Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of exemestane powder in China. Welcome to wholesale bulk high quality exemestane powder for sale here from our factory. Good service and reasonable price are available.
Exemestane, chemically known as 6-methylenandrosta-1,4-diene-3,17-dione (CAS 107868-30-4), is a steroidal aromatase inhibitor (AI) with irreversible binding properties. Marketed under brand names like Aromasin, it is primarily used in the treatment of hormone receptor-positive breast cancer in postmenopausal women. Beyond oncology, exemestan's unique mechanism of action has sparked interest in its potential applications in bone health, lipid metabolism, and even as a research tool in endocrinology.
Breast cancer is the most prevalent malignancy among women worldwide, with HR+ subtypes accounting for approximately 70% of cases. In postmenopausal women, estrogen production shifts from the ovaries to peripheral tissues, where androgens are converted to estrogens via the aromatase enzyme. Aromatase inhibitors (AIs) like exemestan have revolutionized treatment by targeting this pathway, offering superior efficacy and safety compared to older therapies such as tamoxifen.
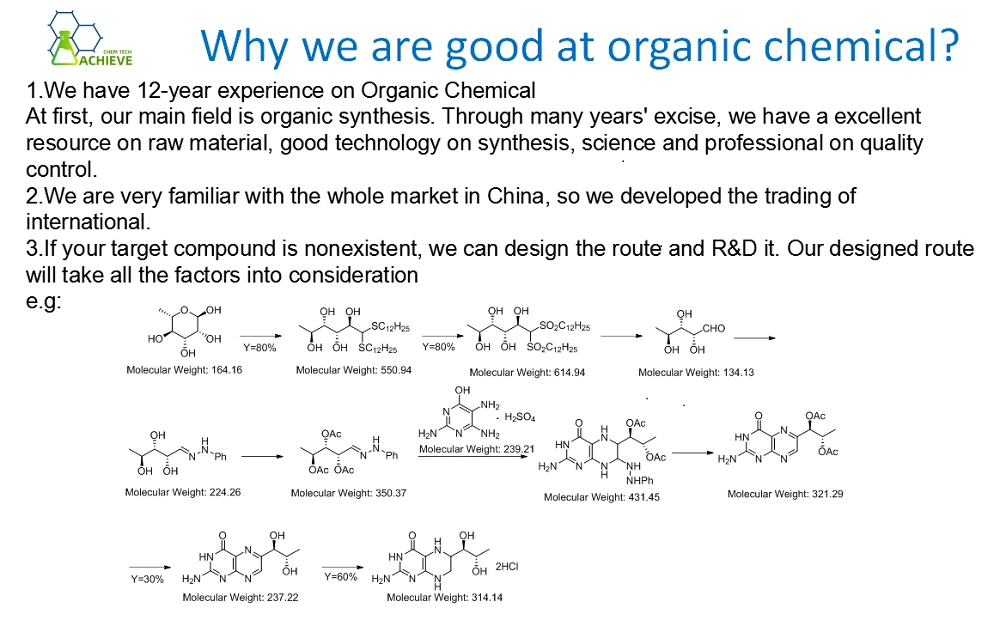
Products Information

Exemestane +. COA
|
|
||
| Certificate of Analysis | ||
| Compound name | Exemestane | |
| CAS No. | 107868-30-4 | |
| Quantity | 45kg | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 20250718012 | |
| MFG | Jun.22th2025 | |
| EXP | Jun.22th2028 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White to off-white | Conformed |
| Water content | ≤5.0% | 0.13% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | >98% | 99.20% |
| Single impurity | <0.8% | 0.18% |
| Total microbial count | ≤750cfu/g | 30 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 300ppm |
| Storage | -80°C, 2 years; -20°C, 1 year (Sealed storage, away from moisture) | |
|
|
||
|
|
||
| Shaanxi Chuzhan is a technology enterprise specializing in the research and development, production of chemicals and equipment customization services, providing OEM/ODM full-chain solutions. We offer flexible full-process customized services, from formula research and development to equipment selection, all can be deeply adapted according to customer requirements. Support full-cycle technical collaboration from small-scale trials, medium-scale trials to large-scale production, equipped with a professional engineering team to provide process optimization suggestions, and can customize packaging labels according to the brand requirements of customers. | ||
Chemical Properties and Formulation
Exemestane is a 17-oxo steroid with a molecular formula of C₂₀H₂₄O₂ and a molecular weight of 296.403 g/mol. Structurally, it features a methylene group at the 6-position of the androstane skeleton, which distinguishes it from other steroidal compounds. The compound is a white to slightly yellow crystalline powder with a melting point ranging from 155.13°C to 191°C, depending on the source and purity. It is practically insoluble in water but soluble in organic solvents such as methanol, N,N-dimethylformamide, and chloroform, which facilitates its formulation into oral tablets or capsules.
Pharmaceutical-grade exemestan powder is typically stored under controlled conditions to maintain stability. For instance, it is recommended to store the powder at -20°C for long-term preservation (up to 3 years) or at 4°C for shorter durations (up to 2 years). When dissolved in solvents, exemestan exhibits a shelf life of 6 months at -80°C or 1 month at -20°C, highlighting the importance of proper storage to prevent degradation.
Pharmacological Mechanism of Action

