Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of sodium tetraborate decahydrate (borax) cas 1303-96-4 in China. Welcome to wholesale bulk high quality sodium tetraborate decahydrate (borax) cas 1303-96-4 for sale here from our factory. Good service and reasonable price are available.
Sodium tetraborate decahydrate (borax), called sodium borate, is also known as Moonstone. Borax includes sodium tetraborate decahydrate, sodium tetraborate pentahydrate, and anhydrous sodium tetraborate. Colorless translucent crystal or white crystalline powder, monoclinic system. It is odorless, salty, soluble in water and glycerin, insoluble in ethanol and acid, and the aqueous solution is weakly alkaline. Its density is 1.73g/cm3, and it weathered in dry air. When the temperature is higher than 56 ℃, pentahydrate is precipitated from the solution; When the temperature is lower than 56 ℃, decahydrate will be precipitated; Heating to 350 ~ 400 ℃, complete dehydration to anhydrous salt; Heat it to 878 ℃ and melt it into a glass. Molten borax can dissolve many metal oxides to form compound salt of metaboric acid with characteristic color.

This property of borax is called the "borax bead test." Sodium tetraborate decahydrate, also known as sodium pyroborate, is an inorganic compound, generally written as Na2B4O7 · 10h2o, with a molecular weight of 381.37. Borax is an essential boron-bearing mineral and boron compound. It is usually a white powder containing colorless crystals and is easily soluble in water. Borax has a wide range of uses. It can be used as a detergent, cosmetics, insecticide, buffer solution, and other boron compounds. Borax is highly toxic, and it is forbidden to be used as a food additive in many countries worldwide. If the human body ingests too much boron, it will cause cumulative poisoning of multiple organs.
|
Chemical Formula |
B4H20Na2O17 |
|
Exact Mass |
382 |
|
Molecular Weight |
381 |
|
m/z |
382 (100.0%), 381 (74.5%), 380 (37.0%), 381 |
|
Elemental Analysis |
B, 11.34; H, 5.29; Na, 12.06; O, 71.32 |
|
|
|
|
Boron is a necessary growth element for the human body, animals, and plants (especially beans, vegetables and fruits). It mainly exists in the form of boric acid, borax, and their complexes with sodium, magnesium, and calcium. The chemical composition of borax is sodium tetraborate, which exists in the form of boric acid under acidic conditions. Trace boron is beneficial to the human body, such as preventing osteoporosis and promoting calcium metabolism in the human body. After eating borax, the substance will be converted into boric acid by gastric acid and absorbed by the gastrointestinal tract. Although the intake is not much each time, the discharge speed is plodding, so little makes a lot, and eventually, it will accumulate in the body and be poisoned. Borax is toxic to many organs of the human body, endangers digestive tract enzymes, decreases appetite, and inhibits the absorption of various nutrients, thus speeding up the decomposition of fat, reducing weight, acute vomiting, diarrhea, dizziness, headache, and other symptoms, even erythema, circulatory system disorders, shock, coma, and other boric acid symptoms.

Sodium tetraborate decahydrate (borax) is the raw material for preparing boron-containing compounds. Almost all borides can be designed by borax. They are widely used in metallurgy, steel, machinery, military, cutting tools, papermaking, electronic tubes, chemical, textile, and other sectors.

Glass and enamel industry: in glass, it can enhance the transmittance of ultraviolet rays and improve glass's transparency and heat resistance. In enamel products, the glaze is not easy to fall off and has luster. It is also widely used in special optical glass, glass fiber, non-ferrous metal welding agents, jewelry binder, printing and dyeing, washing (silk and wool products, etc.), gold refining, cosmetics, pesticides, fertilizers, borax soap, preservatives, antifreeze, and medical disinfectants.
Medical field: borax is used for the treatment of skin mucous membrane, some tinea pedis, and also for the treatment of some inflammation, such as acute tonsillitis, pharyngitis, sore tongue, gingivitis, otitis media, fungal vaginitis, cervical erosion, epilepsy, tumor, etc. It also has remarkable effects in treating some animal diseases, such as chicken laryngotracheitis, goat infectious pustulosis, swine mycoplasma pneumonia, bovine chronic mucinous endometritis, etc. Boron is an essential element for plant growth, human beings, and animals.


Other applications: borax is widely used in industrial and agricultural production. For example, it can be used as filler and enhance surface activity (cross-linking) in washing powder and soap; It is also widely used in glass fiber, welding non-ferrous metals, bonding jewelry, cosmetics anti-corrosion, pesticides, fertilizers, antifreeze, and medical disinfectants. It is used as a herbicide in agriculture and for killing weeds in non-cultivated areas.
Industrial field: borax is an essential industrial boron mineral in industry. Borax is the most critical compound of boron. Boron is often listed as a rare element in foreign countries, but there are abundant borax ores in China. Therefore, boron is not a rare element in China but a high-yield element. Borax is also a solid lubricant for metal wire drawing in the industry. In the welding and maintaining refrigerators, freezers, air conditioners, and other refrigeration equipment, it is often used as an (inactive) flux to purify the metal surface. Remove oxides from metal surfaces; Add a certain proportion of sodium chloride to borax. Sodium fluoride, potassium chloride, and other compounds can be used as an active flux for welding copper pipes and steel pipes and between steel pipes in refrigeration equipment.


