Pure samarium is a metal element with the chemical symbol Sm and atomic number 62. This is a silver white metal of medium hardness, which is easy to oxidize in the air. As a typical component of the lanthanide series, samarium usually assumes the oxidation state of+3. SmO, SmS, SmSe and SmTe are the most common compounds of samarium (II). Samarium has no significant biological effect, only slight toxicity. It coexists with other rare earth elements in monazite sand. The rare earth elements contained in monazite, as well as calcium and thorium, are distributed in the river sands of India and Brazil and the coastal river sands of Florida. The mass fraction of rare earth elements in monazite sand is usually 50%, of which samarium accounts for 2.8%. In addition, samarium also exists in bastnaesite, which is mostly distributed in southern California. Ion exchange technology is needed to separate samarium from its minerals.

|
Chemical Formula |
Sm |
|
Exact Mass |
150 |
|
Molecular Weight |
152 |
|
m/z |
152 (100.0%), 154 (85.0%), 147 (56.0%), 149 (51.7%), 148 (42.0%), 150 (27.6%), 144 (11.5%) |
|
Elemental Analysis |
Sm, 100.00 |


Pure samarium, as an important member of the rare earth element family, has demonstrated irreplaceable application value in multiple fields due to its unique physical and chemical properties.
Samarium cobalt permanent magnet (SmCo) is a pioneer in rare earth permanent magnet materials, with magnetic properties second only to neodymium iron boron. However, it has significant advantages in high temperature stability, demagnetization resistance, and corrosion resistance.
Technical characteristics: Samarium cobalt magnets are divided into two categories: SmCo ₅ series and Sm ₂ Co ₁ series. The latter has become mainstream due to its higher magnetic energy product (up to 32MGOe) and coercivity (>25kOe). Its maximum working temperature reaches 350 ℃, and some models can withstand extreme environments of 538 ℃. The magnetic change rate is less than 0.03%/℃, ensuring the reliability of precision systems under temperature differences.


Military applications:
F-35 fighter jet: Each aircraft requires 23 kilograms of samarium cobalt magnets to drive radar servo systems and high-temperature motors (capable of withstanding 538 ℃), ensuring precise control under extreme working conditions. Neodymium iron boron magnets cannot be replaced due to high temperature magnetic attenuation issues.
Missile guidance system: The samarium cobalt magnet drives the cone motor of the warhead, maintaining magnetic stability in the high-temperature friction environment generated by supersonic flight. Its anti electromagnetic interference characteristics ensure the reliability of laser/infrared guidance signals.
Nuclear submarine: The sonar detection system of the Virginia class nuclear submarine relies on samarium cobalt magnets to capture weak sound waves, and the propulsion motor uses samarium magnets to achieve silent operation, avoiding exposure of magnetic characteristics.
Industrial applications: In fields such as satellite navigation, high-frequency tubes, microwave equipment, etc., samarium cobalt magnets provide stable magnetic fields to ensure system accuracy. For example, the positioning instruments of the Apollo 11 spacecraft used samarium cobalt magnets to adapt to the extremely low temperature environment of the moon.

Nuclear Industry: Control Valve for Safe Utilization of Nuclear Fission

The isotope samarium-149 (Sm-149) of samarium has an extremely high thermal neutron capture cross section (42000 bar) and is a key control material for nuclear reactors.
Neutron absorption: Sm-149 regulates the rate of nuclear reactions by absorbing neutrons, preventing uncontrolled chain reactions. Its absorption capacity far exceeds traditional materials such as cadmium, and its performance is stable at high temperatures.
Structural material: Samarium alloy can be used to manufacture reactor shielding layers, effectively blocking gamma rays and neutron radiation, protecting personnel and the environment.
Strategic significance: After China imposed export controls on rare earths such as samariu, the US F-35 production line stalled due to a lack of samariu cobalt magnets, and the upgrade of nuclear submarines was delayed, exposing the vulnerability of the Western military supply chain. The Pentagon's 500 ton rare earth reserve is only sufficient for short-term emergencies, highlighting the strategic value of samarium in the field of nuclear energy safety.
The radioactive isotope samarium-153 (Sm-153) of samarium plays a crucial role in medical imaging and cancer treatment.
Lai Xijue Nan Samarium Injection: Used to treat pain caused by osteogenic bone metastasis, its emitted beta particles can target and destroy cancer cells while reducing damage to normal tissues. Clinical statistics show that the pain relief rate of the drug for bone metastasis of breast cancer, lung cancer and prostate cancer is more than 80%.
Medical imaging: Sm-153 serves as a tracer to locate tumor metastases through bone scanning, helping doctors develop precise treatment plans.
Technical advantage: Pure samarium compounds can withstand high temperatures above 700 ℃ without losing magnetism, ensuring the stability of drugs during preparation and storage.

Optics and Electronic Materials: "Additives" for Functional Upgradestions

Samarium compounds promote technological innovation in the fields of optics and electronics by improving material properties.
Laser material: samariu doped yttrium aluminum garnet (Sm: YAG) crystal is the core component of solid-state lasers, and its emitted 1.06 μ m laser wavelength is suitable for medical, industrial processing, and military directed energy weapons. The doping of samariu can improve laser efficiency by more than 30%.
Optical glass: Adding samarium oxide (Sm ₂ O3) can increase the refractive index (above 1.8) and wear resistance of the glass, while endowing the glass with special yellow fluorescence properties, which are used to manufacture high-precision optical instruments such as microscopes and telescopes.
Piezoelectric ceramics: Samariu oxide as an additive can improve the sintering and density of ceramics, producing suitable piezoelectric effects, and is widely used in fields such as sensors and ultrasonic transducers.
Samariu compounds exhibit efficient catalytic performance in chemical reactions and ceramic manufacturing.
Petroleum refining: Samariu based catalysts can promote heavy oil cracking, increase gasoline yield by 10% -15%, and reduce sulfur oxide emissions.
Hydrogen energy storage: Lanthanum nickel alloy (containing samarium) can absorb a large amount of hydrogen gas to form metal hydrides, which is expected to solve the problem of safe storage and transportation of hydrogen energy.
Ceramic capacitors: Doping with samarium oxide can increase the dielectric constant of ceramics, reduce dielectric losses, and is suitable for high-frequency circuits and pulse power systems.

Emerging fields: the forefront of technological breakthroughs

With the development of technology, the potential applications of samarium in stealth materials, superconducting technology, and other fields are gradually emerging.
Stealth material: Samariu based metamaterials can regulate radar waves and infrared radiation, achieving radar infrared dual band stealth for fighter jets/ships, breaking through the bandwidth limitations of traditional absorbing materials.
Superconducting technology: Certain compounds of samarium exhibit superconductivity at low temperatures, providing a material foundation for maglev trains and quantum computing.
Hypersonic missile: Samariu oxide, as a high-temperature resistant ceramic additive, can protect the thermal protection layer of the missile from damage at a flight speed of 5 Mach.
Samariu, with its unique physical and chemical properties, has become a "bridge" connecting traditional industry with cutting-edge technology. From high-temperature permanent magnets to nuclear reactor control rods, from cancer pain treatment drugs to laser crystals, the application of samarium runs through multiple dimensions of human society. With China's strategic control and technological upgrading of rare earth resources, the global supply chain pattern of Samariu will continue to evolve, and its dominant position in key areas will be further consolidated. In the future, with breakthroughs in emerging fields such as stealth materials and superconducting technology, the potential value of samarium will be more fully unleashed.


For the preparation of pure samarium, metal samarium can be prepared by reducing samarium oxide with barium or lanthanum.
Reduction distillation method of Samariu oxide: The advantage of reduction distillation method is that rare earth oxides are directly used as raw materials, and the reduction and distillation processes are carried out simultaneously, thus simplifying the process. The purity of the obtained metal products is high. In addition, the residue from reduction distillation is also a rare earth oxide, which can be recycled.
Because samarium has a high vapor pressure, while the vapor pressure of reductant lanthanum is low. La: at 1754 ℃, the vapor pressure is 1.33 Pa; at 2217 ℃, the vapor pressure is 133.32 PaSm; at 722 ℃, the vapor pressure is 1.33 Pa; at 964 ℃, the vapor pressure is 133.32 Pa; therefore, the lanthanum reduction distillation method of oxides can be used to prepare metal samarium: 2La (l)+Sm2O3 (s) 1600La2O3 (s)+2Sm (g). The Samariu produced in the reaction can be removed from the reactor by volatilization, which can promote the complete reaction.
In the reduction distillation process, heat samarium oxide in air at 800 ℃ for 15h to remove the possible absorption of H2O and CO2. Lathe the molten metal lanthanum at 1800 ℃ into metal chips. Mix 550g of calcined Sm2O3 and 540g of La metal chips [excess 15% (mass fraction)], and pass through the ingot pressing (9.8-49) × 107Pa] put it into a Ta crucible with a diameter of 6.4cm and a length of 25.4cm, and attach a 20cm Ta condenser and a Ta baffle on the upper part of the crucible to prevent excessive oxide particles from being taken out. Put the device into the high temperature zone of the vacuum induction furnace. When the system is evacuated to a pressure less than 0.1Pa, it starts to heat up. After 2h, it rises to the maximum temperature of 1600 ℃, and remains at this temperature for another 2h. It is important to raise the temperature slowly, because if the temperature rises too fast, La will melt and run to the bottom of the crucible, affecting the contact of reactants. The reduced metal is distilled out of the reaction zone and condensed on the condenser. About 465g of samariu can be obtained with a yield of 98%. When the temperature of the condenser is 300~500 ℃, the condensed metal has large crystalline particles and is stable in the air. However, when the condensation temperature is low, the condensed metal particles are fine and flammable in the air. The purity of the product from one reduction distillation can reach 99.5% or more, but it still contains hundreds of La, O and H in the order of 10-6. These impurities can be further reduced after re distillation or sublimation. The sublimation temperature is 800 ℃, and the condensation temperature is~500 ℃. The crucible used for reduction distillation can be used in sublimation. However, the crucible should be pickled with acid in advance and degassed under vacuum at 1800 ℃.
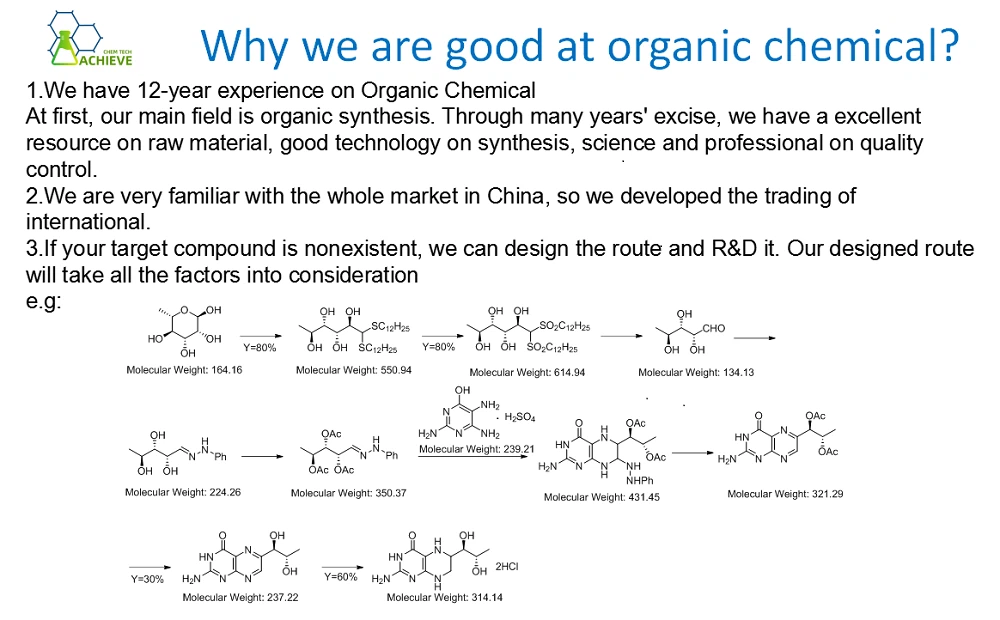
![]()
Discovery process:pure samarium is one of lanthanide elements (belonging to rare earth elements), which entangled and confused chemists in the 19th century. Its history began with the discovery of cerium in 1803.
Cerium is supposed to contain other metals. Carl Mosander claimed to obtain lanthanum and didymium from it in 1839, but didymium is actually a mixture of praseodymium and neodymium. In 1879, Paul ¦ mile Lecoq de Boisbaudran again extracted the didymium from niobium yttrium ores. Later, he made the solution of nitric acid didymium and added ammonium hydroxide. It was found that the sediment was formed in two stages. He concentrated on the first deposit and measured its spectrum, and concluded that it was a new element samarium. (In fact, europium was found in samarium in 1901)
Mineral distribution: coexists with other rare earth elements in monazite sand. The rare earth elements contained in monazite, as well as calcium and thorium, are distributed in the river sands of India and Brazil and the coastal river sands of Florida. The mass fraction of rare earth elements in monazite sand is usually 50%, of which samarium accounts for 2.8%. In addition, samarium also exists in bastnaesite, which is mostly distributed in southern California. Ion exchange technology is needed to separate samarium from its minerals.
Frequently Asked Questions
What are the hazards of samarium?
+
-
Specific Hazards Arising from the Material: Flammable in the form of dust when exposed to heat, spark or flame. May react with water under fire conditions liberating flammable hydrogen gas. May emit fumes of samarium oxide under fire conditions.
Is samarium used in headphones?
+
-
Samarium has its main use in creating alloys with cobalt for headphones and small motors. Radioactive Samarium 153 is used in the treatment of cancers.
What is samarium used for in the military?
+
-
Samarium is central to Samarium-Cobalt (SmCo) magnets, prized for their thermal stability and resistance to demagnetisation. These magnets are used in radar seekers, gyroscopes, and inertial navigation systems, ensuring accurate targeting and trajectory control under extreme conditions.
Hot Tags: pure samarium cas 7440-19-9, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CHICAGO SKY BLUE 6B, Iridium III chloride, Consumable, Cerium sulfate powder, 1 AMINO 2 NAPHTHOL 4 SULFONIC ACID


