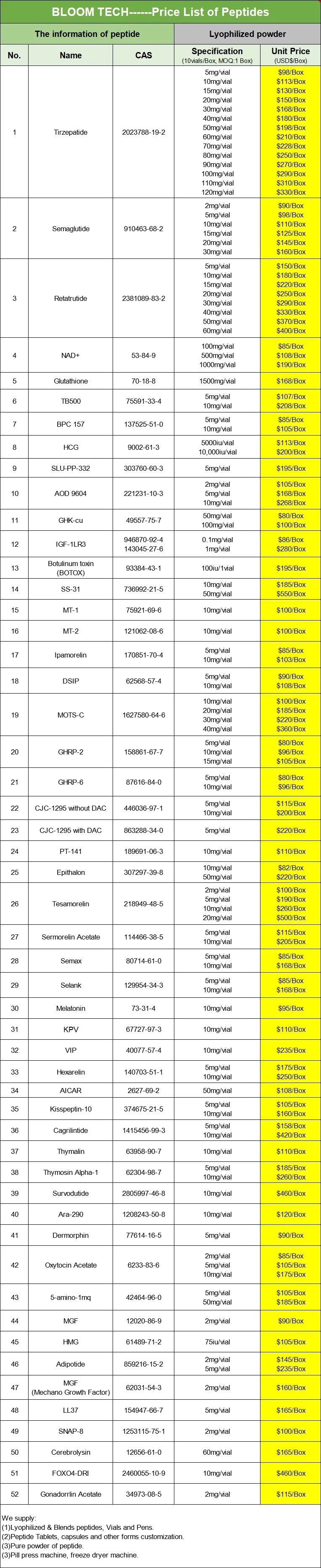
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of Bioglutide Tablets in China. Welcome to wholesale bulk high quality Bioglutide Tablets for sale here from our factory. Good service and reasonable price are available.
Bioglutide Tablets, an oral small-molecule quadruple receptor agonist, offers significant advantages in metabolic health while demonstrating unique potential for depression intervention. Through multi-pathway synergy, it modulates emotion-related physiological processes by directly protecting neurons, regulating neuroinflammation and stress responses, and improving brain energy metabolism. It also engages the gut-brain axis to influence gut health and signaling, while improving systemic metabolism to break the "metabolic dysfunction–inflammation–depression" cycle. Particularly suitable for depressed patients with comorbid obesity or glycemic instability, it provides dual benefits of neuroprotection and metabolic improvement, offering a novel approach to integrated depression management beyond single-mechanism interventions.
Our products





Bioglutide NA-931 COA

The significant pharmacological advantages of Bioglutide
1. Oral Small Molecule Design: Unlike GLP-1 receptor agonists (such as semaglutide and tirzepatide), oral administration avoids local irritation associated with tablets. Additionally, blood drug concentrations rise steadily without sharp post-injection peaks, which prevents concentrated gastrointestinal reactions.
2. Food Independence and Absorption Profile: Its oral absorption is not affected by food intake, eliminating the need for fasting during administration. This reduces gastrointestinal discomfort caused by interactions between diet and medication while improving convenience and tolerability.

3. Mild Gastrointestinal Regulation Effect: Compared to certain long-acting injectables that significantly delay gastric emptying (e.g., semaglutide can prolong gastric emptying for over 12 hours), Bioglutide tablets exerts a gentler gastrointestinal regulatory effect, reducing the incidence of symptoms such as bloating and nausea.

II. Clinical Evidence and Dosing Strategy
1. Phase II Clinical Data Support: In the 150 mg dose group, the incidence of nausea and vomiting over 13 weeks was 7.3%, and diarrhea was 6.3%. These rates are significantly lower than those of some injectable weight-loss drugs (e.g., gastrointestinal adverse event rates exceeding 80% in the semaglutide 2.4 mg dose group). Moreover, no severe side effects were reported, and discontinuation rates were low.
2. Low-Dose Initiation and Titration: The clinical strategy involves starting with a low dose and gradually adjusting it, allowing the body to adapt progressively to the drug's effects. This approach reduces the risk of gastrointestinal reactions and offers better tolerability compared to some injectables initiated at higher doses.
III.advantage
Incidence of Gastrointestinal Reactions: Mostly mild.
Severe Side Effects: No reports of severe side effects.
Formulation-Related Risks: Oral administration eliminates local injection reactions.
Metabolic Stress: IGF-1 pathway protects muscles and reduces metabolic stress.
Bioglutide's pharmaceutical research and innovation tool
Bioglutide tablets (NA-931) is not only an investigational therapeutic drug but also a critical tool and technology validation platform in the field of metabolic disease drug development.
I.Validation of Drug Delivery and Formulation Technology: A Benchmark for Oral Metabolic Drug Formulation Optimization
The oral formulation characteristics of it provide key technical validation for the development of oral small-molecule peptide/metabolic modulator formulations, including:
Technical Reference for Enhancing Oral Bioavailability: Small-molecule metabolic drugs often face challenges such as poor gastrointestinal absorption and significant first-pass effects. Through formulation optimization, It achieves stable oral bioavailability without relying on absorption enhancers (e.g., SNAC). Its formulation design, excipient selection, and gastrointestinal stability strategies can directly serve as a technical reference for improving the oral bioavailability of other small-molecule metabolic drugs, addressing a core challenge in oral administration.

Validation of Food-Independent Dosing Formulation Logic: Existing oral GLP-1 drugs (e.g., oral semaglutide tablets) require fasting for administration, limiting patient compliance. Bioglutide's oral absorption is unaffected by food intake. Its formulation design avoids interference from food on drug dissolution and absorption, validating the feasibility of "food-independent oral formulations." This provides formulation design insights for developing highly compliant oral metabolic drugs and optimizes the patient medication experience.

Technology Exploration Platform for Long-Acting Oral Formulations
It supports once-daily oral dosing. Its formulation technologies for controlling in vivo half-life and maintaining stable blood drug concentrations can be leveraged to explore the development of long-acting oral metabolic drug formulations (e.g., once-weekly oral dosage forms). This serves as a technology validation platform to overcome the industry bottleneck of long-acting injectables versus short-acting oral formulations.
II.Industrial and academic application value
Innovative Cross-Industry Commercial Collaboration Models
The oral formulation of Bioglutide enables integration with consumer-grade health management scenarios. Its combination therapies can facilitate collaborations with pharmaceutical companies and healthcare institutions to develop integrated "drug + metabolic management service" solutions. This drives the transformation of the pharmaceutical industry from "single drug sales" to "full-cycle disease management," expanding the commercial boundaries of metabolic drugs.
Core patents of major GLP-1 receptor agonists (such as semaglutide and liraglutide) will gradually expire, intensifying competition from generic drugs. The combination therapy patents for this with these drugs provide a compliant solution based on "low-dose originator drugs + innovative multi-target drugs".

This approach not only avoids price competition from generics but also achieves product differentiation through enhanced efficacy and safety, offering a practical commercialization pathway for both originator and innovative pharmaceutical companies after patent expiration.
Reference for Tiered Market Positioning in Metabolic Drug Commercialization
This drug is positioned as an oral, convenient weight-loss/metabolic drug, while its combination therapies target high-efficacy, intensive treatment regimens. This creates a tiered product portfolio ranging from "basic treatment to intensive therapy," catering to diverse patient needs (such as mild to moderate obesity, severe obesity, and patients with metabolic comorbidities). This model serves as a commercial reference for metabolic drug companies in building product matrices and implementing market segmentation strategies.
III.Core Experimental Carrier for Metabolic Biology Fundamental Research
Bioglutide tablets, as a unique multi-target tool molecule, serves as a critical resource in metabolic biology and pharmacological basic research. Its core academic value is reflected in the following areas:

Research Tool for Crosstalk Between Metabolic Pathways
Metabolic disorders involve dysregulation across multiple signaling pathways. It can precisely activate four core metabolic pathways, making it a valuable tool for studying the crosstalk between these pathways (e.g., the synergistic effects of GLP-1 and IGF-1 pathways on muscle metabolism, and the regulatory relationship between glucagon and GIP pathways in lipolysis). This helps elucidate the pathogenesis of diseases such as obesity, type 2 diabetes, and MASH, filling a gap in tools for studying multi-pathway synergy.
Research Model for Receptor Pharmacology and Signal Transduction
As a tool molecule, Bioglutide enables the study of combined activation of G protein-coupled receptors (GPCRs, including GLP-1, GIP, and glucagon receptors) and the IGF-1 receptor. It aids in analyzing the activation kinetics of different receptors, crosstalk among downstream signaling pathways, and the molecular basis of biased signaling. This provides an experimental model for receptor pharmacology and structural biology research, driving theoretical breakthroughs in GPCR family drug development.
With comprehensive data spanning from preclinical studies to Phase II clinical trials, it establishes a translational chain connecting "basic mechanism research to clinical efficacy validation." It can be used to analyze the relationship between target mechanisms and clinical endpoints, the correlation between pharmacokinetics and pharmacodynamics, and the screening of safety biomarkers. This provides robust data support for the clinical translation research of metabolic drugs, accelerating the translation of basic research findings into clinical applications.

The significant effect of Bioglutide on depression

Bidirectional Regulation Pathways of Emotion and Metabolism
Depression is often accompanied by disruptions in the brain-gut axis. this achieves bidirectional modulation through the following mechanisms:
1. Balance of Enteric Neurotransmitters
Promotes the secretion of 5-HT by intestinal L-cells, which enters the central nervous system via the bloodstream and vagus nerve pathways, increasing 5-HT levels in the raphe nuclei and exerting SSRI-like antidepressant effects.
Inhibits intestinal inflammation and reduces intestinal permeability, thereby decreasing LPS entry into the bloodstream, lowering systemic inflammatory load, and indirectly ameliorating central neuroinflammation.
2. Microbiota-Metabolite-Brain Signaling
Modulates the composition of the gut microbiota, increasing the abundance of beneficial bacteria such as *Akkermansia*, which enhances the production of short-chain fatty acids (SCFAs). SCFAs cross the blood-brain barrier to regulate neuroinflammation and neurotransmitter synthesis.Improves gastrointestinal motility and nutrient absorption, reducing psychological stress induced by gastrointestinal discomfort, thereby enhancing overall tolerability and emotional stability.
Integration of Metabolism Inflammation Neuroplasticity: The Overall Effect of Antidepressants
Improves Insulin Resistance and Blood Glucose Fluctuations:Reduces neuroinflammation and neuronal damage caused by abnormal central glucose metabolism, thereby restoring metabolic homeostasis in the hippocampus (e.g., levels of NAA and glutamate).
Modulates the Tryptophan-Kynurenine Pathway:Reduces the production of neurotoxic metabolites (such as quinolinic acid) and increases the availability of tryptophan, a precursor for 5-HT synthesis.

Reduces Stress-Induced Cortisol Elevation:Decreases glucocorticoid toxicity to hippocampal neurons, enhancing stress adaptability and resilience.
Regulates Sleep-Wake Cycles:Alleviates sleep disorders associated with depression and restores the stability of circadian rhythms.
fAQ
- Is this drug the same as semaglutide?
Do not confuse it with already marketed GLP-1 receptor agonists (such as semaglutide and tilpotide), as they have essential differences in composition, approval, and use.
- What are the side effects of Bioglutide?
Common side effects observed in clinical studies include muscle spasms, diarrhea, and acne.
- How to alleviate the side effects of it?
Choose light, low-fat, easily digestible foods, such as oats, steamed pumpkin, porridge, and cooked vegetables. Avoid spicy, fried, high sugar, and dairy products to reduce intestinal irritation. Adopting a small and frequent meal pattern (5-6 meals per day) to reduce gastrointestinal burden.
Hot Tags: Bioglutide Tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale






