Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of lixisenatide injection in China. Welcome to wholesale bulk high quality lixisenatide injection for sale here from our factory. Good service and reasonable price are available.
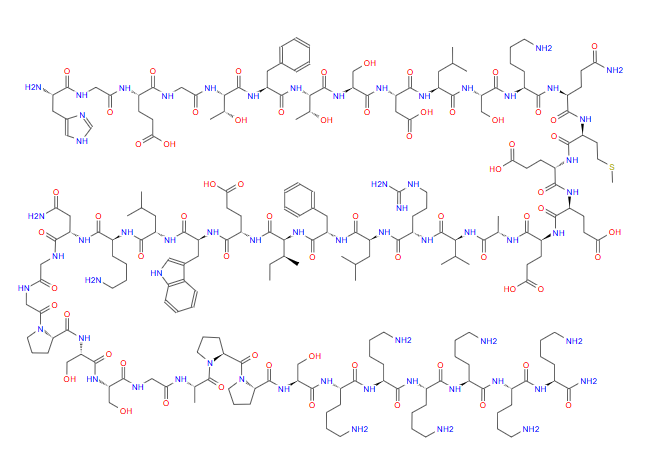
Lixisenatide injection is an innovative drug for the treatment of type 2 diabetes, belonging to glucagon like peptide-1 (GLP-1) receptor agonists. It simulates the natural secretion of GLP-1 hormone by the human body, activates corresponding receptors, and thus exerts various hypoglycemic effects.
Approved for listing in Europe in 2013 under the brand name Lyxumia ®, It can be used alone or in combination with oral hypoglycemic drugs and insulin to treat type 2 diabetes. Its unique pharmacokinetic characteristics and good safety make it one of the important choices for comprehensive management of diabetes.
Our Products Form





Lixisenatide COA
 |
||
| Certificate of Analysis | ||
| Compound name | Lixisenatide | |
| Grade | Pharmaceutical grade | |
| CAS No. | 320367-13-3 | |
| Quantity | 40g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090057 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.47% |
| Loss on drying | ≤1.0% | 0.35% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.90% |
| Single impurity | <0.8% | 0.56% |
| Total microbial count | ≤750cfu/g | 170 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 400ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||

In recent years, the widespread distribution of various organs throughout the body, especially the central nervous system, has attracted high attention from scientists. The research on lixisenatide injection has far exceeded the traditional field of endocrinology and entered the field of neurodegenerative diseases.
Theoretical basis of neuroprotective mechanisms

Research has shown that neurodegenerative diseases such as Parkinson's Disease (PD) and Alzheimer's Disease (AD) are closely related to insulin resistance, chronic neuroinflammation, oxidative stress, and mitochondrial dysfunction in the brain. It can penetrate the blood-brain barrier, activate GLP-1 receptors on neurons, and through the cAMP mediated signaling pathway:
Inhibit the excessive activation of microglia, thereby reducing the inflammatory cascade reaction in the central nervous system.
Upregulating the expression of anti apoptotic proteins (such as Bcl-2), inhibiting apoptosis, and directly protecting dopaminergic neurons from toxic damage.
Improve cellular energy metabolism and mitochondrial function.
Data source:
Nature Reviews Neuroscience, Mechanism of GLP-1 in Neurodegenerative Diseases, 2020;
MCE Scientific Product Data Platform, Lixisenatide Pharmacological Applications, 2023
Milestone clinical trial for Parkinson's disease (PD) (LIXIPARK)
In the field of Parkinson's disease treatment, a recent breakthrough research on this substance has caused a global shock in the medical community due to the lack of "disease modifying" drugs that can reverse or slow down the disease process.
In April 2024, the top international medical journal, the New England Journal of Medicine (NEJM), published the results of a multicenter, randomized, double-blind, placebo-controlled phase II clinical trial called LIXIPARK.
Experimental design: The study was led by a team from the French National Institute of Health and Medical Research (Inserm). The study included 156 patients diagnosed less than three years ago who were receiving standard treatment for early-stage Parkinson's disease. The patients were randomly assigned to receive subcutaneous injection of levocetirizine once a day or placebo for 12 months.


Surprising results: At the end of the 12-month treatment period, researchers evaluated patients using the MDS-UPDRS Part III (Motor Function Score). The results showed that patients in the placebo group had a 3.04 point deterioration in their motor symptom scores (consistent with the natural progression of the disease), while patients in the group treated with liraglutide not only did their motor scores not deteriorate, but also improved by 0.04 points.
Clinical significance: This result has epoch-making significance. For the first time in rigorous clinical trials, it has been demonstrated that the hypoglycemic drug, liraglutide, can substantially slow down the progression of motor symptoms in Parkinson's disease, indicating its true protective effect on dopaminergic neurons in the brain. Although about 46% of patients in the experimental group experienced nausea as a gastrointestinal side effect, the potential of this drug is immeasurable compared to the huge benefit of delaying neurodegenerative disease.

Data source: The New England Journal of Medicine, Clinical trial LIXIPARK on the treatment of early Parkinson's disease with Lisilar, April 2024
Other potential research directions

In addition to neurological disorders, basic research is also exploring the potential therapeutic value of liraglutide in rheumatoid arthritis (due to its anti-inflammatory properties) and non-alcoholic steatohepatitis (NASH)/metabolic associated fatty liver disease (MASLD). It has been shown to alleviate liver steatosis in animal models by reducing the synthesis of fatty acids in the liver and promoting the oxidation of free fatty acids.
Data source: Hepatology, Target Study of GLP-1 Analogs in NASH Treatment, 2022

Clinical efficacy and key clinical trials (GetGoal series studies)
Lixisenatide injection effectiveness and safety have been mainly confirmed through a massive global phase III clinical trial program - the GetGoal series of studies. The plan covered more than 5000 patients with type 2 diabetes, and evaluated its efficacy as a single drug, in combination with oral hypoglycemic drugs (such as metformin and sulfonylurea), and in combination with basic insulin.
Data source: Comprehensive analysis of diabetes, Obesity and Metabolism, GetGoal series tests, 2015
In the GetGoal Mono trial, for patients with poor control solely through diet and exercise, once daily treatment with liraglutide for 12 weeks resulted in a significant decrease of approximately 0.7% to 0.9% in glycated hemoglobin (HbA) levels compared to baseline, accompanied by weight loss. In the GetGoal-M trial, its addition to metformin background therapy demonstrated superior hypoglycemic and weight loss effects compared to placebo.
GetGoal-X is an open label non inferiority trial that compares once daily administration with twice daily administration of Exenatide. The research results showed that liraglutide was not inferior to exenatide in reducing HbA, and the liraglutide group had better gastrointestinal tolerance (especially lower incidence of nausea) and lower risk of hypoglycemia.
Data source: diabetes Care, GetGoal-X, 2012
Cardiovascular and Renal Safety Assessment (ELIXA Trial)
For all new hypoglycemic drugs, the US FDA requires a cardiovascular outcome test (CVOT) to demonstrate that the drug does not increase the risk of cardiovascular events. Its cardiovascular outcome trial is called ELIXA (Evaluation of Lixsenatide in Acute Coronary Syndrome).
Data source: The New England Journal of Medicine, ELIXA trial complete results, 2015
ELIXA is a landmark study, because it specifically recruited patients at high cardiovascular risk: 6068 patients with type 2 diabetes who had recently (within 180 days) developed acute coronary syndrome (ACS, including myocardial infarction or unstable angina pectoris).
Primary endpoint: Four components of major adverse cardiovascular events (MACE-4), including cardiovascular death, non fatal myocardial infarction, non fatal stroke, or hospitalization for unstable angina.
Research results: The median follow-up time was 25 months. The proportion of primary endpoint events occurring in this group was 13.4%, compared to 13.2% in the placebo group. The hazard ratio (HR) is 1.02 (95% confidence interval 0.89-1.17). This demonstrates the excellent cardiovascular safety of liraglutide in this extremely high-risk population (meeting non inferiority criteria), although it did not show the same cardiovascular benefits as some later long-acting GLP-1 (such as the LEADER trial of liraglutide) (i.e., efficacy was not demonstrated).
Further post hoc analysis of ELIXA data and related microvascular complication studies have found that it has potential renal protective effects. Data shows that liraglutide can effectively reduce the incidence of massive albuminuria and decrease the progression of urinary albumin/creatinine ratio (UACR). This renal benefit is independent of its hypoglycemic effect and may be related to its direct anti-inflammatory, antioxidant stress, and improved renal hemodynamics mechanisms.
Data source: CJASN, Clinical Journal of the American Society of Nephrology, Riseratide can prevent kidney damage in patients with type 2 diabetes, secondary analysis based on ELIXA data, 2017
In conclusion, as a short-term GLP-1 receptor agonist during meals, lixisenatide injection has proved its outstanding value in controlling postprandial blood glucose fluctuations, reducing weight, and maintaining high cardiovascular safety in patients with type 2 diabetes in its clinical application for more than 10 years. Especially its combination with insulin glargine, the compound formulation Soliqua, provides an efficient, highly compliant, and side effect hedging innovative treatment option for patients with poor monotherapy control.

The pharmacokinetic parameters determine the frequency of once daily administration.
Absorption: Rapid absorption after subcutaneous injection (usually in the abdomen, thighs, or upper arms). The time to reach the maximum plasma concentration is approximately between 1 and 3.5 hours after administration.
Distribution: The apparent volume of distribution in the body is approximately 100 liters. It mainly exists in free form in the blood, with a moderate plasma protein binding rate (about 55%).
Metabolism: As a peptide, lixisenatide injection is not metabolized by the cytochrome P450 (CYP) enzyme system in the liver. It is mainly filtered in the kidneys and extensively degraded by proteolytic enzymes into smaller peptide segments and monomeric amino acids.
Elimination&Half life: Degradation products are mainly excreted through the kidneys. Its terminal elimination half-life is relatively short, about 3 hours. This short half-life makes it particularly suitable for injection before the main meal of the day (usually breakfast or dinner) to cover postprandial blood sugar fluctuations.
Special populations: Patients with mild to moderate renal insufficiency typically do not require dose adjustment, but in patients with severe renal insufficiency (creatinine clearance rate below 30 mL/min) and end-stage kidney disease, the use of levocetirizine is not recommended due to decreased drug clearance rate.
Data source: EMA Lyxumia pharmacokinetic evaluation report, 2013
Data source: FDA Adlyxin prescription information, 2016
The following are the core data and factual sources cited during the writing process of this article:
1. World Health Organization (WHO): Registration information on Lixisenatide in the International List of Non Patent Medicines (INN).
2. European Medicines Agency (EMA): 2013 Public Evaluation Report (EPAR) - Lyxumia, covering the research and development history, pharmacokinetics, use in special populations, and reproductive toxicity data of Lysimab.
3. US Food and Drug Administration (FDA): 2016 Adlyxin drug instructions and prescription information, including usage and dosage, incidence of adverse reactions (such as nausea), contraindications, and post marketing safety warning data.
4. National Medical Products Administration (NMPA): Chinese drug instructions and clinical indications guidelines for Soliqua (insulin glargine injection).
5. PubChem: Compound Database (CID: 56841944/CAS: 320367-13-3) provides precise chemical formula, amino acid sequence, and relative molecular weight data for Liragliflozin.
6. Journal of Medicinal Chemistry: A study published in 2014 on the structural modification mechanism of GLP-1 analogs provided a chemical mechanism by which the poly-Lysine tail resists DPP-4 degradation.
7. Molecular Endocrinology: A literature published in 2012 that provides a detailed analysis of the glucose dependent insulin secretion pathway that causes an increase in cAMP levels and intracellular calcium release after GLP-1 binding to receptors.
8. The Lancet diabetes&Endocrinology: A comparative study published in 2015 confirmed the core pharmacodynamic difference between short-term and long-term GLP-1 RA in gastric emptying and postprandial glucose control.
FAQ
What is the drug lixisenatide used for?
Lixisenatide injection is used to treat type 2 diabetes mellitus. Lixisenatide is used together with diet and exercise to help control your blood sugar. This medicine is a glucagon-like peptide-1 (GLP-1) receptor agonist. This medicine is available only with your doctor's prescription.
Has lixisenatide been discontinued?
Lixisenatide (Lyxumia®) 10 microgram and lixisenatide treatment initiation pack (10 microgram and 20 microgram) have been discontinued. This means no new patients can be initiated on lixisenatide.
Hot Tags: lixisenatide injection, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale