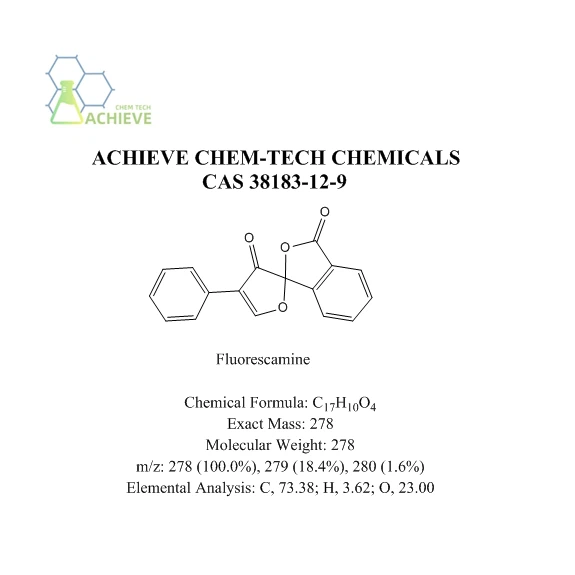
Fluorescamine, chemical formula C17H10O4, molecular weight 278.26, CAS number 38183-12-9, is an important chemical reagent widely used in the fields of biochemistry and analytical chemistry. It usually exists in the form of a white to off white powder, and sometimes may also appear pale yellow. Its powdered form makes it easy to operate and weigh in the laboratory. The solubility in water is low, appearing slightly soluble. However, it can dissolve well in various organic solvents such as acetonitrile, acetone, ethanol, chloroform, and dimethyl sulfoxide. This solubility characteristic allows fluorescent amines to be prepared and reacted in the laboratory by selecting appropriate solvents. It does not have fluorescence properties itself, but it can react with compounds containing primary amino groups (such as proteins, peptides, amino acids, etc.) to generate compounds with strong fluorescence. This characteristic makes it an important fluorescent derivative reagent. Can react with primary amino groups of amino acids under mild conditions to form stable, high fluorescence, low background complexes. This complex has high sensitivity in fluorescence detection and is therefore widely used for quantitative analysis and detection of amino acids. In protein analysis, it can also react with the primary amino groups of proteins to generate fluorescent derivatives. By detecting the fluorescence intensity of these derivatives, quantitative analysis of protein content can be achieved.

|
|
|
|
Chemical Formula |
C17H10O4 |
|
Exact Mass |
278 |
|
Molecular Weight |
278 |
|
m/z |
278 (100.0%), 279 (18.4%), 280 (1.6%) |
|
Elemental Analysis |
C, 73.38; H, 3.62; O, 23.00 |

Fluorescamine is an important chemical reagent with wide applications in the fields of biochemistry and analytical chemistry.
1. Fluorescence detection and analysis
Amino acid detection: It can react with the primary amino group of amino acids under mild conditions to form stable, high fluorescence, and low background complexes. This complex has high sensitivity in fluorescence detection and is commonly used for quantitative analysis of amino acids.
Protein analysis: It can also react with the primary amino groups of proteins to generate fluorescent derivatives, which are used for quantitative detection and structural research of proteins. By detecting the fluorescence intensity of these derivatives, the protein content and structural changes can be evaluated.


2. Liquid chromatography fluorescence detection
High performance liquid chromatography (HPLC) combined with fluorescence detection is a commonly used technique in biochemical analysis. As a fluorescent derivative reagent, it can enhance the fluorescence signal of the target compound, improve the sensitivity and selectivity of detection. This technology is widely used in fields such as proteomics and drug metabolism.
3. Protein sequencing and structural research
In protein sequencing and structural research, it can be used to label specific positions or residues on proteins, helping researchers determine the sequence and structure of proteins. This labeling method helps to understand the function and interactions of proteins.


4. Detection of proteolytic enzymes
Can react with substrates of proteolytic enzymes to generate fluorescent products. By monitoring the fluorescence intensity changes of these products, the activity and kinetic characteristics of proteolytic enzymes can be evaluated. This is of great significance for studying the catalytic mechanism of enzymes and screening inhibitors.
1. Drug screening
In the process of drug development, it can be used to screen potential drug targets. By binding and reacting with the target, fluorescent amines can reveal the interaction mechanism between drugs and targets, providing important information for drug design and optimization.
2. Drug metabolism research
It can also be used for drug metabolism research. By labeling specific functional groups on drugs or their metabolites, fluorescent amines can help researchers track the metabolic pathways and rates of drugs in the body. This is crucial for evaluating the safety and efficacy of drugs.

Other applications

1. Environmental monitoring
Although its direct application in environmental monitoring is limited, its characteristics as a fluorescent derivative reagent provide a new approach for environmental monitoring. By combining with other detection technologies, it is possible to play an important role in environmental monitoring, such as detecting pollutants in water bodies.
2. Teaching and Research Tools
In the fields of teaching and research, it is one of the indispensable tools. By demonstrating the reaction process and results of fluorescent amines with different compounds, students and researchers can better understand the basic principles and methods of biochemistry and analytical chemistry.

The synthesis of Fluorescamine amines usually involves raw materials such as benzoic acid, ethylenediamine, chloroform or other solvents, catalysts (such as acids or bases). In some cases, oxidants or reducing agents may also be used, but these are less common in the direct synthesis of fluorescent amines.
Synthesis steps and chemical equations
Firstly, benzoic acid can undergo amidation reaction with ethylenediamine to produce N-phenylethylenediamine amide (here we assume a simplified intermediate product, as the actual intermediate product may be more complex). This step usually needs to be carried out under acidic or alkaline conditions to promote the reaction between carboxyl and amino groups.
Chemical equation (assuming the intermediate product is N-phenylethylenediamine amide):
Benzoic Acid + Ethylenediamine → N-Phenylethylenediamide + Water
Note: This equation is a schematic representation, and different amide products may be generated in actual reactions, requiring catalysts (such as acids or bases) and appropriate reaction conditions (such as temperature and solvents).
The next steps may involve cyclization reactions, which involve converting a part of the amide structure into a cyclic structure. However, the cyclization reaction directly converting N-phenylethylenediamine amide to fluorescent amine does not exist because the structure of fluorescent amine is more complex, including the benzoxazolone structure. Therefore, this step requires the introduction of other functional groups or further structural modifications.
Assumed cyclization reaction (for illustration only, not actual pathway):
N-Phenylethylenediamide → Intermediate Cyclic Compound
Here, 'Intermediate Cyclic Compound' represents a fictional intermediate cyclic compound that requires further reaction to be converted into a fluorescent amine.
In order to convert cyclic intermediates into fluorescent amines, oxidation, halogenation, condensation, or other types of functional group conversion reactions may be required. The specific types and conditions of these reactions depend on the structure of the intermediate and the structure of the target product fluorescent amine.
Due to the complex and varied synthesis pathways of fluorescent amines, an exact chemical equation cannot be provided here. But generally speaking, these reactions may involve the use of oxidants (such as hydrogen peroxide, potassium permanganate, etc.) to introduce oxygen atoms, or the use of halogenating agents (such as chlorine gas, sodium bromide, etc.) to introduce halogen atoms, and further reactions to form the characteristic structure of fluorescent amines.
Regardless of the synthesis pathway used, the reaction mixture ultimately needs to be purified to obtain high-purity fluorescent amines. Purification methods may include filtration, washing, drying, crystallization, recrystallization, chromatographic separation, etc. These steps aim to remove impurities and unreacted raw materials to improve the purity and yield of the Fluorescamine.

The principle of fluorescent amine detection for amino groups is a chemical reaction based fluorescent detection technology, which utilizes the specific reaction between fluorescent amines and amino groups (especially primary amine groups) to generate products with strong fluorescence, thereby achieving sensitive detection of amino groups.
types of CNC machining
Basic characteristics of fluorescent amines
Fluorescent amine is an organic compound that contains specific functional groups in its molecular structure. These functional groups can undergo chemical changes when reacting with amino groups, generating new fluorescent substances. Fluorescent amines themselves do not have significant fluorescence under normal conditions, but once they react with amino groups, their fluorescence properties undergo significant changes, allowing the generated products to emit strong fluorescence under specific wavelength light excitation.
mill CNC machine
Reaction mechanism between fluorescent amine and amino group
The reaction between fluorescent amine and amino group mainly occurs between the carbonyl group (C=O) of fluorescent amine and the nitrogen atom of amino group. Under appropriate reaction conditions (such as a certain pH value, temperature, etc.), the carbonyl group of fluorescent amine will undergo condensation reaction with the amino group, forming a Schiff base structure. This reaction process is usually accompanied by the breaking and formation of chemical bonds, as well as changes in electronic structure, resulting in significant changes in fluorescence properties.
Specifically, when fluorescent amine reacts with an amino group, the carbonyl oxygen atom in the fluorescent amine molecule attacks the nitrogen atom of the amino group, forming a new carbon nitrogen double bond (C=N). This reaction not only changes the electronic structure of fluorescent amine molecules, but also enables the generated products to emit strong fluorescence under excitation of ultraviolet or visible light. The intensity and wavelength of this fluorescent signal are usually related to the concentration of reactants, reaction conditions, and the structure of the product.
FAQ
What is the reaction of fluorescamine with amine?
Fluorescamine is defined as an amine-reactive dye that forms intensely fluorescent adducts upon reaction with protein primary amines, allowing for the staining of proteins prior to SDS tube gel electrophoresis. It exhibits an excitation maximum at approximately 390 nm and an emission maximum at approximately 475 nm.
Which amino acids are fluorescent?
The three naturally fluorescent amino acids are tryptophan (Trp), tyrosine (Tyr), and phenylalanine (Phe). While all three are fluorescent, tryptophan is the most widely used in biological applications because it has the strongest fluorescence properties, though its wavelengths are in the ultraviolet range.
What chemicals are fluorescent?
Xanthene derivatives: fluorescein, rhodamine, Oregon green, eosin, and Texas red.
Cyanine derivatives: cyanine, indocarbocyanine, oxacarbocyanine, thiacarbocyanine, and merocyanine.
Squaraine derivatives and ring-substituted squaraines, including Seta and Square dyes.
Squaraine rotaxane derivatives: See Tau dyes.
What does fluorescence detect?
Fluorometry is the technique of measuring any parameter using fluorescent emission. In the context of modern bioscience, fluorometry is used to detect biological macromolecules such as proteins and measure their concentration in the experimental sample.
Hot Tags: fluorescamine cas 38183-12-9, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Articaine Hcl powder, CAS 662903 77 5, 1r 3r spiro adamantane 2 9 fluoren 2 yl boronic acid, 2 bromo 8 chlorodibenzo b d thiophene, CAS 222044 85 1, 1H BenziMidazole 1 4 broMophenyl 2 phenyl