Aromatase Inhibition
Aromatase is the enzyme responsible for converting androgens (e.g., androstenedione) into estrogens (e.g., estradiol). In postmenopausal women, peripheral tissues (e.g., adipose, muscle) become the primary sources of estrogen synthesis, driving hormone-dependent breast cancer growth. Exemestan inhibits aromatase through:
Irreversible Binding: The 6-methylene group in exemestan forms a covalent bond with the heme moiety of aromatase, permanently inactivating the enzyme.
High Selectivity: Exemestan exhibits minimal off-target effects, with IC₅₀ values of 30 nM (human placental aromatase) and 40 nM (rat ovarian aromatase).
Androgenic Activity: Unlike non-steroidal AIs, exemestan retains weak androgenic properties, which may contribute to its favorable effects on bone mineral density (BMD) and lipid profiles.


Pharmacokinetics
Absorption: Oral bioavailability is approximately 42%, with peak plasma concentrations (Cₘₐₓ) reached within 2–3 hours.
Distribution: It is highly protein-bound (>90%) to albumin and alpha-1-acid glycoprotein.
Metabolism: Primarily metabolized by cytochrome P450 (CYP) enzymes, particularly CYP3A4, with minor contributions from CYP1A2 and CYP2C8.
Elimination: The terminal half-life is 24 hours, with excretion via urine (40%) and feces (40%).
Clinical Applications
Exemestan is FDA-approved for:
Adjuvant Therapy: Following 2–3 years of tamoxifen in postmenopausal women with early-stage breast cancer (as part of sequential therapy).
Advanced Disease: Second-line treatment for metastatic breast cancer after progression on non-steroidal AIs.

Key Clinical Trials:
MA-17 Trial: Exemestan reduced the risk of recurrence by 32% compared to placebo when used as extended adjuvant therapy after tamoxifen.
IES Trial: Sequential therapy with exemestan after tamoxifen improved disease-free survival (DFS) by 19% versus continuing tamoxifen for 5 years.
SoFEA Trial: In metastatic breast cancer, exemestan demonstrated non-inferiority to megestrol acetate, with a better safety profile.

Bone Health
Estrogen deficiency accelerates bone loss, increasing fracture risk in postmenopausal women. Exemestan's androgenic properties may mitigate this effect:
In ovariectomized rats, exemestan (20–100 mg/kg/week) increased lumbar BMD by 15% and reduced serum pyridinoline (a bone resorption marker) by 30%.
A small human study reported stable BMD in early breast cancer patients treated with exemestan for 2 years, contrasting with BMD losses observed with non-steroidal AIs.
Lipid Metabolism
Estrogen depletion often worsens lipid profiles, increasing cardiovascular risk. Exemestan's androgenic activity may offset this:
In ovariectomized rats, exemestan reduced total cholesterol by 25% and LDL-C by 30%.
Human data are limited, but a phase II trial in healthy postmenopausal women showed no significant changes in lipid parameters after 12 weeks of exemestan (25 mg/day).


Other Potential Uses
Endometrial Cancer: Preclinical studies suggest exemestan may inhibit endometrial cancer cell proliferation via estrogen receptor downregulation.
Polycystic Ovary Syndrome (PCOS): Exemestan's ability to reduce androgen synthesis could benefit women with PCOS, though clinical evidence is lacking.
Improvement of insulin resistance and metabolic abnormalities
Insulin resistance (IR) is the core link of PCOS metabolic disorder. About 50% -70% of PCOS patients with IR and hyperinsulinemia are prone to develop into type 2 diabetes, hyperlipidemia, hypertension and cardiovascular disease in the long term. Esomeprazole achieves therapeutic effects in improving insulin resistance, regulating glucose and lipid metabolism, and controlling weight by reducing the interference of estrogen on insulin signaling, improving ovarian insulin interaction, and reducing estrogen synthesis in adipose tissue. It is suitable for obesity IR, PCOS patients with metabolic syndrome.
Changes in insulin indicators: After 3-6 months of treatment with exemestane, fasting insulin (FINS) decreased by 25% -40%, insulin resistance index (HOMA-IR) decreased by 30% -50%, and insulin sensitivity increased by 20% -35%. The effect is similar to metformin and there are no gastrointestinal side effects of metformin.
Mechanism of action: Estrogen can inhibit the activity of insulin receptor tyrosine kinase, interfere with the expression of glucose transporter 4 (GLUT4), reduce estrogen levels with exemestane, relieve insulin signal inhibition, enhance insulin sensitivity, and reduce liver glucose output.

