Breast cancer remains one of the most prevalent and life-threatening malignancies among women worldwide. The development of effective treatment strategies has been a focal point of medical research, leading to the discovery and utilization of various therapeutic agents. Exemestane tablets, a member of the aromatase inhibitor class, have emerged as a crucial component in the management of hormone receptor-positive breast cancer, particularly in postmenopausal women. This article aims to provide a detailed and in-depth exploration of Isimetan tablets, covering their mechanism of action, pharmacokinetics, clinical applications, adverse effects, drug interactions, and future directions.
Products Information

Exemestane +. COA

Pharmacokinetics
● Absorption
Isimetan is administered orally in tablet form. After ingestion, it is rapidly absorbed from the gastrointestinal tract. Studies have shown that the bioavailability of Isimetan is relatively high, with at least 42% of the drug being absorbed into the systemic circulation. The absorption of Isimetan can be influenced by food intake. Taking Isimetan with a high-fat meal has been reported to increase the plasma levels of the drug by approximately 40%, likely due to enhanced solubility and absorption in the presence of dietary fats.
● Distribution
Once absorbed, Isimetan is widely distributed throughout the body. It has a high plasma protein binding rate of around 90%, which means that the majority of the drug in the bloodstream is bound to plasma proteins, primarily albumin. This binding affects the drug's distribution to different tissues and its overall pharmacokinetic profile. Isimetan can cross the blood-brain barrier to some extent, although the clinical significance of this is not fully understood.
● Metabolism
Isimetan undergoes extensive metabolism in the body, primarily through oxidation of the 6-methylene group and reduction of the 17-keto group. The metabolism of Isimetan is mainly mediated by the cytochrome P450 (CYP) enzyme system, particularly CYP3A4, as well as aldoketoreductase. However, it is important to note that Isimetan does not inhibit any of the major CYP isozymes, which reduces the likelihood of significant drug-drug interactions through CYP inhibition.
● Excretion
The metabolites of Isimetan are excreted from the body through both urine and feces, with approximately equal contributions from each route (around 40% each). The excretion of the unchanged drug in the urine is less than 1% of the administered dose, indicating that Isimetan is almost completely metabolized before excretion. The average terminal half-life of Isimetan is approximately 24 hours, which allows for once-daily dosing in clinical practice.
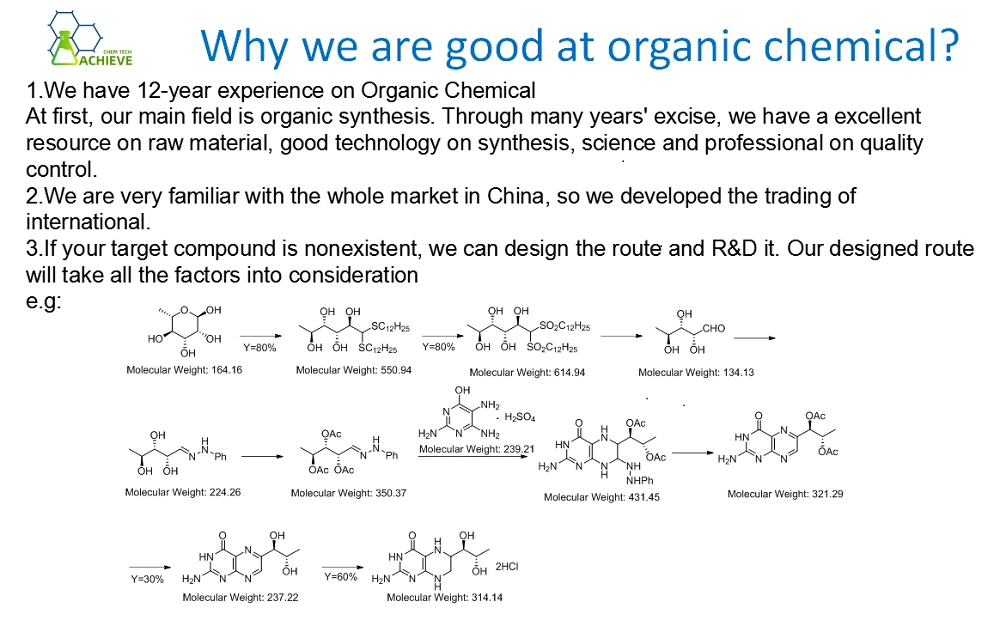
Clinical Applications

Indications
Isimetan tablets are primarily indicated for the treatment of postmenopausal women with hormone receptor-positive breast cancer. It is commonly used in two main clinical scenarios:
Adjuvant Therapy: Following initial treatment with surgery, radiation, and/or chemotherapy, Isimetan can be used as adjuvant therapy to reduce the risk of cancer recurrence in postmenopausal women with early-stage hormone receptor-positive breast cancer. It is often prescribed after a period of treatment with tamoxifen, another anti-estrogen drug, to further improve disease-free survival and overall survival rates.
Advanced or Metastatic Breast Cancer: Isimetan is also effective in the treatment of advanced or metastatic hormone receptor-positive breast cancer in postmenopausal women. It can be used as a first-line or subsequent therapy, depending on the patient's previous treatment history and response to other drugs.
Efficacy
Numerous clinical trials have demonstrated the efficacy of exemestane in the treatment of breast cancer. In the adjuvant setting, the Intergroup Exemestane Study (IES) showed that switching from tamoxifen to exemestane after 2 - 3 years of tamoxifen therapy significantly improved disease-free survival and overall survival compared to continuing tamoxifen for a total of 5 years. In the treatment of advanced breast cancer, exemestane has been shown to produce objective response rates and prolong progression-free survival in a significant proportion of patients.

Adverse Effects

Common Adverse Effects
Like all medications, Isimetan tablets can cause a range of adverse effects, although most are mild to moderate in severity. The most commonly reported adverse effects include:
Hot Flashes: This is one of the most frequent side effects, occurring in a significant proportion of patients. Hot flashes are characterized by sudden feelings of intense heat, often accompanied by sweating and flushing of the skin. They can be disruptive to daily life and may require symptomatic treatment.
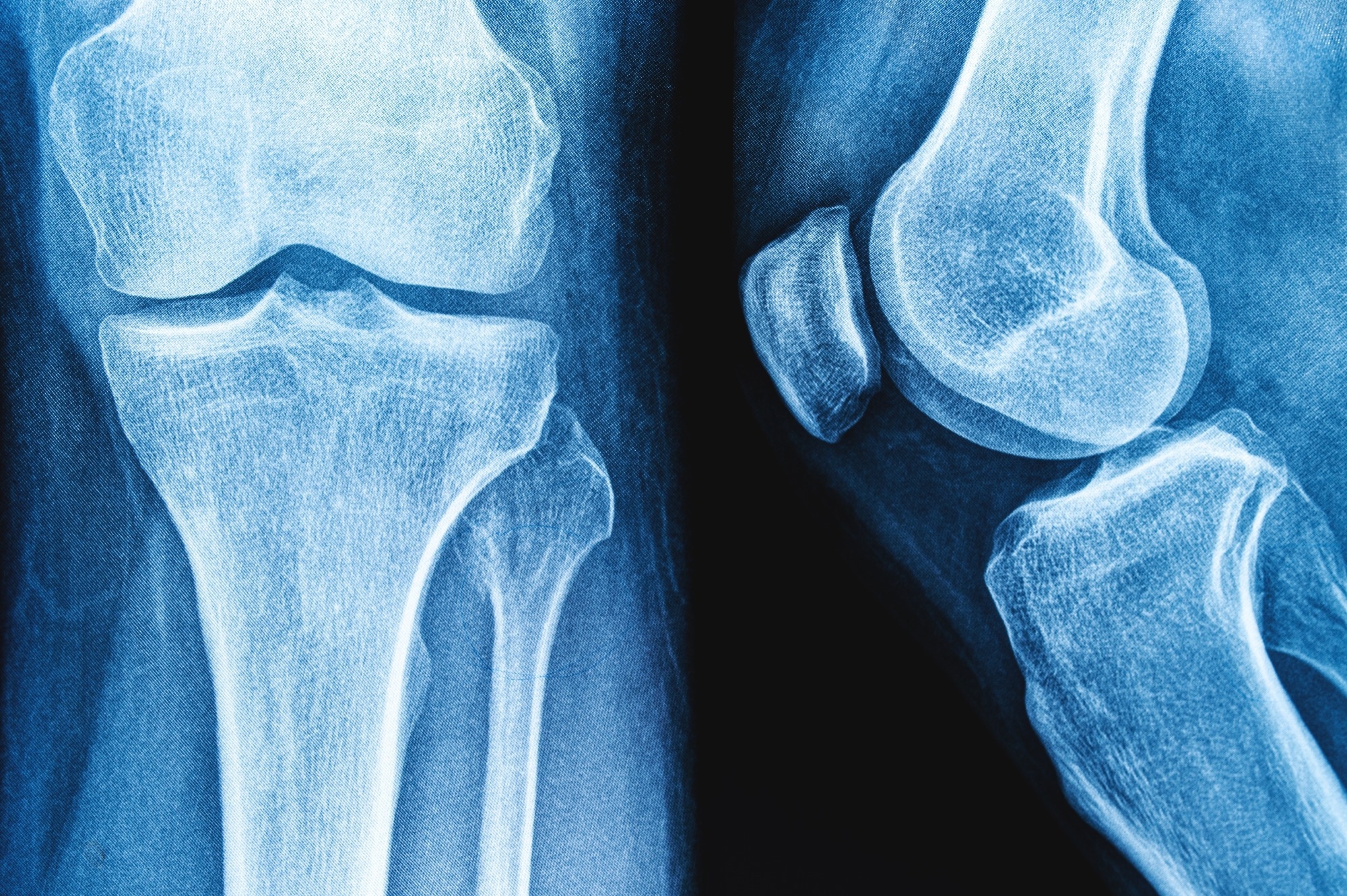
Joint Pain: Many patients experience joint pain, stiffness, or swelling while taking Isimetan. This can affect various joints in the body, such as the knees, hips, and hands, and may limit mobility and physical activity.
Less Common but Serious Adverse Effects
Osteoporosis and Bone Fractures: Isimetan, by reducing estrogen levels, can have a negative impact on bone health. Long-term use of Isimetan has been associated with an increased risk of osteoporosis and bone fractures, especially in postmenopausal women who are already at risk for bone loss. Regular bone density monitoring and appropriate interventions, such as calcium and vitamin D supplementation and bisphosphonate therapy, may be necessary to prevent bone-related complications.
Cardiovascular Events: Although the exact relationship is not fully understood, some studies have suggested a potential increased risk of cardiovascular events, such as heart attacks and strokes, in patients taking Isimetan.
Drug Interactions
● CYP3A4 Inducers and Inhibitors
As mentioned earlier, Isimetan is primarily metabolized by CYP3A4. Although it does not inhibit CYP isozymes, the use of strong CYP3A4 inducers, such as rifampin and phenytoin, may potentially decrease the plasma levels of Isimetan by increasing its metabolism. This could reduce the drug's efficacy in treating breast cancer. On the other hand, strong CYP3A4 inhibitors, like ketoconazole, do not appear to have a significant impact on Isimetan's pharmacokinetics, as demonstrated in clinical studies. However, caution should still be exercised when co-administering Isimetan with other drugs that are known to interact with the CYP3A4 enzyme system.
● Estrogen-Containing Medications
Isimetan should not be used concomitantly with estrogen-containing medications, such as hormone replacement therapy or oral contraceptives. Estrogen can counteract the effects of Isimetan by stimulating the growth of breast cancer cells, thereby reducing the drug's therapeutic efficacy. Patients on Isimetan therapy should be advised to avoid using any estrogen-containing products without consulting their healthcare provider.
● Other Drug Interactions
Isimetan may interact with other drugs that are metabolized by the CYP enzyme system or have similar pharmacokinetic properties. For example, drugs that are substrates of CYP3A4 may have their plasma levels affected by Isimetan, although the clinical significance of these interactions is often minimal. It is important for healthcare providers to review a patient's complete medication list before prescribing Isimetan to identify potential drug interactions and make appropriate dosage adjustments or select alternative medications if necessary.
Patient Case Presentation
Patient Information
Mrs. Smith, a 62 - year - old postmenopausal woman, presented to the oncology clinic with a history of a palpable lump in her right breast. She had no significant past medical history, except for well - controlled hypertension managed with a low - dose diuretic. She had a family history of breast cancer, with her mother being diagnosed at the age of 65.
Diagnosis and Initial Treatment
A mammogram and subsequent biopsy confirmed the presence of an invasive ductal carcinoma of the right breast, which was estrogen - receptor - positive (ER +) and progesterone - receptor - positive (PR +), with a low level of human epidermal growth factor receptor 2 (HER2) expression.
Disease Progression and Switch to Isimetan
During a routine follow - up visit, imaging studies revealed new bone metastases in the lumbar spine and left femur, indicating disease progression. Given her postmenopausal status and the progression of her estrogen - receptor - positive breast cancer after tamoxifen therapy, a decision was made to switch her to Isimetan tablets.
Frequently Asked Questions
Is exemestane a chemotherapy drug?
+
-
No, exemestane is not a traditional chemotherapy drug; it is a hormone therapy drug used to treat certain types of breast cancer. It works by lowering estrogen levels in the body to slow or stop the growth of estrogen-sensitive breast cancer cells.
Which is better, exemestane or letrozole?
+
-
Exemestane (steroidal, irreversible) and letrozole (nonsteroidal, reversible) are both aromatase inhibitors (AIs) for ER-positive breast cancer, but differ in mechanism, potency, and side effects, with letrozole typically causing deeper estrogen suppression but exemestane potentially offering benefits in sequential therapy due to a "non-cross-resistance" effect and milder androgenic activity, potentially reducing joint pain. Letrozole is a stronger estrogen suppressor, while exemestane's irreversible action and mild androgenic profile can affect side effect profiles, leading to its use when letrozole fails.
What to avoid when taking exemestane?
+
-
When taking exemestane, you must avoid estrogen-containing products (like HRT), certain medications (like rifampin, St. John's Wort, some anticonvulsants/antibiotics), grapefruit/pomelo, and pregnancy, while also managing side effects by limiting alcohol, caffeine, and spicy foods, and discussing bone health and contraception with your doctor. Always check with your healthcare team about any supplements, OTC drugs, or new prescriptions.
Does exemestane cause hair loss?
+
-
Yes, exemestane can cause hair loss or thinning, which is often a mild and temporary side effect that typically improves after treatment ends. This is a known side effect of aromatase inhibitors like exemestane, though it may not be severe for everyone.
Can you take vitamin D with exemestane?
+
-
There were no interactions found between exemestane and Vitamin D3. However, this does not necessarily mean no interactions exist. Always consult your healthcare provider.
Hot Tags: exemestane tablet, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale






