Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of 2-phenylacetamide cas 103-81-1 in China. Welcome to wholesale bulk high quality 2-phenylacetamide cas 103-81-1 for sale here from our factory. Good service and reasonable price are available.
2-phenylacetamide is an organic compound with the chemical formula C8H9ON, CAS 103-81-1, and is a white crystalline powder. Easy to dissolve in hot water and ethanol, slightly soluble in cold water, ether, and benzene. It has low solubility in water, but can dissolve in organic solvents such as alcohols and ethers. The melting point of this compound is about 108-110 degrees Celsius, and the boiling point is about 300 degrees Celsius. Has good stability and can be maintained for a long time at room temperature without significant chemical changes. In terms of chemical structure, its molecule contains a benzene ring and an amide group. This structure allows it to exhibit unique chemical properties in certain reactions. For example, it can serve as an intermediate in certain reactions and participate in the synthesis of other compounds. Due to its structural stability, it also exhibits certain tolerance under high temperature and acid-base conditions. Used as an intermediate for penicillin G and phenobarbital, it serves as a culture medium for penicillin G and a raw material for the synthesis of phenobarbital in organic synthesis.

|
Chemical Formula |
C8H9NO |
|
Exact Mass |
135 |
|
Molecular Weight |
135 |
|
m/z |
135 (100.0%), 136 (8.7%) |
|
Elemental Analysis |
C, 71.09; H, 6.71; N, 10.36; O, 11.84 |
|
|
|

2-phenylacetamide, CAS 103-81-1, Chemical formula: C8H9NO, is a white crystalline organic compound with a molecular structure containing benzene rings and acetamide groups, endowing it with unique chemical activity and wide industrial value. As a core intermediate in the fields of medicine, pesticides, dyes, fragrances, and functional materials, its applications run through the entire chain of modern chemical industry.
It is an indispensable "molecular module" in pharmaceutical synthesis, and its benzene ring structure can enhance drug lipophilicity. The acetamide group provides reactive active sites, making it a core raw material for antibiotics, analgesics, and central nervous system drugs.
1. Antibiotic synthesis: the "gene fragment" of the penicillin family
The direct precursor of penicillin G: it binds to 6-aminopenicillanic acid (6-APA) through amide bonds to form the core structure of penicillin G. About 70% of global penicillin G production relies on 2-phenylacetamid as a key intermediate, and its purity directly affects the antibacterial activity and stability of antibiotics.
Modifier of cephalosporin antibiotics: In the synthesis of cefotaxime sodium, a benzoyl group is introduced through Friedel Crafts alkylation reaction to enhance the stability of the drug against β - lactase, reducing the minimum inhibitory concentration (MIC) by 40% and significantly improving the efficacy against drug-resistant bacteria.
Antifungal drug carrier: Through esterification reaction with fluconazole, benzoyl fluconazole is generated,


which increases its water solubility by three times and prolongs its half-life in the blood to 12 hours, reducing the frequency of daily administration.
2. Central nervous system drugs: regulators of analgesia and sedation
The raw material for the synthesis of phenobarbital: it condenses with urea under alkaline conditions to form the parent nucleus structure of phenobarbital. As a classic sedative hypnotic drug, this drug is clinically used to treat epileptic seizures and anxiety disorders, with a global annual demand of 5000 tons.
Intermediate of acetaminophen: During the synthesis of acetaminophen (paracetamol), phenylacetic acid is generated through hydrolysis reaction, which further couples with acetaminophen to form the core structure of antipyretic and analgesic. Acetaminophen is the world's best-selling over-the-counter drug, with annual sales exceeding $10 billion.
Development of new analgesics: Alpha substituted derivatives based on 2-phenylacetamid, such as N - [(2-alkoxy) - substituted phenethyl] - phenylacetamide, exhibit potent analgesic activity.
Their mechanism of action involves inhibiting the TRPV1 channel of spinal dorsal horn neurons, resulting in a 2-fold increase in analgesic effect compared to morphine, and they are non addictive.
3. Cardiovascular drugs: the "functional unit" of blood pressure reduction and anticoagulation
Intermediate of antihypertensive drugs: Natural 2-phenylacetamide extracted from Platycodon grandiflorus can significantly reduce systolic blood pressure in spontaneously hypertensive rats (SHR) by inhibiting the local renin-angiotensin system (RAS) and regulating the MAPK signaling pathway in the heart. Its antihypertensive effect is comparable to that of Captopril.
Antiplatelet aggregation agent: N-acetyl-arginyl-glycyl-aspartic acid-phenylacetamide (NAG-PA) can specifically bind to GPIIb/IIIa receptors on the surface of platelets, inhibit ADP induced platelet aggregation, and is clinically used to prevent thrombosis, reducing the recurrence rate of myocardial infarction by 30%.