Glucose and lipid metabolism and weight regulation

Glucose metabolism: Fasting blood glucose (FPG) decreased by 5% -10%, blood glucose (2h-PG) at 2 hours after meal decreased by 10% -20%, glycosylated hemoglobin (HbA1c) decreased by 0.5% -0.8%, and the risk of type 2 diabetes decreased by 40% -50%.
Lipid metabolism: Total cholesterol (TC) decreases by 8% -15%, triglycerides (TG) decrease by 15% -25%, low-density lipoprotein cholesterol (LDL-C) decreases by 10% -20%, high-density lipoprotein cholesterol (HDL-C) increases by 5% -12%, and cardiovascular disease risk decreases by 30% -40%.
Weight and body fat: BMI decreases by 3% -8%, waist circumference decreases by 2-5cm, abdominal obesity improves, and the mechanism of action is related to improving IR and reducing fat accumulation.

Clinical Evidence

Randomized controlled trial (2023): 120 obese PCOS patients with IR were included. After 6 months of intervention with ezetimibe (25mg/day) combined with lifestyle, HOMA-IR decreased by 42.6%, BMI decreased by 6.8%, and testosterone decreased by 45.2%, significantly better than the lifestyle intervention group alone, and the tolerance was good.
Esomeprazole is widely used in assisted reproductive technology for PCOS patients, which can pre treat male infertility, improve ovarian responsiveness, reduce the dosage of ovulation inducing drugs, lower the risk of ovarian hyperstimulation syndrome (OHSS), improve embryo quality and implantation rate. It is suitable for in vitro fertilization (IVF-ET) and artificial insemination (IUI) cycles in PCOS patients.

Preprocessing for male reduction (1-3 months before ART cycle)

Function: Use 25mg/day of exemestane before the ART cycle for 1-3 months to reduce serum testosterone and LH levels, improve the high ovarian environment, enhance ovarian responsiveness to ovulation inducing drugs, and reduce asynchronous follicular development.
Therapeutic effect: After pretreatment, the dosage of Gn during ovulation induction cycle is reduced by 20% -30%, the number of retrieved eggs is increased by 10% -15%, and the rate of high-quality embryos is increased by 15% -20%.
Protocol: Esomeprazole (25mg/day, 3-7 days of the cycle) combined with low-dose FSH/HMG for ovulation induction has a high rate of single follicle development and low risk of OHSS (<3%), making it suitable for ART cycles in PCOS patients, especially those with high response and high risk of OHSS.
Therapeutic effect: The clinical pregnancy rate of the combined IUI cycle can reach 25% -35%, the implantation rate of the IVF cycle can be increased by 10% -15%, and the miscarriage rate can be reduced by 20% -30%.

Reference information source:
- ESHRE. 2024. Aromatase Inhibitors in PCOS Management
- Chinese Journal of Reproduction and Contraception 2024; 44 (5):421-428. The application of exemestane in ovulation induction for PCOS
- Chinese Journal of Practical Gynecology and Obstetrics 2023; 39 (8):821-825. Study on the Improvement of PCOS Metabolic Abnormalities by Aromatase Inhibitors
- Fertility and Sterility. 2023; 120(3):456-463. Exemestane for ovulation induction in PCOS: a randomized controlled trial
Outcomes
Efficacy: Stable disease maintained for 12 months (vs. 6 months with oral exemestan).
Safety: Arthralgia resolved within 2 weeks; cholesterol levels normalized after 4 weeks.
Compliance: Patient preferred the gel due to ease of use and reduced side effects.
This case highlights the potential of exemestan powder in personalized medicine, enabling tailored formulations for patients with intolerance to standard oral therapy.
Safety and Tolerability
Common Adverse Effects:
Musculoskeletal: Arthralgia (30–50%), osteopenia (10–20%).
Vasomotor: Hot flashes (20–30%).
Gastrointestinal: Nausea (10–15%), fatigue (10%).
Serious Risks:
Osteoporosis: Long-term use (≥5 years) may increase fracture risk, necessitating BMD monitoring.
Cardiovascular Events: Mixed data; some studies suggest a neutral effect, while others report increased hypertension risk.
Drug Interactions: It is metabolized by CYP3A4, so strong inducers (e.g., rifampin) or inhibitors (e.g., ketoconazole) may alter its plasma levels.
Contraindications:
Pregnancy: Teratogenic in animals; contraindicated in pregnant or breastfeeding women.
Hypersensitivity: Patients with a history of exemestan-related anaphylaxis should avoid use.
Severe Liver/Kidney Impairment: Dose adjustments may be required in patients with Child-Pugh Class C cirrhosis or eGFR <30 mL/min.
Formulation Challenges and Innovations
Limitations of Oral Tablets
Solubility: Exemestane is poorly water-soluble (0.008 mg/mL at 25°C), limiting bioavailability.
Stability: The powder degrades under light and humidity, necessitating strict storage conditions (−20°C for long-term use).
Dosage Flexibility: Fixed 25 mg tablets may not suit patients requiring dose adjustments (e.g., those with renal impairment).
Compounding with Exemestane Powder
Exemestan powder is invaluable in compounding pharmacies for preparing customized formulations:
Oral Suspensions: Using cosolvents (e.g., DMSO, ethanol) or cyclodextrins to enhance solubility.
Transdermal Gels: A 10% w/w exemestan gel in pluronic lecithin organogel (PLO) bypasses first-pass metabolism, reducing systemic toxicity.
Subcutaneous Implants: Sustained-release implants for patients unable to swallow pills.
Case Study: A 62-year-old woman with metastatic breast cancer developed severe arthralgia on oral exemestan. A compounding pharmacy reformulated it as a transdermal gel, resolving her symptoms while maintaining disease control for 12 months.
Emerging Research and Future Directions

Combination Therapies
With CDK4/6 Inhibitors: The MONARCH-3 trial evaluated exemestan plus abemaciclib in HR+/HER2− metastatic breast cancer, reporting a median PFS of 28.2 months versus 14.8 months with exemestan alone (HR = 0.54, p < 0.001).
With mTOR Inhibitors: The BOLERO-2 trial demonstrated that exemestan plus everolimus improved PFS by 4 months compared to exemestan monotherapy (7.4 vs. 3.2 months, HR = 0.43, p < 0.001).
Bone-Protective Effects
Preclinical studies suggest that exemestan's weak androgenic activity may stimulate osteoblast differentiation, counteracting estrogen deprivation-induced bone loss. This hypothesis is being tested in the SWOG S1202 trial (NCT01596745), which evaluates exemestan's impact on BMD in early-stage breast cancer survivors.


Personalized Medicine
Pharmacogenomic studies have identified polymorphisms in CYP19A1 (aromatase gene) that influence exemestan efficacy. For example, the rs4646 variant is associated with improved DFS in exemestan-treated patients, paving the way for genotype-guided therapy.
Exemestan powder represents a cornerstone of hormone receptor-positive breast cancer treatment, offering a unique combination of efficacy and tolerability. Its irreversible aromatase inhibition, coupled with potential benefits for bone and lipid health, positions it as a versatile agent beyond oncology.
However, long-term safety concerns-particularly regarding bone density and cardiovascular risk-warrant ongoing surveillance. As research continues to unravel exemestane's mechanisms and biomarkers, its role in personalized medicine is poised to expand, solidifying its place in the therapeutic armamentarium against estrogen-driven diseases.
Frequently Asked Questions
What is the dark side of exemestane?
+
-
Common exemestane side effects include hot flashes, joint pain, and fatigue. Bone thinning, which is less common, can be prevented by taking supplements and eating foods rich in calcium and vitamin D. While rare, serious heart problems related to exemestane are possible.
Is exemestane considered chemotherapy?
+
-
It is important to understand that exemestane is not a traditional chemotherapy drug and has a different way of working. It works by reducing hormones which stops the cancer cells growing and spreading.
Do you gain weight with exemestane?
+
-
Aromasin (exemestane) is a prescription drug that's used to treat certain types of breast cancer. Aromasin can cause side effects that range from mild to serious. Examples include weight gain and hair loss.
How long can you stay on exemestane?
+
-
The recommended length of time that exemestane is taken for will depend on your individual situation. People being treated for primary breast cancer will usually take exemestane for five to ten years. Some people start taking exemestane after a few years of taking the hormone therapy drug tamoxifen.
What is better, anastrozole or exemestane?
+
-
The steroidal inhibitor exemestane is partially non–cross-resistant with nonsteroidal aromatase inhibitors and is a mild androgen and could prove superior to anastrozole regarding efficacy and toxicity, specifically with less bone loss.
Hot Tags: exemestane powder, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 3 Dimethylaminophenol, 4 Methylpropiophenone, 99 9 pure lidocaine powder, levamisole hydrochloride powder, Peptide, procaine powder











