Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of edta ferric sodium salt cas 15708-41-5 in China. Welcome to wholesale bulk high quality edta ferric sodium salt cas 15708-41-5 for sale here from our factory. Good service and reasonable price are available.
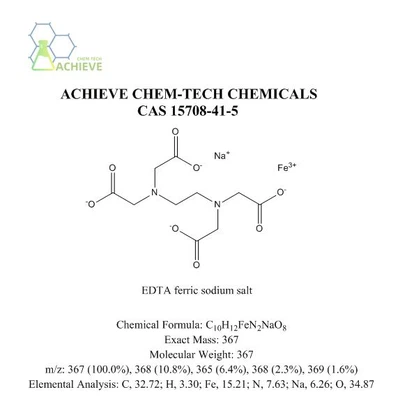
EDTA ferric sodium salt is a light yellow or brown powder that is easily soluble in water. M.W.C10H12FeN2NaO8, CAS 15708-41-5. When heated, some or all of the crystal water will be lost. Has good chemical stability and solubility, and can form stable complexes with metal ions. The structure of this compound has two acetate groups attached to iron ions, forming a stable five membered ring structure. In addition, the ethylenediamine moiety also attaches to the iron ion, forming a six membered ring. This unique structure gives it excellent chelating ability and can effectively chelate iron ions.

|
Chemical Formula |
C10H12FeN2NaO8 |
|
Exact Mass |
367.05 |
|
M.W |
367.05 |
|
m/z |
367 (100.0%), 368 (10.8%), 365 (6.4%), 368 (2.3%), 369 (1.6%) |
|
Elemental Analysis |
C, 32.72; H, 3.30; Fe, 15.21; N, 7.63; Na, 6.26; O, 34.87 |
|
|
|

Ethylenediaminetetraacetic acid sodium iron, as a chelating iron compound, has shown wide application value in food, medicine, agriculture, industry, and daily chemical industries due to its high stability, strong absorption rate, and low side effects. The following systematically summarizes its core uses and scientific basis from five dimensions.
1. Core function: Efficient iron supplemntation and anemia intervention
It is internationally recognized as an efficient iron fortifier, with an iron absorption rate 2-3 times that of traditional ferrous sulfate. This advantage stems from its unique chelating structure: EDTA molecules tightly encapsulate iron ions through six coordinating atoms, forming stable water-soluble complexes. This structure remains stable in the gastric acid environment, avoiding gastrointestinal irritation caused by premature release of iron ions; After entering the duodenum, EDTA dissociates from iron ions, and the released iron ions can be efficiently absorbed by intestinal mucosal cells.


Clinical validation:
In 2004, the Chinese Center for Disease Control and Prevention conducted a 3-month intervention study among Miao school-age children in Guizhou. The results showed that daily intake of 2mg NaFeEDTA could reduce anemia rate from 34.2% to 12.7%, and increase hemoglobin concentration by an average of 11.2g/L.
In 2005, a 2-year follow-up study conducted by the Global Alliance for Improved Nutrition (GAIN) on 1200 Mexican women confirmed that NaFeEDTA fortified flour significantly improved iron storage indicators (serum ferritin increased by 38%).
2. Application scenarios: from basic foods to special diets
Staple food reinforcement: China, India and other countries have added EDTA ferric sodium salt to flour and rice to solve the iron deficiency problem of mainly grain based cannibals. For example, the Chinese Ministry of Health has approved its use in soy sauce (with an added amount ≤ 180mg/kg) to achieve implicit iron supplemntation through daily seasoning.
Dairy products and beverages: Adding NaFeEDTA to infant formula milk powder and sports drinks can avoid precipitation caused by the binding of iron and milk protein, while improving bioavailability.
Special diet: In nutritiona supplemnts designed for pregnant women and the elderly, NaFeEDTA is often combined with vitamin C to further promote iron absorption.
Medical and Health: From Anemia Treatment to Heavy Metal Detoxification

Innovative application of heavy metal detoxification:
The strong chelating ability of EDTA makes it an antidote for heavy metal poisoning such as lead and cadmium. NaFeEDTA can preferentially bind with heavy metal ions in the body, forming stable soluble complexes that are excreted from the body through urine.
Clinical case: In 2010, a lead contaminated area in China used NaFeEDTA (500mg per day) combined with dimercaptopropanol to treat lead poisoning in children. After 3 months, the blood lead concentration decreased from 45.2 μ g/dL to 18.7 μ g/dL, with an effective rate of 92%.
Mechanism study: The binding constant between EDTA and lead ions (logK=18.3) is much higher than that of iron ions (logK=14.3), ensuring that iron ions are not heavily chelated during the detoxification process and avoiding aggravating anemia.
Soilless cultivation and water fertilizer integration
NaFeEDTA is an ideal iron source for soilless cultivation and integrated water fertilizer systems
Stability: It is not easy to precipitate in nutrient solution and can maintain effective concentration for a long time.
Compatibility: Has no antagonistic effect on nutrients such as calcium and magnesium, avoiding nutritiona imbalance.
Precise regulation: Supply according to demand through drip irrigation system, reducing waste and environmental pollution.
Data support: Research from Wageningen University in the Netherlands shows that the utilization rate of NaFeEDTA in tomato hydroponics is 62%, which is 2.3 times higher than that of ferrous sulfate.

Industrial sector: from catalysts to environmentally friendly mterials

1. Multi functional catalysts in the chemical industry
NaFeEDTA has a unique catalytic effect in organic synthesis:
Oxidation reaction: As a Fe ³ ⁺ source, it catalyzes the oxidation of phenol to p-benzoquinone with a selectivity of 95%.
Aggregation reaction: In the polymerization of styrene butadiene rubber, EDTA ferric sodium saltchelates ferrous ions to control the polymerization rate and improve the uniformity of moleular weight distribution.
Electroplating industry: As a stabilizer for plating solution, it prevents Fe ² ⁺ from oxidizing to Fe ³ ⁺ and reduces coating defects.
2. Water treatment agents in the field of environmental protection
NaFeEDTA can remove heavy metal ions from water through chelation:
Wastewater treatment: In electroplating wastewater treatment, NaFeEDTA forms soluble complexes with Pb ² ⁺ and Cd ² ⁺, and heavy metals are recovered through ion exchange resin.
Soil remediation: Spraying NaFeEDTA solution can activate heavy metals in soil, combined with plant extraction technology to remediate polluted soil.
Case: A chemical industrial park in Jiangsu Province uses NaFeEDTA (50mg/L) to treat chromium containing wastewater, with a chromium removal rate of 98% and effluent that meets the Comprehensive Wastewater Discharge Standard (GB 8978-1996).


3. Stabilizers and chelating agents in daily chemical products
In cosmetics and detergents, NaFeEDTA has the following functions:
Anti hard water: chelates calcium and magnesium ions to prevent shampoo and laundry detergent from settling in hard water.
Anti corrosion and Efficiency Enhancement: Used in combination with preservatives such as sodium benzoate to extend the shelf life of the product.
Skin protection: In anti-aging skincare products, EDTA chelates metal ions to reduce the generation of free radicals.
Formula example: 0.5% NaFeEDTA added to a brand of facial cleanser can keep the product stable in the pH range of 5-9, and increase the foam content by 20%.
Nanocatalysis and Energy Storage
NaFeEDTA has unique advantages in the field of nanocatalysis:
Photocatalysis: As a Fe ³ ⁺ source, Fe TiO ₂ nanocomposites are synthesized to improve the efficiency of visible light photocatalytic degradation of organic pollutants.
Battery mterial: In lithium-ion batteries, EDTA ferric sodium salt chelated iron can stabilize the electrode structure and extend the cycle life.
Experimental data: LiFePO ₄/C cathode mterial prepared with NaFeEDTA as the iron source has a first discharge capacity of 165mAh/g at a rate of 0.1C, and a capacity retention rate of 92% after 100 cycles.


Synthesis Methods
This process typically begins with the preparation of specific precursors or intermediates that are then combined to yield the desired compound.
One commonly used method involves the reaction of iron (III) chloride with ethylenediaminetetraacetic acid (EDTA) and a sodium salt. In this process, the reactants are mixed in a controlled environment, often with the assistance of heat, to facilitate the formation of the desired chelate. The reaction conditions, such as temperature and pH, are carefully monitored to ensure optimal product formation and purity.
Another method involves the use of carbonate precursors. In this approach, a solution of ferrous sulfate is mixed with a carbonate salt, such as sodium carbonate, to produce a carbonate intermediate. This intermediate is then reacted with an EDTA salt solution, often under heated conditions, to yield the EDTA iron (II) sodium salt. The reaction is carefully controlled to ensure complete conversion and to minimize any side reactions.
In both methods, the resulting solution containing the EDTA iron (II) sodium salt is then subjected to purification steps to remove any impurities and obtain a high-purity product. This typically involves filtration, crystallization, washing, and drying steps.
It is worth noting that the specific synthesis method chosen can depend on various factors, including the availability of reactants, the desired purity of the product, and the specific application for which the EDTA iron (II) sodium salt is intended. Therefore, it is important to carefully consider these factors when selecting a synthesis route.
In summary, the synthesis of EDTA ferric sodium salt involves the controlled reaction of iron and EDTA precursors, followed by purification steps to obtain a high-purity product. The specific method chosen should be tailored to the available reactants and the desired properties of the final product.
Hot Tags: edta ferric sodium salt cas 15708-41-5, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Hinokitiol powder, 5 hydroxymethylfurfural synthesis, s allyl l cysteine, spermidine powder, Pamoic acid