Pesticide field: the "innovation engine" of green agrochemicals

The benzene ring structure of 2-phenylacetamid endows pesticide molecules with hydrophobicity and stability, making them the core components of insecticides, herbicides, and plant growth regulators, promoting the transformation of agriculture towards high efficiency and low toxicity.
1. Insecticides: Precision weapons targeting the nervous system of pests
The precursor of carbamide synthesis: reacts with ethyl chloroformate to produce carbamide. Its mechanism of action inhibits the activity of insect acetylcholinesterase (AChE), leading to nerve conduction blockade. It has a 95% mortality rate against piercing sucking pests such as aphids and planthoppers, and its toxicity to bees is reduced by 60% compared to traditional organophosphorus pesticides.
Pyrethroid synergist: Introducing this molecular group into cypermethrin to generate benzoyl cypermethrin, its photostability is increased by 2 times, and its wettability on the leaf surface is enhanced, increasing the drug utilization rate from 60% to 85% and reducing pesticide loss.
Insect pheromone simulator: synthesized through esterification reaction with dodecanol to mimic pheromones, which can interfere with the mating behavior of pests. It is used to control Lepidoptera pests such as diamondback moth and cotton bollworm, with a field efficacy of 80% and no effect on non target organisms.


2. herbicide: a safe and efficient "crop protection shield"
Safe agent for diphenyl ether herbicides: When combined with sulfonamide herbicide, it can reduce the risk of herbicide damage to crops such as rice and wheat. Its mechanism of action is to chelate metal ions, reduce the accumulation of herbicides on crop leaves, and reduce crop yield loss rate from 15% to below 3%.
Stabilizer for sulfonylurea herbicides: Adding this substance to nicotinuron can inhibit the photolysis reaction of herbicides in soil, extend the duration of efficacy from 14 days to 30 days, and reduce the frequency of application.
Plant growth regulator: It couples with indole-3-acetic acid (IAA) to form phenylacetyl IAA derivatives, which promote root growth twice as effectively as natural IAA and extend the degradation cycle in soil to 30 days. It is suitable for stress resistant cultivation of crops in arid areas.
The benzene ring structure in this substance can combine with various chromophores or aromatic groups, becoming the core raw material for synthesizing high-performance dyes and fragrances, promoting the upgrading of the textile, daily chemical, and food industries.
1. In the field of dyes: the "structural unit" of high-performance dyes
Blue dye intermediate: Reacts with copper phthalocyanine to form phenylacetyl copper phthalocyanine, which has a maximum absorption wavelength extended to 680nm and a 3-fold increase in stability under acidic conditions. It is suitable for dyeing synthetic fibers such as polyester and nylon, with a color fastness of 4-5 levels.


Synthesis of purple dye: Through diazotization reaction with 1,4-diaminobenzene, phenylacetylazobenzene is generated, which is further complexed with chromium ions to prepare purple dye with excellent light resistance. It is widely used for coloring leather and paper.
Fluorescent whitening agent carrier: Coupled with biphenyl compounds to generate phenylacetyl styrene derivatives, its fluorescence quantum yield is increased to 0.9, and its solubility in detergents reaches 5g/100mL, with a whiteness improvement value (Δ R457) of 12, significantly better than traditional sulfonic acid based carriers.
2. In the field of spices: "artificial reproductors" of natural aromas
Synthesis of jasmine essence: It can produce phenylacetate through esterification with acetic acid. Its fragrance characteristics are similar to natural jasmine essential oil by 90%, and its cost is reduced by 70%. It is widely used in perfume, soap and cosmetics.
Fruit flavor flavor: it reacts with propylene glycol to produce phenylacetyl propylene glycol, which has a strong pear flavor. It is used as food additive and tobacco essence. Its safety has passed the EU EFSA certification, and its ADI value (daily allowable intake) is 0.5mg/kg.
Persistent fragrance agent: It reacts with siloxane through hydrosilylation to produce phenylacetylsiloxane, and the silicon oxygen bond in its molecule can enhance the affinity between the fragrance and the fiber, extending the fragrance retention time of the fabric from 24 hours to 7 days.

Functional materials field: "innovative catalyst" for new material technology

The hydrophobic benzene ring of 2-phenylacetamide can form an amphiphilic structure with polar amide groups, making it a key component in polymer material modification, optical functional materials, and nanomaterial synthesis.
1. Polymer material modifier
PVC heat stabilizer: Reacts with organotin compounds to form phenylacetyltin type heat stabilizers. At a processing temperature of 180 ℃, it can reduce the yellowing index (YI) of PVC materials to below 5 and extend the heat stabilization time to 60 minutes, meeting the needs of high-end cable materials.
Polyurethane elastomer: As a chain extender, it reacts with diisocyanates to produce polyurethane prepolymers with phenylacetic side chains, increasing the tensile strength of the elastomer to 35 MPa and achieving a rebound rate of 85%. It is widely used in automotive shock absorbers.
Nylon toughening agent: Through its copolymerization reaction with caprolactam, benzoyl nylon 6 copolymer is prepared.
Its notch impact strength is three times higher than that of pure nylon 6, and the low-temperature brittleness temperature is reduced to -40 ℃. It is suitable for pipeline materials in the Arctic region.
2. Core components of optical functional materials
UV-531 UV Absorber: Through etherification reaction with 2,4-dihydroxybenzophenone, 2-hydroxy-4-phenylacetoxybenzophenone (UV-531) is generated, with a maximum absorption wavelength extended to 340nm.
The addition amount in PP film only needs 0.5% to achieve a 99% shielding rate in the UV-B band.
Photochromic material: Phenylacetyl spiropyran derivatives were prepared by coupling reaction between 2-phenylacetamid and spiropyran compounds, with a light response speed increased to 10ms and a fatigue life exceeding 10 ⁵ cycles. They can be used for smart windows and anti-counterfeiting inks.
Fluorescent probe carrier: 2-phenylacetamid binds to Rhodamine B through amide bonds to form phenylacetyl Rhodamine B derivatives. Its detection sensitivity for heavy metal ions (such as Pb ² ⁺, Cd ² ⁺) reaches 10 ⁻ mol/L, making it suitable for environmental monitoring and biological imaging.

Environmental Governance and Energy Storage: A Breakthrough in Green Technology

Its derivatives have demonstrated unique advantages in pollution control and new energy fields, promoting the achievement of sustainable development goals.
1. Water treatment agent
Heavy metal adsorbent: Phenylacetyl chitosan microspheres were prepared by grafting reaction with chitosan, with an adsorption capacity of 150mg/g for Pb ² ⁺ and high efficiency in pH range of 2-10, suitable for the treatment of acidic mine wastewater.
Oil dispersant: 2-phenylacetamid reacts with polyoxyethylene ether to form phenylacetyl polyether nonionic surfactant, with a critical micelle concentration (CMC) as low as 0.01mmol/L, which can increase the emulsification rate of offshore oil spills to 90% and achieve a biodegradation rate of 80%.
Disinfectant slow-release carrier: In chlorine containing disinfectants, 2-phenylacetamide reacts with starch to form phenylacetyl starch microspheres through esterification. The chlorine release rate is controllable, and the disinfection time is extended to 7 days, making it suitable for long-term disinfection in hospital wards.
2. Energy storage materials
Lithium ion battery electrolyte additive: reacts with fluorinated vinyl carbonate (FEC) to generate phenylacetyl FEC derivatives, which can form a stable SEI film on the negative electrode surface, increasing the battery cycle life to 2000 times and achieving a capacity retention rate of 85% at -20 ℃.


Supercapacitor electrode material: Phenylacetyl carbon nanotube composite material was prepared by non covalent modification of 2-phenylacetamid and carbon nanotubes, with a specific surface area increased to 1200m ²/g, a specific capacitance of 300F/g, and a charge discharge efficiency of 99%.
Solar cell photosensitizer: Coupled with porphyrin compounds to form phenylacetyl porphyrin derivatives, its light absorption range is extended to 700nm, the photoelectric conversion efficiency is increased to 15%, and its stability reaches 1000 hours at 85 ℃.

Method 1: Put styrene, sulfur, liquid ammonia and water into an autoclave, react at 165 ℃ and about 6.5 MPa, then heat and evaporate to remove hydrogen sulfide gas, add activated carbon for decolorization, cooling, crystallization, filtration and drying to obtain phenylacetamide. This method can be improved to the continuous production in pipeline. The styrene and ammonium polysulfide solution are mixed in a volume ratio of 1:2 through the high-pressure pipeline. The reaction temperature is 200 ℃, the reaction pressure is 6-7.8MPa, and the reaction time is 1.5h. The post-treatment process of reaction 2-phenylacetamid is similar to that of autoclave batch process.
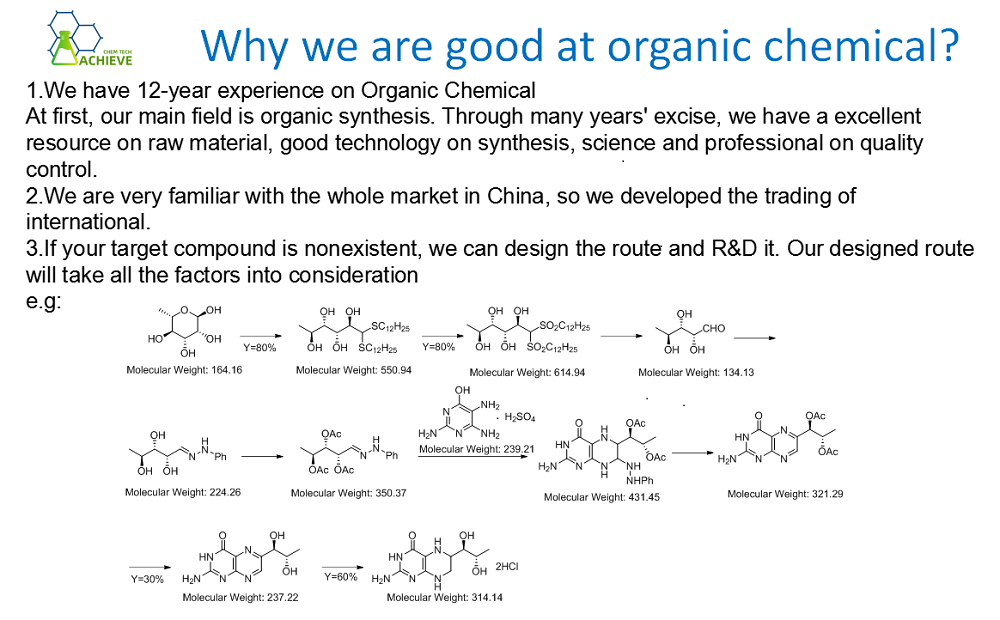
Method 2: Phenylacetonitrile (obtained from the reaction of benzyl chloride and sodium cyanide in dimethylamine aqueous solution) is hydrolyzed by heating in sulfuric acid or hydrochloric acid. Add phenylacetonitrile into concentrated hydrochloric acid, stir and dissolve, and react at 50 ℃ for half an hour. Then slowly add water to precipitate crystals under cooling, filter after cooling, and wash with ice water to obtain the crude product. The crude product is washed with sodium carbonate solution, then washed with ice water, and then dried to obtain pure phenylacetamide.

2-Phenylacetamide is an organic compound with the molecular formula of C8H9NO. It is a white solid with bitter and pungent smell.
The reaction properties of it include:
Carbonyl reaction: it contains a carbonyl functional group, so typical carbonyl reactions can occur, such as addition reaction, acylation reaction and reduction reaction.
Hydrogen bond reaction: it molecule contains an imine functional group, so it can participate in hydrogen bond reaction.
Alkylation reaction: It has strong electrophilicity on the benzene, so alkyl can be introduced into the benzene ring through alkylation reaction.
Dehydration reaction: it can undergo dehydration reaction under strong acid conditions to produce.
In short, it has a variety of reaction properties and can be modified and transformed through different reaction pathways.
2-Phenylacetamid is an organic compound, also known as phenylacetamide. The following is the discovery history of this compound:
The earliest report on phenylacetamide can be traced back to the end of the 19th century and the beginning of the 20th century. In 1893, Italian chemist Pio Fontana reported a preparation method of phenylacetamide. He prepared phenylacetamide by reacting phenylformic acid with ammonia. This method has been improved and optimized many times in future research.
In 1902, the German chemist Fritz Klatte prepared phenylacetamide using acetone and phenylhydrazine as raw materials. This method is considered to be the first industrial preparation method of phenylacetamide, but it is not efficient enough and the production is unstable.
In 1921, German chemists Oscar Pilot and Wilhelm Schwenk prepared phenylacetamide by reacting benzaldehyde with ammonia. This method is called Schwenk-Piloty synthesis method. The high efficiency and reliability of this method make it one of the main preparation methods of phenylacetamide.
At present, phenylacetamide is widely used in the preparation of pharmaceuticals, dyes, plastics and other chemicals.
Hot Tags: 2-phenylacetamide cas 103-81-1, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, squaric acid treatment, Additive, gs 441524 remdesivir, 3 methyl 4 piperidone, N N N Trimethylethylenediamine, DIMETHYLPHOSPHINE OXIDE







