Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of cobalt acetate cas 71-48-7 in China. Welcome to wholesale bulk high quality cobalt acetate cas 71-48-7 for sale here from our factory. Good service and reasonable price are available.
Cobalt acetate, has a molecular formula of C4H6CoO4. Red purple crystal or crystalline powder easy to deliquescence. With slight acetic acid smell, it becomes anhydrous when heated to 140 ℃. It is soluble in water, ethanol, dilute acid and amyl acetate. Toxic and carcinogenic substances should be stored in a closed container. Inhalation and ingestion are harmful to the body. Inhalation may cause pharyngitis, followed by gastrointestinal irritation symptoms, such as vomiting, abdominal cramps, temperature rise, calf weakness, etc. Skin contact can cause allergic dermatitis and contact dermatitis. It is used as an analytical reagent, catalyst, drying paint, and also as the ingredient of ceramic glaze.

|
Chemical Formula |
C4H6CoO4 |
|
Exact Mass |
177 |
|
Molecular Weight |
177 |
|
m/z |
177 (100.0%), 178 (4.3%) |
|
Elemental Analysis |
C, 27.14; H, 3.42; Co, 33.29; O, 36.15 |
|
|
|

There are three most common methods for the synthesis of cobalt acetat:
1. Cobalt metal or cobalt oxide method
Cobalt nitrate solution is prepared by reaction of metal cobalt or cobalt oxide with nitric acid, and then reacts with ammonium bicarbonate (or soda ash) solution to generate cobalt carbonate precipitation; Washing and separating the precipitate, adding acetic acid for acidification to obtain cobalt acetat; After filtering, adjust the reaction solution to pH=2, and concentrate it until the relative density of the solution is 1.17-1.20. Then add 1kg acetic acid every 200L of concentrated solution, crystallize it for 7-10d at a temperature lower than 20 ℃, centrifugate it, and dry it at 30-40 ℃ to obtain the finished product.
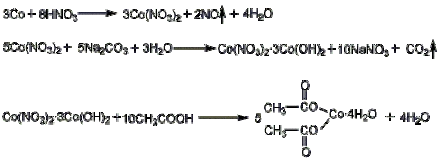
2. Cobalt sulfate method
Cobalt sulfate is heated to react with soda ash to generate cobalt carbonate, which reacts with acetic acid. The reaction product is filtered, concentrated, crystallized, dehydrated and packaged to obtain the product.

3. Take 5g of cobalt nitrate hexahydrate and add it to 25mL of acetic anhydride, mix it evenly, heat it and boil it for 10min, and then precipitate it through standing crystal. Filter by suction and wash with a small amount of acetic anhydride and ether. Drain and vacuum dry at 100 ℃ for 1h.
Purification method of cobalt acetate:
The purification method of cobalt acetat includes the following steps:
Cobalt acetat acetic acid solution obtained by soda precipitation method is adsorbed by an adsorption column containing modified ATBS copolymer medium at room temperature and 0.1-0.25MPa. The adsorbed cobalt acetat acetic acid solution is concentrated to 20-95wt%, and then cooled to 5-25 ℃ for recrystallization. The purified cobalt acetat product is collected. In the invention, the cobalt acetat acetic acid solution obtained by soda precipitation method is column adsorbed through the modified ATBS copolymer medium, and the ATBS copolymer medium has strong adsorption capacity for impurity metal ions in the solution, effectively removing the impurity metal ions. In this method, the ATBS copolymer is modified with sodium carbonate weak base solution, which can effectively remove the acidic monomer in the copolymer and prevent it from reacting with cobalt ion to generate other impurities. The modified ATBS copolymer medium of this method can be eluted with 0.1mol/L sodium carbonate solution after the use effect decreases, and can be reused. The method has simple operation, stable quality, low energy consumption and is suitable for industrial production.

