Pure trisodium citrate dihydrate is a chemical substance with the molecular formula of Na3C6H5O7.2H2O, white granular crystal or white crystalline powder, odorless and salty. Slightly soluble in ethanol, easily soluble in water and glycerin. The aqueous solution is alkaline. It is obtained by neutralization of citric acid with sodium hydroxide or sodium bicarbonate. The substance is widely used in food additives, emulsion enhancers and pharmaceutical industry. It is an anticoagulant and can also be used as a buffer and food preservative.

|
Chemical Formula |
C6H9Na3O9 |
|
Exact Mass |
294 |
|
Molecular Weight |
294 |
|
m/z |
294 (100.0%), 295 (6.5%), 296 (1.8%) |
|
Elemental Analysis |
C, 24.50; H, 3.08; Na, 23.45; O, 48.96 |
|
|
|

Pure trisodium citrate dihydrate, as a multifunctional food additive, occupies an important position in the food industry due to its unique chemical properties and safety. Its core functions include regulating acid-base balance, chelating metal ions, stabilizing food structure, and improving taste. It is widely used in various fields such as beverages, dairy products, meat products, baked goods, etc.
Acidity regulation and flavor optimization

Beverage industry
In carbonated drinks, fruit juice drinks, and sports drinks, by neutralizing acidic components (such as citric acid and phosphoric acid), the pH value is stabilized within the range of 3.5-6.0, which not only prevents strong acidity from stimulating the oral mucosa, but also avoids excessive alkalinity from damaging nutrients such as vitamin C. For example, adding 0.2% -0.5% sodium citrate to orange juice beverages can significantly alleviate the sharp acidity of citric acid, create a soft fruity taste, and extend the shelf life to more than 12 months.
Improvement of dairy products
In fermented dairy products such as yogurt and cheese, sodium citrate chelates calcium ions (Ca ² ⁺) to inhibit protein coagulation and prevent granular precipitation of the product. For example, adding 0.3% sodium citrate to processed cheese can make the texture finer, lower the melting point by 10-15 ℃, and improve the applicability of hot processed foods such as pizza. In addition, its slightly alkaline properties can neutralize the sour taste produced by lactic acid fermentation and balance the flavor.


Preservation of meat products
In cured meat products such as sausages and ham, sodium citrate inhibits the taste of meat caused by fat oxidation by chelating iron ions (Fe ³ ⁺), while synergistically working with phosphates to improve the water retention of meat. Experimental data shows that ham sausage with 0.5% sodium citrate added has a moisture content 8% -12% higher than the untreated group, reduces juice loss by 30% during slicing, and extends shelf life to 90 days.
Structural stability and texture improvement
Construction of gel system
In gel foods such as jelly and soft candy, sodium citrate, gelatin, carrageenan and other colloids form a composite network structure to enhance elasticity and transparency. For example, adding 0.4% sodium citrate to mango jelly can increase the strength of gel by 25%, and the elongation at break by 40%. Even at high temperature (40 ℃), it still remains stable.


Stable emulsion system
In emulsified foods such as ice cream and cream, sodium citrate promotes uniform dispersion of fat globules by reducing interfacial tension. For example, adding 0.2% sodium citrate to vanilla ice cream can increase the expansion rate by 15%, enhance the melt resistance by 20%, and make the taste denser. In addition, its chelating effect can prevent emulsifiers (such as monoglycerides) from failing to bind with metal ions.
Anti caking and dispersibility
In powdered foods such as milk powder and seasonings, sodium citrate adsorbs onto the surface of particles, forming electrostatic repulsion to prevent clumping. For example, adding 0.1% sodium citrate to infant formula can increase the flowability of the powder by 30% and accelerate the dissolution rate by 50% during mixing.

Nutritional reinforcement and functional extension

Mineral supplements
Sodium citrate, as a sodium source (with a sodium content of about 21%), can replace some sodium chloride (NaCl) and reduce the sodium content in food. For example, in low sodium salt, replacing 30% NaCl with sodium citrate can reduce the sodium content by 25% while retaining the salty taste, making it suitable for people with hypertension to consume. In addition, its citrate ions (C ₆ H ₅ O ₇³ ⁻) can promote the absorption of minerals such as calcium and magnesium, enhancing bone health.
Synergistic antioxidant effect
The combination of sodium citrate with antioxidants such as vitamin C and vitamin E can significantly enhance its stability. For example, adding 0.1% sodium citrate to fruit juice can increase the retention rate of vitamin C from 60% to 85% and extend the shelf life by 3-6 months. Its mechanism of action is to chelate metal ions (such as Fe ² ⁺, Cu ² ⁺) and block free radical chain reactions.


Fermentation accelerator
In fermented foods such as bread and yogurt, sodium citrate provides a suitable growth environment for yeast or lactic acid bacteria by adjusting the pH value. For example, adding 0.3% sodium citrate to bread dough can shorten fermentation time by 20%, increase dough expansion rate by 15%, increase bread volume by 10%, and improve internal tissue structure.

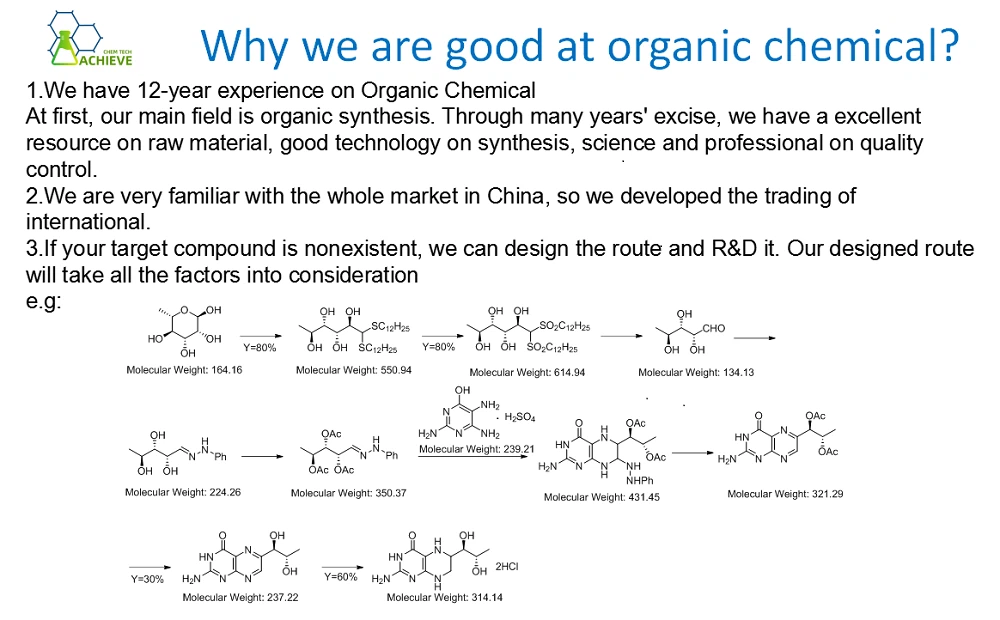
Synthesis of pure trisodium citrate dihydrate:
1. It is obtained by neutralization of citric acid with sodium hydroxide or sodium bicarbonate. Dissolve sodium bicarbonate in water under stirring and heating, add citric acid, continue to raise the temperature to 85-90 ℃, and adjust the pH to 6.8. Decolorize with activated carbon and filter while hot. The filtrate is concentrated under reduced pressure and crystallized after cooling. Filter, wash and dry to obtain sodium citrate.
First, stir and dissolve sodium bicarbonate in hot water, then add citric acid to neutralize it at 85~90 ℃ to a Ph value of 6.8. Add activated carbon to decolorize and filter while hot. The filtrate is concentrated under reduced pressure, cooled and crystallized, centrifuged, washed and dried to obtain sodium citrate.
C6H8O7+3NaHCO3→C6H5Na3O7·2H2O+3CO2↑+H2O
2. Use anhydrous citric acid dried at 70 ℃ and anhydrous sodium carbonate dried at 160 ℃ as raw materials. Dissolve 1mol citric acid in 600mL water, add a small amount of stoichiometric sodium carbonate for many times, and keep stirring. Boil the solution for 30min. Filter, collect the filtrate in an evaporating dish of known weight, and concentrate it on a water bath until crystals are separated. The crystals were air dried and then dried at 70 ℃.
3. It is obtained by neutralization of citric acid with sodium hydroxide or sodium bicarbonate.
Pure trisodium citrate dihydrate Excellent performance:
Sodium citrate is the most important citrate at present, which is mainly produced by starch substances through fermentation to produce citric acid, and then neutralized with alkali substances. It has the following excellent properties:
It is safe and non-toxic. Since the raw materials for preparing sodium citrate are basically from grain, it is absolutely safe and reliable, and will not harm human health. The Food and Agriculture Organization of the United Nations and the World Health Organization do not impose any restrictions on its daily intake, so it can be considered as non-toxic.
It is biodegradable. After being diluted by a large amount of water in nature, sodium citrate partially becomes citric acid, and both of them coexist in the same system. Citric acid is easily biodegradable in water under the action of oxygen, heat, light, bacteria and microorganisms. Its chemicalbook decomposition pathway is generally through aconitic acid, itaconic acid and limoconic acid, which are converted into carbon dioxide and water.
It has metal ion complexing ability. Sodium citrate has good complexing ability to Ca2+, Mg2+and other metal ions, and also has good complexing ability to other metal ions, such as Fe2+plasma.
Excellent solubility, and solubility increases with water temperature.
It has good pH regulation and buffering performance. Sodium citrate is a weak acid and strong base salt, which can form a strong pH buffer when it is mixed with citric acid. Therefore, it is of great use in some occasions where it is not suitable for a wide range of pH changes. In addition, sodium citrate has excellent retarding and stabilizing properties.
Emergency treatment measures:
First aid:
Inhalation: If inhaled, move the patient to fresh air. Skin contact: Take off the contaminated clothes and wash the skin thoroughly with soap and water. If you feel unwell, see a doctor.
Eye contact: separate the eyelids and wash them with flowing water or normal saline. Get medical attention immediately.
Ingestion: Rinse mouth, do not induce vomiting. Get medical attention immediately.
Advice to protect the rescuer: transfer the patient to a safe place. Consult a doctor. Show this chemical safety data sheet to the doctor on site.
Fire fighting measures
Extinguishing agent: use water mist, dry powder, foam or carbon dioxide extinguishing agent to extinguish the fire. Avoid using direct water to extinguish the fire. Direct water may cause splashing of flammable liquid and spread the fire.
Special risk: no data available.
Precautions and protective measures for fire fighting: firefighters must wear air breathing apparatus and full body fire-fighting clothes to extinguish the fire in the upwind direction. Move containers from the fire site to an open place as much as possible. If the container in the fire site has changed color or made a noise from the safety relief device, it must be evacuated immediately. Isolate the accident site and prohibit irrelevant personnel from entering. Receive and treat fire water to prevent environmental pollution.
Leakage emergency treatment
Protective measures, protective equipment and emergency response procedures for operators:
It is recommended that emergency treatment personnel wear air breathing apparatus, anti-static clothing and rubber oil resistant gloves. Do not touch or cross the leakage. All equipment used in operation shall be grounded. Cut off the source of leakage as much as possible. Eliminate all sources of ignition. The warning area shall be demarcated according to the influence area of liquid flow, steam or dust diffusion, and irrelevant personnel shall evacuate to the safe area from the crosswind and upwind direction.
Environmental protection measures: collect the leakage to avoid environmental pollution. Prevent leakage from entering sewers, surface water and groundwater. Receiving and removal methods of leaked chemicals and disposal materials used:
Small amount of leakage: collect the leaked liquid in a sealable container as far as possible. Absorb with sand, activated carbon or other inert materials and transfer to a safe place. Do not flush into the sewer.
Large amount of leakage: build a dike or dig a pit to receive. Close the drainage pipe. Cover with foam to inhibit evaporation. Use explosion-proof pump to transfer to tank car or special collector for recycling or transport to waste disposal site.
faq
What is trisodium citrate dihydrate used for?
Trisodium citrate dihydrate is widely used in foods, beverages and various industrial applications mainly as buffering, sequestering or emulsifying agent. Trisodium citrate dihydrate occurs as white, granular crystals or as white, crystalline powder with a pleasant, salty taste.
What is another name for trisodium citrate dihydrate?
2.4 Synonyms
Trisodium citrate dihydrate.
6132-04-3.
Sodium citrate dihydrate.
Sodium citrate hydrate.
Sodium citrate hydrous.
B22547B95K.
Citric acid, trisodium salt, dihydrate.
Nauzene.
What is the difference between sodium citrate and trisodium citrate dihydrate?
Sodium Citrate Dihydrate is the hydrated form of trisodium citrate, with the chemical formula Na₃C₆H₅O₇·2H₂O. It contains two molecules of water of crystallization, giving it slightly different handling and solubility characteristics compared to its anhydrous counterpart.
Is trisodium citrate dihydrate a preservative?
Hot Tags: pure trisodium citrate dihydrate cas 6132-04-3, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, Pamoic acid, s allyl l cysteine, spermidine powder, Hinokitiol powder, 5 hydroxymethylfurfural synthesis









