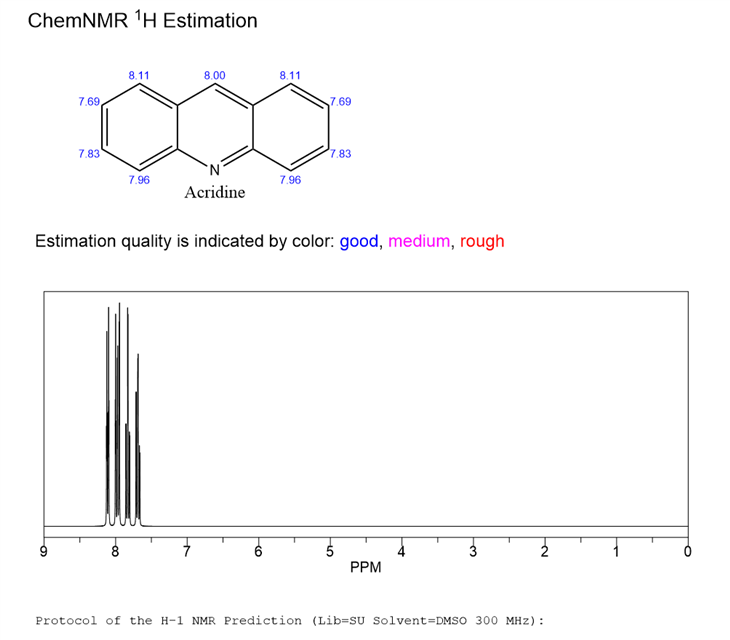
Acridine is an organic compound belonging to the class of aromatic amines. Acridine has the chemical formula C13H9N and the molecular weight is 179.22. It consists of a central pyridine ring and two benzene rings, forming a triangular structure. This structure gives Acridine unique physical and chemical properties.Acridine is usually a yellow to yellow-brown crystalline powder with a melting point of about 106~109°C and a boiling point of 346°C. It has good solubility in organic solvents such as dioxane.Acridine has a certain degree of alkalinity, and it can react with acids to form salts. It can also react chemically with certain reagents by substitution, addition and oxidation.
Acridine itself may not be used directly as a product, but it may be used as a raw material or intermediate in certain specific areas or products. In the laboratory, Acridine may be used in a variety of chemical studies and experiments, including organic synthesis, photophysical and photochemical studies.
|
|
|

|
|
|
|
Chemical Formula |
C13H9N |
|
Molecular Weight |
179.22 |
|
Exact Mass |
179.07 |
|
m/z |
179.07 (100.0%), 180.08 (14.1%) |
|
Elemental Analysis |
C, 87.12; H, 5.06; N, 7.82 |
|
Melting point |
106-109 °C |
|
Boiling point |
346 °C(lit.) |
|
Density |
1,005 g/cm3 |
|
Storage conditions |
Refrigerator |
|
Solubility |
dioxane: 0.1 g/mL, clear |
|
Flash Point |
346°C |
|
Index Of Refraction |
1.7270 (estimate) |
|
Acidity Coefficient (pKa) |
5.58(at 20℃) |
|
Form |
Crystalline Powder |
|
Color |
Yellow to yellow-brown |
|
Water Solubility |
57.35mg/L(24 ºC) |
|
Stability |
Stable. Combustible. Incompatible with strong oxidizing agents. |

Acridine, as an organic compound, has its applications in several fields. Below are the main application areas of Acridine:

Chemical Research
In the laboratory, Acridine is commonly used in organic synthesis reactions, where it can be used as a catalyst or intermediate for the reaction due to its unique chemical structure. In addition, Acridine is also used in photophysical and photochemical research, where it can be used as a fluorescent probe or photosensitiser for the study of light-substance interactions.
Biochemical Studies
Acridine and its derivatives have a wide range of applications in biochemistry, especially as fluorescent dyes for DNA and RNA. These dyes are able to bind to and fluoresce nucleic acids, thus facilitating researchers to visualise nucleic acids within cells.
For example, Acridine Orange (Acridine Orange) is a commonly used fluorescent dye that binds to DNA and emits green fluorescence for cell staining and fluorescence microscopy.


Environmental Monitoring
Some derivatives of Acridine can be used as indicators for environmental monitoring to detect pollutants in water or other environmental parameters.
Dye Industry
Acridine and its derivatives can also be used as precursors or intermediates of dyes for the synthesis of dyes of various colours, which are widely used in textile and printing industries.
It should be noted that since Acridine and its derivatives are toxic and carcinogenic, it is necessary to strictly follow the safety operation procedures when using them, avoid contact with skin, eyes, etc., and ensure that the workplace is well ventilated. Meanwhile, when handling waste liquid or waste containing Acridine, appropriate environmental protection measures should also be taken to prevent pollution to the environment.

Acridine is a multifaceted compound with a rich history of research and diverse applications. Its unique chemical structure and biological activities make it a valuable tool in medicine, biotechnology, and materials science. However, the environmental and safety considerations associated with acridine use highlight the need for sustainable and responsible approaches in its applications. Future research on acridine is expected to focus on the development of novel derivatives, green chemistry approaches, and multidisciplinary collaborations, paving the way for new and exciting applications of this fascinating compound.
Biological Applications
Antimicrobial and Antiparasitic Agents
Acridine derivatives have long been recognized for their antimicrobial properties:
Proflavine (3,6-Diaminoacridine): Used as a topical antiseptic in World War I, though its use declined due to mutagenicity concerns.
Acriflavine (Mixture of Proflavine and Euchoflavine): Effective against Gram-positive bacteria and some fungi, historically employed in wound disinfection and aquaculture.
Quinacrine (Mepacrine): An acridine derivative used to treat giardiasis and malaria before the advent of chloroquine.
Mechanism of Action: Acridines intercalate into DNA, disrupting replication and transcription by stabilizing DNA-protein complexes and inducing single-strand breaks.
Anticancer Activity
Several acridine derivatives exhibit potent antitumor effects:
Amsacrine (m-AMSA): A 9-aminoacridine derivative used to treat acute myeloid leukemia (AML) and non-Hodgkin lymphoma. It inhibits topoisomerase II, causing DNA double-strand breaks and apoptosis.
Doxorubicin (Adriamycin): Though not a pure acridine, its anthracycline structure (derived from acridine) is critical for its efficacy against breast, ovarian, and lung cancers.
Challenges: Cardiotoxicity and drug resistance limit long-term use, prompting research into nanotechnology-based delivery systems to enhance targeting and reduce side effects.
Fluorescent Probes and Stains
Acridine's fluorescence makes it invaluable in biological imaging:
Acridine Orange (AO): Stains nucleic acids (DNA: green; RNA: red) and is used in flow cytometry and microscopy to differentiate cell types and assess viability.
Acridine Yellow: A pH-sensitive dye for tracking lysosomal activity in live cells.
DNA Intercalators and Mutagenesis
While intercalation is therapeutic in cancer treatment, it can also induce mutations. Acridine derivatives like 2-aminofluorene are classic mutagens used in the Ames test to assess carcinogenic potential.

Laboratory research
Acridine is often used in the laboratory in the fields of organic synthesis, photophysical and photochemical research. Due to its unique chemical properties, it can be used as a catalyst for organic reactions, fluorescent probes and photosensitisers.
Pharmaceuticals
Although Acridine itself is not directly used in drug preparation, its derivatives have certain applications in the pharmaceutical field. For example, some Acridine derivatives can be used as candidates for antitumour or antibacterial drugs.
Biochemical research
Acridine can also be used in biochemical research, such as staining and labelling of DNA and RNA. Some Acridine derivatives can be used as fluorescent probes for the detection of nucleic acid molecules in cells.
Dye Industry
Acridine and its derivatives also have applications in the dye industry. They can be used as precursors or intermediates in the synthesis of dyes of various colours.
However, due to the high toxicity of Acridine, safety procedures should be strictly followed when using it, avoiding contact with skin, eyes, etc. At the same time, it should be ensured that the workplace is well ventilated to avoid inhaling its vapour for a long time. When storing, Acridine should be kept in a dry, cool and ventilated place, avoiding direct sunlight and high temperature.

Acridine is usually synthesised in several ways.
- Method 1: From Acridone
Acridone is obtained from diphenylamine-2-carboxylic acid by cycling with sulphuric acid, and then oxidising acridine by reduction to 9,10-dioxoacridine with pentanol and sodium.
Laboratory preparations can also be made by mixing acridone Chemicalbook with zinc powder and heating.
Acridine sublimation generated by the reaction is dissolved in hydrochloric acid, and then alkalised with sodium hydroxide solution, precipitate, filtered to obtain the crude product.
Recrystallisation with methanol gives a fine product with a melting point of 110°C.
- Method 2: Based on the condensation reaction of aniline and aldehyde or ketone
1.Condensation of aniline
firstly, aniline is condensed with formaldehyde under acidic conditions to produce N-methylaniline.
C6H5NH2 + CH2O → C6H5NHCH3 (N-methylaniline)
2.Bromination of methylaniline
Next, N-methylaniline reacts with bromine under suitable conditions to give N-methyl-N-bromoaniline.
C6H5NHCH3 + Br2 → C6H5N(CH3)Br (N-methyl-N-bromoaniline)
3.Coupling reaction
finally, two N-methyl-N-bromoaniline molecules react by coupling to form Acridine.
2C6H5N(CH3)Br → C13H9N (Acridine) + 2 HBr + 2 CH3Br
Recent Advances and Future Directions




Targeted Drug Delivery: Nanoparticles functionalized with acridine derivatives (e.g., liposomes, dendrimers) aim to improve drug solubility and reduce off-target effects in cancer therapy.
Organic Electronics: Acridine's electron-transport properties make it a candidate for organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs).
Bioorthogonal Chemistry: Acridine-based probes are being designed for live-cell imaging without disrupting native biochemical processes, enabling real-time tracking of metabolites and proteins.
Green Synthesis: Enzymatic and microbial synthesis routes are being explored to produce acridine derivatives under ambient conditions, minimizing chemical waste.
Acridine's unique structure and multifaceted reactivity have sustained its relevance across scientific disciplines for over a century. From its humble beginnings as a coal tar derivative to its modern roles in cancer therapy and sustainable chemistry, acridine continues to inspire innovation. Future research will likely focus on mitigating its toxicity, enhancing its specificity in biological applications, and expanding its utility in emerging technologies. As interdisciplinary collaboration grows, acridine's potential to address global challenges in health, energy, and the environment remains vast and promising.
Future Perspectives
Development of Novel Derivatives
The development of novel acridine derivatives with improved biological activities and reduced side effects is an active area of research. By modifying the structure of acridine, researchers can enhance its specificity for target molecules and reduce its toxicity to non-target cells. For example, the development of acridine derivatives with targeted delivery systems can improve their efficacy in cancer therapy.
Green Chemistry Approaches
The adoption of green chemistry approaches in the synthesis and use of acridine and its derivatives is expected to increase in the future. Green chemistry aims to minimize the environmental impact of chemical processes by using renewable resources, reducing waste generation, and improving energy efficiency. The development of green synthesis methods for acridine and its derivatives can contribute to a more sustainable chemical industry.
Multidisciplinary Research
The future of acridine research is likely to involve multidisciplinary approaches, combining chemistry, biology, materials science, and environmental science. By integrating knowledge from different fields, researchers can gain a deeper understanding of the properties and applications of acridine and develop innovative solutions to complex problems.
Frequently Asked Questions
What does acridine do?
+
-
Acridine is a polycyclic aromatic dye with antineoplastic, antimicrobial and imaging activities. Acridine and its derivatives intercalate within DNA and RNA by forming hydrogen-bonds and stacking between base pairs resulting in DNA crosslinks and strand breaks.
What is the mutation caused by acridine dyes?
+
-
This is called a frameshift mutation. Proflavin is an acridine dye that intercalates itself between base pairs of the DNA chain thereby causing loss or gain of a single nucleotide. The gene mutation alters the base sequence of the whole genetic frame from the point of mutation called frameshift mutation.
Is acridine orange carcinogenic?
+
-
Toxicity and carcinogenicity of AO. Since AO is mutagenic in bacteria (24, 25), it might also be carcinogenic in humans.
Hot Tags: acridine cas 260-94-6, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 5 Pyrimidinecarbonitrile 4 chloro 2 6 diphenyl , lidocaine hydrochloride powder, 2 6 Pyridinedicarboxylic acid, CAS 715 50 4, CAS 97870 64 9, CAS 2396743 57 8