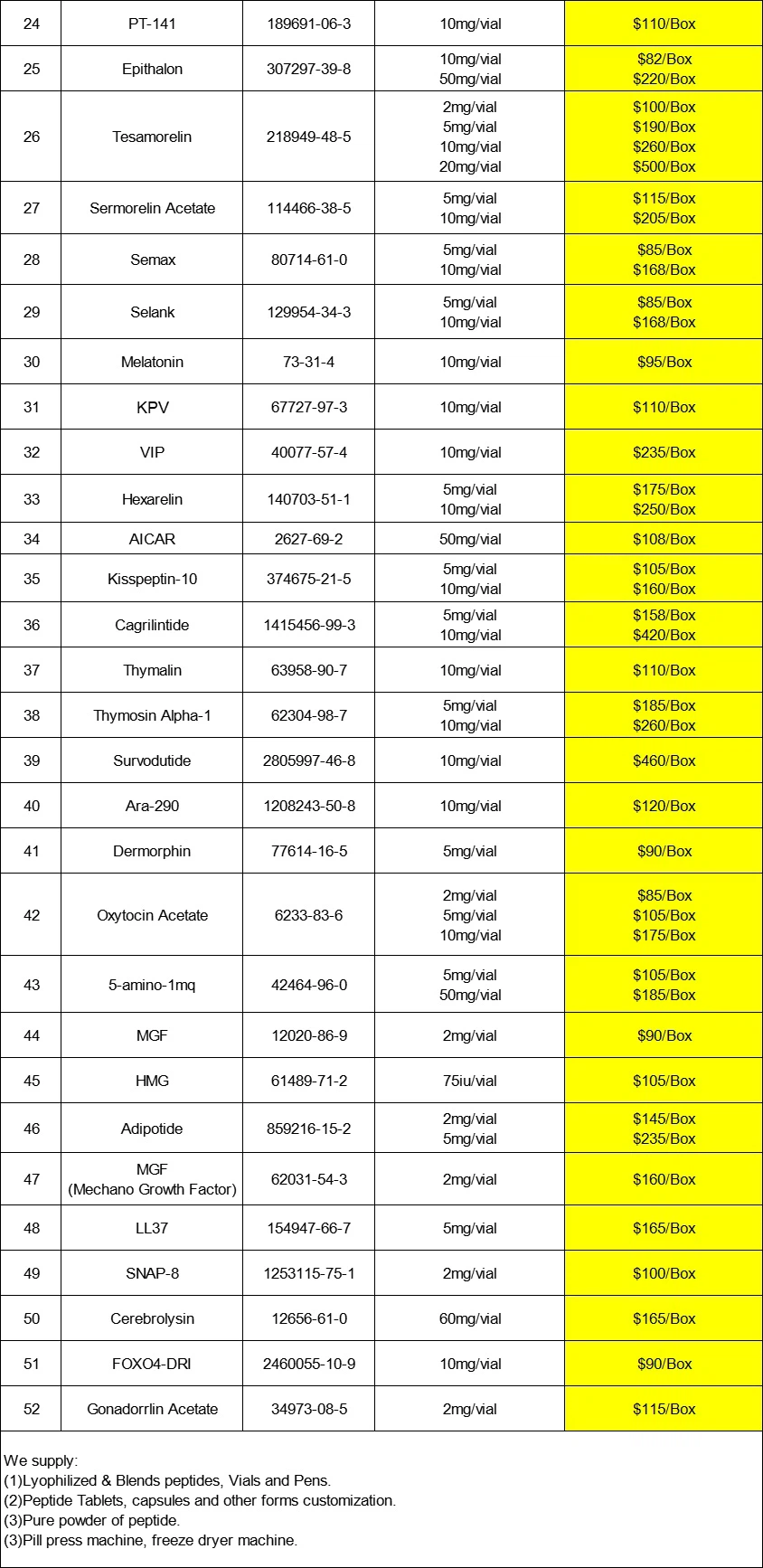
Cagralintide peptide is a novel long-acting acylated amygdalin analogue under development, typically with a purity of ≥ 95% or 98%. It needs to be sealed and stored in the dark at -20 ° C to ensure its stability and activity. It is a cyclic polypeptide composed of 38 amino acids, with a sequence containing 38 amino acids and a pair of disulfide bonds. As a non selective agonist of amylin receptor (AMYR) and calcitonin G protein coupled receptor (CTR), Cagrlintide has multiple biological activities. Cagralintide can significantly reduce weight, which makes it highly promising in obesity research. By activating relevant receptors, Cagrlintide can also reduce food intake, thereby aiding in weight management. At present, Cagralintide is still in the research and development stage and has not yet been widely used in clinical practice. But some studies have shown that it has significant effects in weight loss and reducing food intake. Due to its unique biological activity and potential therapeutic value, Cagrlintide may become an important drug for treating obesity and related metabolic diseases in the future. At the same time, currently available Cagrlintide products on the market are usually for scientific research purposes only and cannot be used for human treatment.
Customized Bottle Caps & Corks
|
|
|



Cagralintide peptide is a novel long-acting acylated amygdalin analogue under development, and is also a peptide hormone. Its CAS number is 1415456-99-3, and its molecular formula is relatively complex, containing multiple amino acid residues and a pair of disulfide bonds. The following is a detailed explanation of the use of Cagralintide:
Cagralintide has shown significant potential in the treatment of obesity. As a non selective agonist of amylin receptor (AMYR) and calcitonin G protein coupled receptor (CTR), it can activate these receptors, triggering a series of physiological reactions, significantly reducing food intake and weight loss. In clinical trials, Cagralintide has shown good weight loss effects. For example, in a phase II clinical trial targeting obese patients, weekly injection of Cagralintide reduced average body weight by 6% to 10.8% after 26 weeks, with weight loss effects comparable or superior to some marketed GLP-1 receptor agonists.

Treatment of type 2 diabetes

In addition to treating obesity, Caglininte is also used to treat type 2 diabetes. It can lower blood sugar levels and help patients better control their blood sugar. This mechanism of action may be related to its activation of amygdalin receptors and calcitonin receptors, whose activation can affect insulin secretion and sensitivity, thereby improving blood glucose levels. In addition, when combined with certain other hypoglycemic drugs such as semaglutide, Cagralintide may exhibit more
significant hypoglycemic effects.
The mechanism of action of Cagrlintide
The mechanism of action of Cagrlintide is mainly based on its properties as an analog of amygdalin. Starch is a hormone secreted by pancreatic beta cells, which has the effect of reducing gastric emptying rate and producing satiety, thereby reducing food intake. However, due to its poor water solubility and tendency to aggregate into amyloid like proteins, the clinical application of amygdalin is limited. Cagralintide, as an analog of amygdalin, has been chemically modified to enhance its water solubility and stability, enabling it to exert therapeutic effects more effectively. Specifically, Cagrlintide triggers a series of intracellular signal transduction processes by activating the amygdalin receptor and calcitonin receptor. These processes include suppressing appetite, reducing food intake, increasing energy expenditure, etc., which helps to reduce weight and improve blood sugar levels. In addition, Cagralintide may also exert therapeutic effects by affecting fat metabolism, insulin resistance, and other aspects.


As a new long-acting acylated amylin analogue under research, Cagralintide peptide shows a broad prospect in the medical field, especially in the treatment of obesity and type 2 diabetes. The following is a detailed analysis of the development prospects of Cagralintide:
Prospects in obesity treatment
Significant clinical trial results
Cagralintide has shown significant weight loss effects in multiple clinical trials. For example, in the phase 3 clinical trial of the fixed dose combination therapy of CagriSema composed of Semaglutide, overweight or obese patients who did not suffer from type 2 diabetes (T2D) lost an average weight of 22.7% after 68 weeks of treatment with CagriSema. This data fully demonstrates the effectiveness of Cagralintide in the treatment of obesity.
The market demand is huge
With the continuous increase of the global obese population, obesity has become a serious public health problem. Therefore, the demand for safe and effective weight loss drugs in the market is increasing day by day. Cagralintide, as a new type of weight loss drug, has enormous market demand potential.
The potential of combination therapy
Cagralintide can not only be used alone, but also in combination with other drugs to enhance weight loss effects. For example, the CagriSema therapy, used in combination with GLP-1 receptor agonist semaglutide, has shown breakthrough weight loss effects in clinical trials. This combination therapy provides more treatment options for obese patients.
Prospects in the treatment of type 2 diabetes
Improve blood sugar control
Cagralintide can regulate insulin secretion and blood glucose levels by activating the amygdalin receptor and calcitonin G protein coupled receptor. In clinical trials, Caglininte has shown the effect of improving blood glucose control in patients with type 2 diabetes. This provides a new treatment option for patients with type 2 diabetes.
Reduce cardiovascular risk
Type 2 diabetes patients are often accompanied by cardiovascular complications. Caglinintide can reduce cardiovascular risk while treating type 2 diabetes. For example, in the clinical trial of CagriSema, participants not only experienced significant weight loss, but also effectively reduced cardiovascular risk. This characteristic makes Caglininide have a broader application prospect in the treatment of type 2 diabetes.
Synergistic effects with other drugs
Caglininide can cooperate with existing type 2 diabetes drugs (such as insulin, oral hypoglycemic drugs, etc.) to improve the therapeutic effect. This synergistic effect helps to better control blood sugar levels and reduce the occurrence of complications.
Market competition and commercialization prospects
Market competition pattern
At present, the global market for weight loss and type 2 diabetes treatment has presented a fierce competition pattern. However, as a novel and effective therapeutic drug, Cagrlintide's unique mechanism of action and significant clinical efficacy give it a unique competitive advantage in the market.
Wide commercial prospects
With the continuous advancement of clinical trials and the publication of positive results, the commercialization prospects of Cagrlintide are becoming increasingly broad. It is expected that in the next few years, Cagilintide will become one of the important drugs in the field of weight loss and type 2 diabetes treatment.
Patent protection and market exclusivity period
Once Cagralintide is approved by relevant regulatory agencies and marketed for sale, it will be protected by patents. This will provide Cagrlintide with a certain exclusivity period in the market, ensuring its market share and profitability.
Research and Development Progress and Future Trends of Cagrlintide
Rapid progress in research and development
In recent years, the research and development of Cagrlintide has made rapid progress. Several pharmaceutical companies are actively advancing their clinical trial processes and planning to bring them to market in the coming years. In addition, with the in-depth study of the mechanism of action of Cagralintide, people have gained a more comprehensive understanding of its therapeutic efficacy and safety.
Future Trends: Diversification
In the future, the application areas of Cagrlintide may be further expanded. In addition to weight loss and treatment of type 2 diabetes, Cagilintide may also play an important role in other metabolic diseases, cardiovascular diseases and other fields. In addition, with the continuous advancement and innovation of pharmaceutical technology, the dosage form and administration method of Cagrlintide may also be further optimized and improved.
Challenges and coping strategies faced by Cagrlintide
Although Cagralintide has shown significant efficacy in clinical trials, it still requires strict approval from relevant regulatory agencies before it can be marketed for sale. This may require a considerable amount of time and effort, and there is a certain degree of uncertainty involved. Response strategy: Pharmaceutical companies should strengthen communication and cooperation with regulatory agencies to ensure the smooth development and registration of Cagrlintide. At the same time, actively prepare and submit necessary application materials and data to accelerate the approval process.
With the continuous increase of similar drugs in the market, Cagralintide will face fierce competition from competitors. This may affect its market share and profitability. Response strategy: Pharmaceutical companies should continue to pay attention to market dynamics and the situation of competitors, and adjust their market strategies and marketing methods in a timely manner. At the same time, we will strengthen brand building and market promotion efforts to enhance the market awareness and competitiveness of Cagrlintide.
Although Cagralintide has shown good safety in clinical trials, there may still be some potential safety risks in practical applications. This may affect the patient's medication experience and treatment effectiveness. Response strategy: Pharmaceutical companies should strengthen safety monitoring and risk assessment of Cagrlintide. Timely identify and address potential safety issues to ensure the safety and effectiveness of medication for patients. At the same time, strengthen communication and education with doctors and patients, and enhance their awareness of medication and safety.
Potential social and economic impacts of Cagrlintide
Improve patients' quality of life
As a new and effective therapeutic drug, Caglininide can significantly improve the symptoms and quality of life of obese and type 2 diabetes patients. This will help alleviate the pain and burden of patients, and improve their life satisfaction and happiness.
Promote the development of the medical industry
The development and promotion of Cagrlintide will drive innovation and growth in the healthcare industry. It will provide doctors with more treatment options and methods, which will help improve medical standards and treatment effectiveness. At the same time, the successful listing of Cagrlintide will also drive the development of related industrial chains, such as pharmaceutical equipment and raw material supply.
Driving economic growth
As a therapeutic drug with broad market prospects, Cagrlintide's launch and sales will drive economic growth in related industries. This will bring considerable economic benefits and employment opportunities to pharmaceutical companies, medical institutions, and others. At the same time, the successful promotion of Cagralintide peptide will also promote exchanges and cooperation in domestic and international markets, and advance the internationalization process of the pharmaceutical industry.
Frequently Asked Questions
What does cagrilintide peptide do?
+
-
Once administered, Cagrilintide binds to the amylin receptors in the brain, which helps modulate appetite and enhance feelings of fullness or satiety. By slowing gastric emptying, it prolongs the duration food stays in the stomach, contributing to reduced caloric intake.
What is the difference between semaglutide and cagrilintide?
+
-
CagriSema leverages synergistic mechanisms: Semaglutide slows gastric emptying, suppresses glucagon, stimulates insulin, and reduces appetite, while Cagrilintide enhances satiety and further delays gastric emptying via brainstem pathways.
Is cagrilintide approved by the FDA?
+
-
This combination therapy is highly investigational. Neither retatrutide nor cagrilintide have received FDA approval, and no clinical trials have studied this specific combination together.
Does cagrilintide slow gastric emptying?
+
-
The addition of cagrilintide, Blaha explained, further slows gastric emptying and induces satiety, creating a kind of "semaglutide squared effect."
How effective is cagrilintide for weight loss?
+
-
The results demonstrated that cagrilintide displayed clinically meaningful weight loss, with an average body weight reduction of 11.8% compared with 2.3% with placebo after 68 weeks, if all patients adhered to treatment.
Hot Tags: cagrilintide peptide cas 1415456-99-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, propylene oxide market, 2 bromo 8 chlorodibenzo b d thiophene, Picene, Procaine Hydrochloride, CAS 1353573 89 3, CAS 1955546 91 4