Antibacterial effect
There are reports indicating that borax is a weak base that has weak antibacterial effects, just like boric acid. Using a plate method to make the culture medium contain 10% borax, it has inhibitory effects on Escherichia coli, Pseudomonas aeruginosa, Bacillus anthracis, Shigella dysentery, Salmonella typhi, Salmonella paratyphoid, Proteus mirabilis, Staphylococcus aureus, and Candida albicans; Using the paper method, sodium tetraborate decahydrate (borax) has been proven that borax can also inhibit bacteria such as diphtheria, Brucella bovis, Streptococcus pneumoniae, meningococcal, and hemolytic streptococcus.
Anticonvulsant and antiepileptic effects
Experimental results have shown that administering 130 mg/kg to 260 mg/kg orally to mice has an anticonvulsant effect, which gradually increases with the number of doses administered. The maximum anticonvulsant effect occurs about one week after administration. Injection administration can accelerate the production of its anticonvulsant effect. Intraperitoneal injection of 260 mg/kg can counteract electroconvulsive seizures, with an inhibition rate of 100%. When combined with other antiepileptic drugs, borax can quickly control epileptic seizures and status epilepticus.


Medicine
Trace amounts of boron are beneficial to the human body, such as preventing osteoporosis and promoting calcium metabolism. However, high intake can lead to toxicity, and continuous intake can accumulate in various organs of the body. Mild cases may cause decreased appetite and indigestion, while severe cases may include symptoms such as vomiting, diarrhea, red spots, circulatory system disorders, shock, and coma. The adult toxic dose of borax is 1-3 grams, the adult lethal dose is 15-20 grams, and the infant lethal dose is 2-5 grams. According toChina's Food Safety Law and the first batch of non edible substances and food additives that may be illegally added to food and are prone to abuse announced by the Ministry of Health in December 2008, the addition of boric acid or borax to food is strictly prohibited.
Other functions
Borax also has anti-corrosion and protective effects on the skin and mucous membranes. Clinically, this product can be used to flush ulcers, abscesses, especially mucosal inflammations such as conjunctivitis and gastritis. As it is alkaline, it can remove mucosal scaling; Oral administration is used for urethral sterilization, especially when urine is acidic, it can make it alkaline.


Glass and enamel industry
In glass, it can enhance the transmittance of ultraviolet rays, improve the transparency and heat resistance of the glass. In enamel products, it can make the glaze less likely to peel off and give it a glossy appearance. It is also widely used in special optical glass, glass fiber, welding agents for non-ferrous metals, bonding agents for jewelry, printing and dyeing, washing (silk and woolen fabrics, etc.), gold refining, cosmetics, pesticides, fertilizers, borax soap, preservatives, antifreeze agents, and medical disinfectants.
Industrial sector
Borax is also used as a solid lubricant in metal drawing industry. In the welding and maintenance of refrigeration equipment such as refrigerators, electric freezers, and air conditioners, it is often used as a (non active) flux to remove oxides from metal surfaces; Add a certain proportion of sodium chloride to borax. Compounds such as sodium fluoride and potassium chloride can be used as active fluxes for welding copper and steel pipes, as well as between steel pipes in refrigeration equipment.


Other applications
Sodium tetraborate decahydrate (borax) has a wide range of applications in industrial and agricultural production. For example, used as fillers and to enhance surface activity (cross-linking) in laundry detergent and soap; It is also widely used in fiberglass, welding non-ferrous metals, bonding jewelry, cosmetics anti-corrosion, pesticides, fertilizers, antifreeze agents, and medical disinfectants. Used as a herbicide in agriculture and for eliminating weeds in non cultivated areas.

Many methods in our laboratory prepare borax:
(1) the pretreated boron ore powder is mixed with sodium carbonate solution by the carbon alkali method, and the reaction is carried out in the carbon decompressor. The amount of sodium carbonate added is 105% ~ 110% of the theoretical amount. The carbon decompressor is jacketed for heating. The reaction pressure is controlled at 0.5 ~ 0.6 MPa, the temperature is 130 ~ 135 ℃, the reaction time is 13 ~ 15 h, and the carbon dioxide concentration is 25% ~ 30%. The purified kiln gas can be directly used. After carbon hydrolysis, the slurry is filtered to remove the residue, washed in countercurrent, the obtained clear solution is concentrated (if the concentration of the countercurrent washing solution meets the requirements, it is not necessary to concentrate), cooled, crystallized, centrifuged and dried to obtain the finished borax product. The reaction equation is:
![]()
(2) Pressurized alkaline hydrolysis: the pretreated boron ore powder is mixed with sodium hydroxide solution (the amount of sodium hydroxide is 160% ~ 200% of the theoretical amount). It is decomposed by heating and pressurizing in an alkaline hydrolysis device equipped with a stirrer. The reaction pressure is 0.4 MPa, and the reaction time is 6 ~ 8 h. The alkali hydrolyzed slurry is filtered by a blade vacuum filter, washed by countercurrent, and then carbonized by carbon dioxide. The carbonation solution is cooled, crystallized, separated, and dried to obtain the finished borax product. The reaction equation is:
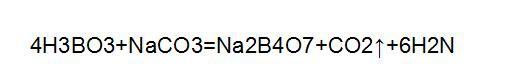
![]()
(3) Generally, industrial borax is used as raw material, heated and dissolved in high-purity water. After complete dissolution, vacuum suction filtration is maintained to remove insoluble impurities. The filtrate is sent to the crystallization tank for cooling and crystallization. When the crystallization solution is cooled to near room temperature, it is centrifuged, washed, and dried to obtain the finished product.
(4) The pure boric acid aqueous solution is reacted with stoichiometric refined sodium carbonate, and the obtained crystalline crude product is refined by synthesizing sodium tetraborate decahydrate. Pentahydrate can be prepared when the relative density of the aqueous solution is 1.19 ~ 1.24 or when the saturated aqueous solution at 80 ℃ is slowly crystallized at 65 ~ 70 ℃. When the hydrate is heated above 400 ℃, anhydrous matter can be prepared. First, it is dried in a steam bath, heated to about 200 ℃ in an air bath, and heated to 700 ℃ in an electric furnace.
(5)Dissolve 120g anhydrous sodium carbonate in 1080ml water, add 100g boric acid, stir to dissolve, filter, concentrate the filtrate to a relative density of 1.16, and cool it. Adsorb and filter out crystals. Dissolve it in 300ml hot water and sift it in to a porcelain dish cooled with ice with filter paper. Keep stirring the solution with a glass rod to precipitate decahydrate crystalline powder. Filter by suction and wash the crystal with a small amount of cold water. Then it is dissolved in water and recrystallized. The obtained crystals are dried in air for 2 ~ 3 days. The product is decahydrate.

(5) Add boric acid to 10% sodium carbonate solution, stir to dissolve, and carry out the reaction: 1303-96-4 preparation filtration, heat and concentrate the filtrate to a density of 1.16, cool it, filter out the crystallization, wash it with a small amount of cold water, and then recrystallize it. The obtained crystallization is dried in the air, and the product is sodium sodium tetraborate decahydrate (borax).
(6) To prepare an anhydrous substance, decahydrate can be dried in a steam bath, heated to 200 ℃, dehydrated and melted in an electric furnace at 700 ℃, and cooled to obtain solid glassy anhydrous sodium tetraborate.
(7) The raw material for industrial production of anhydrous borax is sodium tetraborate decahydrate. Please put it in the melting furnace for decompression melting dehydration, and the dehydration temperature is above 750 ℃. Molten borax flows out from the bottom of the melting furnace and is crushed to the required particle size after cooling.
(8) Boric acid refining and recrystallization method: industrial boric acid is dissolved in distilled water, then purified, filtered, crystallized, separated, washed, and dried.
Remark: BLOOM TECH(Since 2008), ACHIEVE CHEM-TECH is our subsidiary.
FAQ
What is sodium tetraborate decahydrate used for?
Borax Formula - Structure, Properties, Uses, Sample Questions ...Sodium tetraborate decahydrate (borax) is a versatile mineral compound used primarily as a household cleaner, laundry booster, water softener, and insecticide. It acts as a pH buffer, flux in ceramics/glass, and a preservative. Industrial uses include flux for welding/soldering and corrosion inhibition in coolants.
Is sodium tetraborate decahydrate toxic to dogs?
Sodium tetraborate decahydrate (borax) is mildly to moderately toxic to dogs, causing gastrointestinal distress and potential kidney/liver damage in high doses. Symptoms include vomiting, diarrhea, drooling, and tremors, often appearing within 30 minutes to 2 hours of ingestion. Immediate veterinary consultation is advised if a dog consumes this substance.
What is the common name for sodium tetraborate decahydrate?
Sodium tetraborate decahydrate, commonly known as borax, is a versatile compound with a wide range of applications across various industries.
Is sodium tetraborate decahydrate harmful to humans?
Humans. Humans breathing sodium tetraborate decahydrate while employed at a borax processing plant have reported temporary respiratory irritation from exposures equal to or above 4.5 mg borax/m3, but no chronic respiratory effects were found.
Hot Tags: sodium tetraborate decahydrate (borax) cas 1303-96-4, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale





