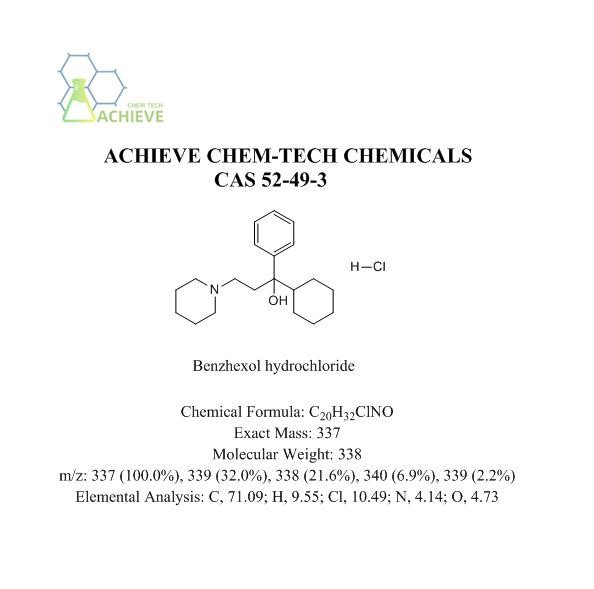
Benzhexol hydrochloride, molecular formula C20H31NO ClH,CAS 52-49-3. Usually a white lightweight crystalline powder, odorless, with a slightly bitter taste, which is beneficial for pain and numbness, and is not easily volatile. It is easily soluble in methanol, ethanol, or trichloromethane, but slightly soluble in water at 670.9ug/L (22.5 ° C). Although the solubility is not high, it can be fully dissolved in suitable solvents to prepare drug formulations. Stable at room temperature and not prone to chemical decomposition or degradation. It is a weakly acidic prodrug with amphiphilic properties, and methylprednisolone succinate is actively loaded into liposomes using a Ca gradient of acetic acid. In short, the [acetic acid Ca] liposome - [acetic acid Ca] medium transmembrane gradient is used as a driving force for long-distance loading of amphiphilic weakly acidic GC prodrugs. The characteristic of this gradient is that the Ca2- ions that cannot penetrate the membrane exceed the acetic acid that can penetrate the membrane.

|
Chemical Formula |
C20H32ClNO |
|
Exact Mass |
337 |
|
Molecular Weight |
338 |
|
m/z |
337 (100.0%), 339 (32.0%), 338 (21.6%), 340 (6.9%), 339 (2.2%) |
|
Elemental Analysis |
C, 71.09; H, 9.55; Cl, 10.49; N, 4.14; O, 4.73 |
|
|
|

Benzhexol Hydrochloride is a central anticholinergic drug that selectively blocks the striatal cholinergic pathway, regulating the balance between dopamine and acetylcholine. It is mainly used to treat Parkinson's disease and related motor disorders.

Core indications: Parkinson's disease and syndrome
1. Parkinson's disease
The core pathological feature of Parkinson's disease is the degeneration and death of dopaminergic neurons in the substantia nigra of the midbrain, leading to an imbalance between dopamine and acetylcholine, causing symptoms such as resting tremors, muscle rigidity, and bradykinesia. By inhibiting acetylcholine receptors, cholinergic neurotransmission is weakened, thereby restoring neurotransmitter balance, improving tremors (especially hand and head tremors) and muscle stiffness, and enhancing joint mobility. For example, patients may transition from slow gait and stiff body to walking freely after taking medication.
2. Parkinson's syndrome
Including secondary Parkinson's syndrome caused by encephalitis, arteriosclerosis, medication, etc. It has a significant effect on the tremor and rigidity symptoms of post encephalitis tremor paralysis, and can also be used for motor disorders associated with neurological diseases such as Wilson's disease and Huntington's disease, by regulating the balance of dopamine and acetylcholine to alleviate symptoms.


Antagonism of drug-induced extrapyramidal reactions
1. Extrapyramidal reactions caused by antipsychotic drugs
Antipsychotic drugs such as phenothiazine (such as chlorpromazine) and butyrylbenzenes (such as haloperidol) may cause symptoms such as sedentary behavior, acute muscle tone disorders (such as neck muscle spasms and eye roll), and late-onset motor disorders. By exerting anticholinergic effects, these symptoms can be effectively alleviated and patients' tolerance to psychotropic drugs can be improved.
For example, symptoms such as body twisting and facial muscle spasms can be alleviated after medication.
2. Movement disorders caused by other medications
Blood lowering drugs such as Li Xue Ping may cause extrapyramidal reactions by affecting neurotransmitters and can also be used in such situations.


Treatment of special motor disorders
1. Muscle tone disorder
Including local muscle tone disorders such as twisting spasms and writing spasms. By inhibiting central cholinergic nerve transmission, reducing muscle tone elevation and spasticity, improving abnormal posture or repetitive movements.
2. Adjuvant treatment for epilepsy and schizophrenia
In recent years, there have been reports that benzoxetine hydrochloride can be used to treat positive symptoms of epilepsy and schizophrenia (such as hallucinations and delusions), with a maximum dose of 20mg/day, but strict monitoring of adverse reactions is required.

The synthetic route of benzhexol hydrochloride can be realized through the following steps:
1. Conduct Knoevenagel condensation reaction of benzaldehyde and acetylacetone to obtain α-Phenyl-β-Acetone (alpha-phenyl-beta-ketoester).
2. Will α-Phenyl-β-Acetone and sodium hydroxide (NaOH) undergo Acyloin condensation reaction in ethanol (EtOH) to obtain α- Phenyl- β- Alpha-phenyl-beta-ethanol.
3. Will α- Phenyl-β-Chlorination reaction of propanol and phosphorus trichloride (PCl3) in chloroform (CHCl3) to obtain 3-chlorine- α- Phenyl-β-3-chloro-alpha-phenyl-beta-ethanol.
4. Add 3-chlorine-α-Phenyl-β-Benzhexol was obtained by Friedel-Crafts alkylation of propanol with triethylamine and ferric chloride in toluene.
5. It is obtained by reacting benzhexol with hydrochloric acid.
In general, the synthetic route of benhexol hydrochloride is complicated and requires multiple reaction steps and reagents. Therefore, the reaction conditions and drug quality should be strictly controlled in actual production and use to ensure the purity and quality of the drug.

It is a drug molecule with certain chemical properties. The following are its common chemical properties:
1. Can form hydrochloride:
It can be formed by reaction of benzhexol with hydrochloric acid.
01
2. Can catalyze esterification reaction:
Phenylhexol can be used as catalyst to catalyze esterification reaction.
02
3. Nuclear magnetic resonance can occur:
Phenylhexol molecule contains multiple nuclei, such as carbon and hydrogen, which can be detected by nuclear magnetic resonance technology.
03
4. Hydrogen bonding can occur:
Benzhexol molecule contains multiple hydrogen bonding receptors and donor sites, so hydrogen bonding can occur.
04
5. Can be oxidized:
Some functional groups in phenylhexol molecule can be oxidized to form corresponding oxidation products.
05
In a word, phenylhexol hydrochloride is a drug molecule with certain chemical properties. In drug research, it is often necessary to consider the influence of its chemical properties on drug efficacy and pharmacokinetics.
Benzhexol hydrochloride is a compound with certain chemical reactivity. The following are its common reaction properties:
1. Reaction with base:
Phenylhexol hydrochloride can react with base to generate corresponding free base and hydrochloride.
01
2. Reaction with acid:
Phenylhexol hydrochloride can react with acid to generate corresponding hydrochloride and corresponding acid.
02
3. Reaction with oxidant:
Phenylhexol hydrochloride can react with oxidant to produce bright sparks and smoke, which is dangerous to burn.
03
4. Reaction with metal ions:
Phenylhexol hydrochloride can react with some metal ions to form corresponding complexes or precipitates.
04
5. Heating decomposition:
It decomposes at high temperature, releasing nitrogen gas and leaving corresponding hydrocarbons.
05
It should be noted that the chemical properties of phenylhexol hydrochloride will be affected by environmental factors, such as temperature, humidity, light, etc. Therefore, it is necessary to avoid adverse environmental conditions during storage and use.
In this type of liposome, methylprednisolone succinate is "pumped" into the aqueous phase of the slightly alkaline liposome in its protonated (uncharged) form through lipid bilayer diffusion. There it loses its proton, becomes negatively charged, and forms insoluble methylprednisolone succinate calcium salt, which precipitates in the aqueous phase inside the liposome. Apply the liposome in the form of isotonic buffer such as saline and/or sucrose containing solution to patients with rheumatoid arthritis. Preferably, methylprednisolone succinate liposomes are slowly intravenously applied to one of the larger veins to reduce adhesion and uptake by endothelial cells. Methylprednisolone succinate liposomes adhere and bind to surrounding monocytes, which engulf methylprednisolone succinate liposomes. During this process, methylprednisolone succinate is released from liposomes and to some extent activates the ribocorticoid receptor. Once monocytes approach the inflammatory arthritis environment, it inhibits subsequent monocyte activation. Therefore, the differentiation of monocytes into macrophages and dendritic cells is inhibited or tends to induce a tolerant phenotype, and the possibility of monocyte migration into inflamed joints is reduced.
FAQ
What is benzhexol hydrochloride used for?
+
-
Trihexyphenidyl (Benzhexol) is an anti-muscarinic muscle relaxant used as an antispasmodic drug for both movement disorder (dystonia) and for control of secretions (drooling) in children. In dystonia, Trihexyphenidyl (Benzhexol) relaxes dystonic, constantly tensed muscles and reduces dystonic movements.
What is benzhexol used for in psychiatry?
+
-
Trihexyphenidyl (benzhexol, Artane) is an anticholinergic drug widely used among the psychiatric population in order to control extrapyramidal symptoms from the use of anti-psychotic drugs.
Is benzhexol a control drug?
+
-
Benzhexol is also indicated to control extrapyramidal disorders (eg akathisia manifested by extreme restlessness and dyskinesia characterised by spastic contractions and involuntary movements) due to central nervous system drugs such as reserpine and the phenothiazines.
Is benzhexol antipsychotic?
+
-
Anticholinergic drugs are used clinically to counteract EPS induced by antipsychotic drugs [21,22]. Benzhexol, also known as trihexyphenidyl, is the most widely used anticholinergic drug in China for the management of EPS.
Hot Tags: benzhexol hydrochloride cas 52-49-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 4 Methylpropiophenone, epinephrine powder, epinephrine hydrochloride powder, tetracaine hcl, isoflurane powder, tetramisole hydrochloride powder