Sodium dodecylbenzene sulfonate, SDBS for short, is a white or light yellow powder, which is easily soluble in water and easy to absorb moisture and agglomerate. It is stable to alkali, dilute acid and hard water.
Discovery history of sodium dodecylbenzene sulfonate: in the late 1930s, people alkylated benzene with chlorinated petroleum, and then sulfonated the generated alkylbenzene to produce alkylbenzene sulfonate. This was the first batch of industrial products of alkyl aromatic sulfonates. At that time, most of the products were used in the textile industry, and then household formulas appeared soon. After the Second World War, dodecylbenzene sulfonate appeared, which is a by-product of oil catalytic cracking. Tetrapolypropylene is prepared by reacting benzene with tetrapolypropylene as alkylation reagent and sulfonation.
The chemical properties of sodium dodecylbenzene sulfonate are as follows:
1. Hydrolysis reaction is the inverse of sulfonation reaction. Under the catalysis of strong acid, sodium dodecyl sulfate can be co heated with water to remove the sulfonyl group. The essence of the reaction is the electrophilic substitution reaction of h+ attacking the aromatic ring as an electrophilic reagent.
3R12-Ph-SO3Na+H2O →R12-Ph+NaHSO4(~200℃,H2SO4)
2. It can establish a balance system with strong acid:
R12-Ph-SO3Na+HCl⇌R12-Ph-SO3H+NaCl
3. The hydroxyl group in the sulfonyl group can also be replaced by the chlorine atom to form sulfonyl chloride:
3R12-Ph-SO3Na+PCl3 →R12-Ph-SO3H+NaCl(~200℃)
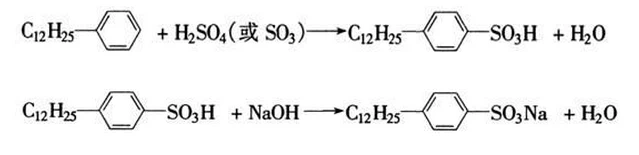
Synthetic sodium dodecylbenzene sulfonate -- Laboratory method
Add 35ml (34.6g) of Dodecylbenzene into a 250ml four mouth bottle equipped with a stirrer, thermometer, dropping funnel and reflux condenser, slowly add 35ml of 98% sulfuric acid under stirring at a temperature not exceeding 40 ℃, and then raise the temperature to 60 ~ 70 ℃ for 2h.
Lower the temperature of the above sulfonated mixture to 40 ~ 50 ℃, slowly add an appropriate amount of water (about 15ml), pour it into the separating funnel, stand still for a while, layer by layer, release the lower layer (water and inorganic salt), and retain the upper layer (organic phase).
Prepare 80ml of 10% sodium hydroxide solution, add it to about 60 ~ 70ml of 250ml four mouth bottle, slowly add the above organic phase under stirring, control the temperature to 40 ~ 50 ℃, adjust ph=7 ~ 8 with 10% sodium hydroxide, and record the total amount of 10% sodium hydroxide.
In the above reaction system, add a small amount of sodium chloride, filter after the ring penetration test is clear, and obtain a white paste product, namely sodium dodecylbenzene sulfonate.
The application scope of sodium dodecylbenzene sulfonate is also very wide. For example, in the washing industry, sodium dodecylbenzene sulfonate is a yellow oil, which can form hexagonal or inclined square strong flake crystals after purification. It has slight toxicity and has been recognized as a safe chemical raw material by the international safety organization. Sodium Alkylbenzene Sulfonate can be used in fruit and tableware cleaning. It is the largest amount used in detergents. Due to large-scale automatic production and low price, Sodium Alkylbenzene Sulfonate Used in detergents has two types: branched chain structure (ABS) and straight chain structure (LAS). The branched chain structure has little biodegradability and will cause pollution to the environment, while the straight chain structure is easy to be biodegradable. The biodegradability can be greater than 90% and the degree of environmental pollution is small.
Sodium dodecylbenzene sulfonate is neutral, sensitive to water hardness, not easy to oxidize, strong foaming power, high decontamination power, easy to mix with various additives, low cost, mature synthesis process and wide application fields. It is a very excellent anionic surfactant. Sodium dodecylbenzene sulfonate has a significant decontamination effect on particle dirt, protein dirt and oily dirt, especially on particle dirt on natural fibers. The decontamination power increases with the increase of washing temperature, and its effect on protein dirt is higher than that of non-ionic surfactants, with rich foam. However, sodium dodecylbenzene sulfonate has two disadvantages. One is that it has poor resistance to hard water, and its decontamination performance can decrease with the hardness of water. Therefore, the detergent with it as the main active agent must be used with an appropriate amount of chelating agent. Second, it has strong degreasing power. It is irritating to the skin when washing by hand. The hand feel of the washed clothes is poor. It is appropriate to use cationic surfactant as a softener for rinsing. In recent years, in order to obtain better comprehensive washing effect, sodium dodecylbenzene sulfonate is often used in combination with non-ionic surfactants such as fatty alcohol polyoxyethylene ether (AEO). Sodium dodecylbenzene sulfonate is mainly used to prepare various types of liquid, powder and granular detergents, detergents and detergents.