Cobalt acetat (chemical formula: (CH3 COO) ₂ Co), also known as cobalt acetat, is a purple red hygroscopic crystal with a weak acetic acid odor. As an important organic chemical raw material, its application fields cover multiple industries such as catalysts, coatings, new energy, environmental protection, pharmaceuticals, and electronic materials.
Ester exchange reaction catalyst:
As an efficient catalyst in organic synthesis, it is widely used in the synthesis of ester compounds such as acrylic esters and methyl methacrylate. Its catalytic mechanism is based on the coordination of cobalt ions, which can reduce the activation energy of the reaction and improve the yield. For example, in the synthesis of methyl methacrylate, the addition of this substance can shorten the reaction time by 30% and reduce the generation of by-products.
liquid-phase oxidation catalyst:
It exhibits excellent performance in liquid-phase oxidation reactions, such as the preparation of terephthalic acid. Through the redox cycle of cobalt ions, the oxidation process of p-xylene can be efficiently catalyzed, and the purity of the product can reach over 99%. In addition, in the reaction of toluene oxidation to benzaldehyde, the synergistic catalytic system of this component and bromobenzyl can increase the conversion rate to 85%.


(2) In the field of coatings and desiccants
Paint drying agent:
As a traditional drying agent, it significantly shortens the drying time by promoting the oxidative crosslinking of unsaturated fatty acids in coatings. Adding 0.5% -1% of this substance to alkyd resin coatings can shorten the surface drying time from 8 hours to 2 hours, while improving the hardness and weather resistance of the coating.
Ceramic glaze additive:
In ceramic glaze formulas, as a coloring agent and flux, it can give the glaze a unique purple red color and reduce the firing temperature. For example, adding 0.3% cobalt acetate to high-temperature porcelain glaze can lower the firing temperature by 50-80 ℃ and improve the glossiness of the glaze surface.
(3) In the field of new energy
Lithium battery positive electrode material:
With the explosive growth of the new energy vehicle industry, cobalt salts, as an important form, have become a key raw material for positive electrode materials in ternary lithium batteries (NCM/NCA). It enhances the stability of the layered structure of the material and improves the charge discharge cycle life by providing cobalt ions. For example, in NCM811 material, doping with this substance can achieve a battery energy density of 280Wh/kg and a cycle life of over 2000 times.
Electrode materials for supercapacitors:
High performance electrode materials can be prepared in solid-state reactions. For example, by mixing and grinding it with oxalic acid and graphene oxide, and then calcining it, cobalt trioxide graphene composite materials can be obtained, with a specific capacitance of up to 1200F/g, which is more than three times that of traditional activated carbon materials.


(4) Environmental protection field
Flue gas desulfurization and denitrification:
As an efficient desulfurizer, it performs well in wet flue gas desulfurization. Research by the Chinese Academy of Environmental Sciences shows that under pH conditions of 5-6, its desulfurization efficiency can reach over 95%, and the by-product cobalt sulfate can be recycled and reused. In addition, it can also be used for the preparation of low-temperature SCR denitrification catalysts, with a NOx conversion rate of up to 90% at 200 ℃.
Industrial wastewater treatment:
It can be used as a heavy metal ion precipitant in electroplating wastewater treatment. For example, adding cobalt acetat to chromium containing wastewater can generate insoluble cobalt chromium composite oxide precipitates, reducing the concentration of chromium ions from 500mg/L to below 0.5mg/L, meeting national discharge standards.
(5) In the fields of medicine and agriculture
Pharmaceutical intermediates:
It is an important intermediate in the synthesis of vitamin B12. In the process of producing vitamin B12 by fermentation, cobalt acetat as a cobalt source can promote microbial metabolism and increase yield. In addition, it can also be used for the synthesis of anticancer drug cisplatin, stabilizing the drug molecular structure through the coordination effect of cobalt ions.
Plant growth regulator:
In the field of agriculture, it can be used as a trace element fertilizer to promote plant root development and photosynthesis. Research has shown that spraying 0.01% cobalt acetat solution in tomato cultivation can increase yield by 15% -20% and enhance plant stress resistance.


(6) In the field of electronic materials
Conducting material:
It can be used in electronic materials to prepare high-performance conductive pastes. For example, by mixing it with silver powder, a conductive paste with a resistivity lower than 10 ⁻⁴Ω· cm can be prepared, which is widely used in fields such as touch screens and solar cells.
Magnetic material:
Cobalt acetate is an important raw material for preparing ferrite magnetic materials. By controlling the calcination conditions, cobalt ferrite (CoFe2O4) with different magnetic properties can be obtained, and its saturation magnetization can reach 80 emu/g, which is suitable for high-frequency transformers, inductors and other devices.
Hot Tags: cobalt acetate cas 71-48-7, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale








