Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of 3,4-difluorophenylboronic acid cas 168267-41-2 in China. Welcome to wholesale bulk high quality 3,4-difluorophenylboronic acid cas 168267-41-2 for sale here from our factory. Good service and reasonable price are available.
3,4-Difluorophenylboronic acid is a compound with important chemical properties. A white to light yellow crystalline solid. Its chemical formula is C6H5BF2O2, CAS 168267-41-2, and its relative molecular weight is 157.91 grams/mole. The melting point is approximately 158-160 degrees Celsius. This refers to the temperature range at which a compound transitions from solid to liquid. The solubility in water is relatively low, approximately 0.1-1 g/100 ml. Used as a reagent in chemical analysis. It can be used to determine or detect target compounds such as alcohols, ketones, and halogenated hydrocarbons. These analytical methods can play an important role in environmental monitoring, food safety, and quality control in the pharmaceutical industry. It has a wide range of applications in chemical analysis. It can be used for the analysis and detection of various target compounds such as alcohols, ketones, halogenated hydrocarbons, fatty acids, sugars, amino acids, and biomarkers. By utilizing the reactivity and selectivity of product, combined with appropriate analytical techniques, quantitative and qualitative analysis of these compounds can be achieved.

|
|
|
|
C.F |
C6H5BF2O2 |
|
E.M |
158 |
|
M.W |
158 |
|
m/z |
158 (100.0%), 157 (24.8%), 159 (6.5%), 158 (1.6%) |
|
E.A |
C, 45.64; H, 3.19; B, 6.85; F, 24.06; O, 20.26 |

3,4-Difluorophenylboronic acid has a wide range of applications in chemical analysis. As an important reagent, it can be used for the determination or detection of various target compounds.
1. Analysis of alcohols and ketones: It can form stable esterification products with alcohols and ketones. This property is widely used in analytical methods such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS). By reacting with alcohols or ketones, derivative compounds that are easy to detect can be formed, thereby achieving quantitative and qualitative analysis of the target compound.


2. Analysis of halogenated hydrocarbons: It can Coupling reaction to form stable esterification products. This property can be used for the analysis and detection of halogenated hydrocarbons. By reacting the tested sample with product, easily detectable derivatives such as 3,4-Difluorophenyl ester can be obtained for analysis using chromatographic techniques such as GC-MS and LC-MS.
3. Analysis of fatty acids: Fatty acids are important metabolic products in organisms, and accurate measurement of them is of great significance. It can Coupling reaction with Fatty acid methyl ester to form detectable derivatives. This method is usually used for qualitative and quantitative analysis of fatty acids. For example, using gas chromatography-mass spectrometry combined with the derivation reaction of It can achieve detailed analysis of fatty acid composition.


4. Analysis of sugars: Sugar plays an important role in fields such as food, biology, and medicine. It can react with sugars to form highly stable esterification products. This is widely used for the analysis and detection of sugars. By utilizing the selective reaction of product, qualitative and quantitative analysis of different types of sugars (such as glucose, fructose, lactose, etc.) can be achieved.
5. Analysis of amino acids: Amino acids are the basic building blocks of proteins in living organisms. It can form stable esterification products with amino acids, which can be used for the analysis of amino acids. This method can be separated and detected through chromatographic techniques such as high-performance liquid chromatography and HPLC.


6. Analysis of biomarkers: Biomarkers refer to chemical substances present in organisms that are related to health conditions, disease diagnosis, or treatment reactions. It can serve as a derivative reagent for specific biomarkers and play an important role in their analysis.

The laboratory synthesis method of 3,4-Difluorophenylboronic acid mainly includes the following steps: first, synthesize the intermediate of product, and then hydrolyze the intermediate to obtain the final product.
Step 1: Obtaining 3,4-Difluoroobenzene:
The synthesis of It requires obtaining 3,4-Difluorobenzene as the starting material. The common synthesis method is to prepare 3,4-Difluorobenzene through fluorination reaction.
Chemical reaction formula:
C6H5F2+Mg → MgF2+C6H4F2
Step 2: Aromatization reaction of 3,4-Difluorobenzene
In this step, 3,4-Difluorobenzene reacts with arylation reagent (such as Phenyllithium or bromobenzene) to generate corresponding aryl bromide.
Chemical reaction formula:
C6H4F2+Br2 → C6H3F2Br+HBr
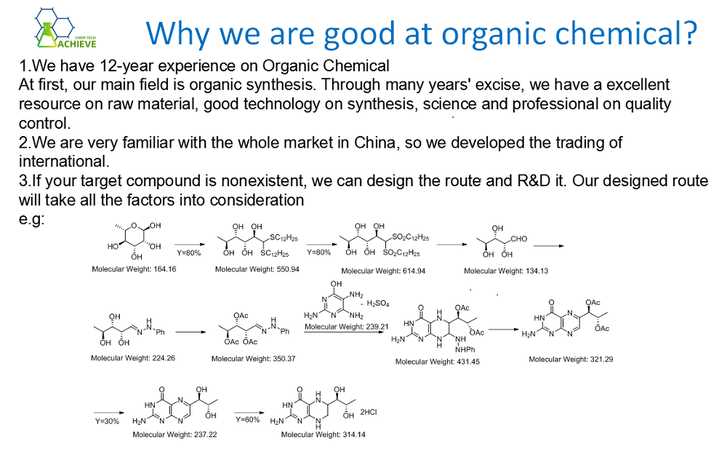
Step 3: Reaction of bromide with Triphenylphosphine sodium
In this step, aryl bromide reacts with Triphenylphosphine sodium to form a biarylphosphine reagent.
Chemical reaction formula:
C6H3F2Br+NaPPh3 → C6H3F2P (Ph) 3+NaBr
Step 4: Borination reaction of bisarylphosphine reagents
In this step, the diarylphosphine reagent reacts with boric acid to generate an intermediate of product.
Chemical reaction formula:
C6H3F2P (Ph) 3+B (OH) 3 → C6H3F2B (OH) 2P (Ph) 3
Step 5: Hydrolysis reaction of intermediate
The final step is to hydrolyze the intermediate to obtain the final product.
Chemical reaction formula:
C6H3F2B (OH) 2P (Ph) 3+H2O → C6H3F2B (OH) 2OH+P (Ph) 3
Through the above synthesis steps, 3,4-Difluorophenylboronic acid can be obtained. It should be noted that when conducting synthesis in the laboratory, strict adherence to chemical experimental operating standards should be followed, necessary safety measures should be taken, and appropriate experimental equipment should be used.
Application of Electronic Energy Level Regulation in Organic Semiconductors
Organic semiconductors have broad application prospects in optoelectronic devices such as organic light-emitting diodes (OLEDs), organic solar cells (OSCs), and organic field-effect transistors (OFETs) due to their advantages of light weight, good mechanical flexibility, and low processing costs. However, the optoelectronic properties and device performance of organic semiconductors are highly dependent on their frontier molecular orbital energy levels (highest occupied molecular orbital HOMO and lowest unoccupied molecular orbital LUMO) and excited state energy levels (first singlet state S1 and first triplet state T1). In traditional organic semiconductor materials, these energy levels are often interwoven with each other, making it difficult to achieve independent control and limiting further improvement in device performance. 3,4-Difluorophenylboronic acid (DFPB), as a type of fluorine-containing boronic acid compound, provides a new approach for regulating the energy levels of organic semiconductors through unique electronic and spatial effects of the fluorine atoms and boronic acid groups in its molecular structure.
The mechanism of action in electronic energy level regulation
HOMO and LUMO energy levels are key parameters that determine the electron injection and transport performance of organic semiconductor materials. DFPB can effectively regulate the positions of HOMO and LUMO energy levels through the fluorine atoms and boronic acid groups in its molecular structure.


The influence of fluorine atoms: The strong electronegativity of fluorine atoms reduces the electron cloud density on the benzene ring, leading to a decrease in the HOMO energy level. Meanwhile, since the LUMO energy level is mainly determined by the π * orbitals of the benzene ring, the electron attraction effect of fluorine atoms has a relatively small impact on the LUMO energy level.
Therefore, the introduction of DFPB can increase the HOMO-LUMO energy gap, enhance the material's electron affinity and ionization potential, thereby optimizing the electron injection and transport performance.


Conjugate effect of boronic acid group: The boron atom in the boronic acid group forms a π - p conjugated system with the π electron on the benzene ring, which can stabilize the LUMO energy level and further regulate the HOMO-LUMO energy gap. In addition, boronic acid groups can indirectly regulate the energy level structure by affecting the stacking mode of molecules through intermolecular interactions such as hydrogen bonds, coordination bonds, etc.
The excited state energy levels (S1 and T1) are key parameters that determine the luminescence performance and charge separation efficiency of organic semiconductor materials. DFPB, through its fluorine atoms and boronic acid groups in its molecular structure, can effectively regulate the position and energy gap of S1 and T1 energy levels (Δ E-ST=E-S1-E-T1).
The influence of fluorine atoms: The electron attraction effect of fluorine atoms can reduce the π electron density of the benzene ring, thereby affecting the positions of the S1 and T1 energy levels.


Research has shown that the introduction of fluorine atoms can lower the S1 energy level, while having a relatively small impact on the T1 energy level, thereby reducing Δ E-ST. A smaller Δ E-ST is beneficial for promoting the conversion of triplet excitons to singlet excitons, improving the luminescence efficiency and charge separation efficiency of the material.
Conjugate effect of boronic acid group: The boron atom in the boronic acid group forms a π - p conjugated system with the π electron on the benzene ring, which can stabilize the S1 and T1 energy levels and further regulate Δ E-ST. In addition, boronic acid groups can indirectly regulate the excited state energy level structure by influencing the molecular stacking mode through intermolecular interactions.
In traditional organic semiconductor materials, the HOMO, LUMO, S1, and T1 energy levels are often intertwined, making it difficult to achieve independent control. DFPB provides the possibility for independent regulation of energy levels through its unique molecular structure.
Selective regulation of fluorine atoms: The electron attraction effect of fluorine atoms mainly affects the HOMO and S1 energy levels, with little effect on the LUMO and T1 energy levels. Therefore, by adjusting the number and position of fluorine atoms, selective control of HOMO and S1 energy levels can be achieved.

Conjugate regulation of boronic acid groups: The boron atom in the boronic acid group forms a π - p conjugated system with the π electron on the benzene ring, which can stabilize the LUMO and T1 energy levels, thereby achieving regulation of the LUMO and T1 energy levels. By adjusting the structure and position of boronic acid groups, the energy level regulation effect can be further optimized.
Synergistic regulation of intermolecular interactions: Hydrogen bonding, coordination bonding, and other interactions between DFPB molecules can affect the stacking mode of molecules, thereby indirectly regulating the energy level structure. By designing intermolecular interactions, synergistic regulation of energy levels can be achieved, further improving the precision of regulation.

Application examples in organic semiconductors
OLED is a light-emitting device based on organic semiconductor materials, which has advantages such as self emission, high contrast, and wide viewing angle. DFPB can significantly improve the luminescence efficiency and stability of OLED by regulating the frontier molecular orbital energy levels and excited state energy levels of OLED materials.
Luminescent layer material: Introducing DFPB into OLED luminescent layer material can lower the HOMO energy level, increase the HOMO-LUMO energy gap, thereby improving the material's electron affinity and ionization potential, optimizing electron injection and transport performance.


Meanwhile, the introduction of DFPB can also reduce Δ E-ST, promote the conversion of triplet excitons to singlet excitons, and improve the luminescence efficiency of the material. For example, OLED devices based on DFPB derivatives have an external quantum efficiency (EQE) of over 20%, significantly higher than traditional OLED devices.
Hole transport layer material: DFPB can also serve as a hole transport layer material, achieving energy level matching with the luminescent layer material by regulating its HOMO energy level, thereby improving hole injection efficiency. Research has shown that hole transport layer materials based on DFPB can significantly reduce the driving voltage of OLED devices, improve device stability and lifespan.
OSC is an organic optoelectronic device that converts solar energy into electrical energy, with advantages such as lightweight, flexibility, and large-area fabrication. DFPB can significantly improve the photoelectric conversion efficiency of OSC by regulating the frontier molecular orbital energy levels and excited state energy levels of OSC materials.
Donor material: Introducing DFPB into OSC donor material can lower the HOMO energy level, increase the HOMO-LUMO energy gap, and thus improve the open circuit voltage (V_oc) of the material.


At the same time, the introduction of DFPB can optimize the absorption spectrum of the material and improve the utilization efficiency of sunlight. For example, OSC devices based on DFPB derivatives can achieve a V_oc of over 0.9 V and a photoelectric conversion efficiency (PCE) of over 10%.
Receptor material: DFPB can also serve as a receptor material, achieving energy level matching with the donor material by regulating its LUMO energy level, thereby improving charge separation efficiency. Research has shown that receptor materials based on DFPB can significantly improve the short-circuit current (Jsc) and fill factor (FF) of OSC devices, further enhancing PCE.
OFET is a field-effect transistor based on organic semiconductor materials, which has advantages such as low power consumption and high integration. DFPB can significantly improve the device performance of OFET by regulating the frontier molecular orbital energy levels and carrier mobility of OFET materials.
Semiconductor layer material: Introducing DFPB into OFET semiconductor layer material can lower the HOMO energy level, increase the HOMO-LUMO energy gap, and thus improve the air stability of the material.


At the same time, the introduction of DFPB can optimize the molecular stacking mode of the material and improve the carrier mobility. For example, OFET devices based on DFPB derivatives have a hole mobility of over 1 cm ²/(V · s), significantly higher than traditional OFET devices.
Interface modification material: DFPB can also be used as an interface modification material to achieve energy level matching with electrode materials by regulating its molecular structure and chemical properties, thereby improving carrier injection efficiency. Research has shown that interface modification layers based on DFPB can significantly reduce the contact resistance of OFET devices, improve the switching ratio and stability of the devices.
Hot Tags: 3,4-difluorophenylboronic acid cas 168267-41-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Consumable, Cerium sulfate powder, Iridium III chloride, CHICAGO SKY BLUE 6B, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID






