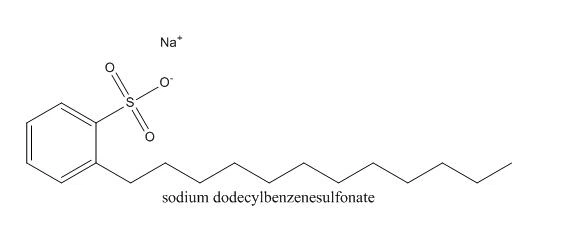
Sodium dodecylbenzene sulfonate, SDBS for short, is a commonly used anionic surfactant. It is a white or light yellow powdery or flaky solid, difficult to volatilize, easily soluble in water, and soluble in water to form a translucent solution. It is stable and slightly toxic to alkali, dilute acid and hard water.
The history of development is as follows: because petrochemical companies can convert a large amount of tetrapolypropylene into Dodecylbenzene, the product quality is high and the price is low, so the detergent with Dodecylbenzene as raw material quickly replaced soap, and dodecylbenzene sulfonate soon became the largest amount of organic surfactant in the United States. At this time, although the surfactant used has good application performance, there is a serious disadvantage, Their biodegradation rate in the sewage treatment plant is very low, and the degradation is incomplete, which has caused great pollution to the environment. In order to solve this problem, in the early 1960s, the detergent industry began to shift from the production of Branched Chain Alkylbenzene Sulfonates to linear chain alkylbenzene sulfonates. Due to the good biodegradability of the straight chain products, the pollution caused by detergent foam, which was the focus of the detergent industry in the 1950s, was solved. After that, the application field of alkyl arylsulfonate anionic surfactants has been expanding, and the demand and sales of products have been increasing.
Physical properties: 1 Anionic surfactant. Because of its low production cost and good performance, it is widely used. It is the synthetic surfactant with the largest amount of household detergent. It also produces some inorganic salts such as magnesium and calcium and organic amine salts such as triethanolamine. Calcium dodecylbenzene sulfonate [27176-87-0] has excellent emulsifying properties and is an important part of mixed emulsifiers for preparing various pesticides. Benzene and α- Olefins are condensed under the catalyst of aluminum trichloride, and the condensed liquid is evaporated to recover benzene after alkali washing and water washing, and then refined alkylbenzene is obtained by vacuum distillation. Then the calcium dodecylbenzene sulfonate was obtained by sulfonation with fuming sulfuric acid and neutralization with white ash (in 2 times the amount of ethanol).
2. it is widely used as detergent raw material to produce various detergents and emulsifiers, such as washing powder in daily chemical industry. Tableware, detergent, detergent and dye for textile industry, degreaser for electroplating industry, leather industry, deinking agent for paper industry, etc. The product is a high-efficiency additive to improve the quality of inorganic and organic chemical products that are easy to absorb moisture and agglomerate. Excellent degreaser for high-grade leather.
Synthetic sodium dodecylbenzene sulfonate - industrial process:
Sodium dodecylbenzene sulfonate is prepared by the reaction of Dodecylbenzene with fuming sulfuric acid or sulfur trioxide, and then neutralization with alkali. The disadvantage of sulfonation with fuming sulfuric acid is that there is always some waste acid in the sulfonated material after the reaction. The sodium sulfate generated after neutralization is brought into the product, which affects its purity. At present, sulfur trioxide air mixture sulfonation is used in industry. Sulfur trioxide can be evaporated from 60% fuming sulfuric acid, or sulfur and dry air can be burned in the furnace to obtain mixed gas containing SO3. The mixed gas is introduced into a sulfonation reactor equipped with alkylbenzene for sulfonation. The sulfonated materials enter the neutralization system for neutralization with sodium hydroxide solution, and finally enter the spray drying system for drying. The obtained product is a powder with good fluidity.
After dehydrogenation of straight run kerosene, dodecyl olefin and benzene enter the alkylator through the feed pump, and then the generated dodecyl benzene (Lab) is sent to the sulfonator 1, where it is sulfonated with sulfur trioxide (3%~5%) entering the sulfonator. After treatment by gas-liquid separator 2, circulating pump 3 and cooler 4, some of the products return to the bottom of the reactor for quenching of sulfonic acid, and some of the reaction products are sent to the aging reactor 5, Adjust the reaction holding time, then enter the hydration reactor 6 to form acid, and finally prepare sodium alkylbenzene sulfonate (LAS) through neutralizer 7. The tail gas is removed from acid mist by demister 8, and then vented after being absorbed by absorption tower 9.

