Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of clonidine hydrochloride powder cas 4205-91-8 in China. Welcome to wholesale bulk high quality clonidine hydrochloride powder cas 4205-91-8 for sale here from our factory. Good service and reasonable price are available.
Clonidine Hydrochloride Powder is a medication primarily used in the management of hypertension, or high blood pressure. It belongs to the class of drugs known as central alpha-2 adrenergic agonists, which work by acting on specific receptors in the brain to decrease the sympathetic outflow, thereby reducing heart rate and peripheral vascular resistance.
This fine, white to off-white powdered substance is the hydrochloride salt of clonidine, a chemical compound with established pharmacological properties. When administered, either orally or transdermally, it helps to lower blood pressure by promoting relaxation of blood vessels and decreasing the heart's workload.
Beyond its antihypertensive effects, it is also indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in some cases, particularly when used as an adjunct therapy. Additionally, it finds application in the management of withdrawal symptoms associated with opioid detoxification and alcohol withdrawal, owing to its ability to reduce anxiety and agitation.
Importantly, the use must be carefully monitored by healthcare professionals due to potential side effects such as drowsiness, dry mouth, dizziness, and constipation. It's crucial to follow prescribed dosages strictly and to be aware of possible interactions with other medications, especially those that affect blood pressure or heart rate.
Overall, clonidine hydrochloride powder represents a valuable therapeutic option in the treatment of hypertension and other conditions, but its benefits must be weighed against potential risks and side effects under medical supervision.

|
|
|
|
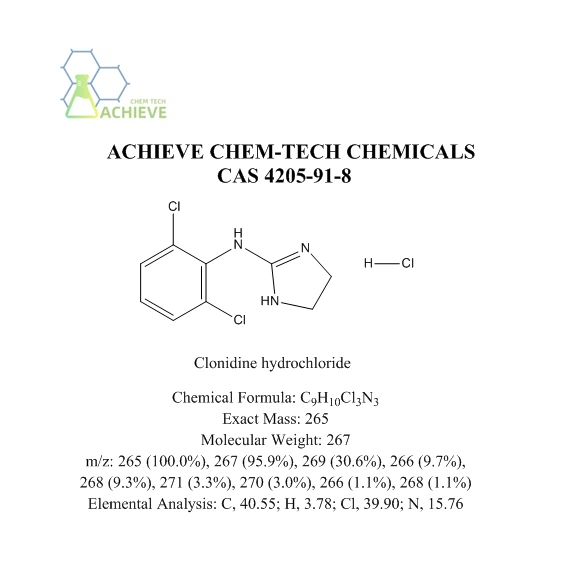
Chemical Formula |
C9H10Cl3N3 |
|
Exact Mass |
265 |
|
Molecular Weight |
267 |
|
m/z |
265 (100.0%), 267 (95.9%), 269 (30.6%), 266 (9.7%), 268 (9.3%), 271 (3.3%), 270 (3.0%), 266 (1.1%), 268 (1.1%) |
|
Elemental Analysis |
C, 40.55; H, 3.78; Cl, 39.90; N, 15.76 |
Synthesis method
A. Synthesis of Intermediate 1
Add 188ml formic acid and 100.0g 2,6-dichloroaniline to the reaction flask, heat and stir in an oil bath to dissolve, stir and reflux at 95℃-100℃ for 5h, then cool to room temperature, then cool to 0℃, stir and crystallize for 2h, filter and dry to obtain 92.0g of white solid with a yield of 78.4%.
B. Synthesis of Intermediate 2
Add 70ml thionyl chloride and 92g intermediate 1 to the reaction flask and stir, cool to 0℃ in an ice-water bath, and add a mixture of 141ml thionyl chloride and 156ml sulfonyl chloride. After the addition is completed, the temperature is naturally raised to room temperature, and then raised to 40°C and stirred for reaction for 20 hours. After the system is dissolved, it is distilled under reduced pressure until there is no fraction, 300 ml of dichloromethane is added, and the temperature is cooled to 5°C in an ice-water bath. A dichloromethane solution of 131 g of ethylenediamine is added dropwise. After the addition is completed, the temperature is naturally raised to room temperature. The reaction is stopped for 2 hours. After the reaction is completed, 200 ml of water is added to the system, stirred for 30 minutes, and allowed to stand for stratification. The aqueous phase is extracted with 100 ml*2 of dichloromethane, the aqueous phase is discarded, the organic phases are combined, washed with water 3 times, washed with 10% sodium hydroxide twice, and washed with saturated brine once, and the dichloromethane layer is collected and concentrated to obtain intermediate 2. Add 300ml of anhydrous ethanol to intermediate 2, add activated carbon, stir and heat to reflux, filter while hot, add 20ml of hydrogen chloride-ethanol solution to the filtrate, keep warm and stir for 30min, cool naturally to room temperature, cool to 0℃ and stir to crystallize for 3h, filter, rinse the filter cake with a small amount of ethanol, dry to obtain clonidine hydrochloride, dissolve clonidine hydrochloride in 300ml of water, stir at room temperature for 1h, filter to remove insoluble matter, cool to below 10℃ in an ice-water bath, adjust pH to 10 with 25% sodium hydroxide, generate white precipitate, filter, rinse the filter cake with a small amount of water, and obtain 60.3g of off-white solid.
C. Synthesis of clonidine hydrochloride
Add 250ml of ethanol and 60.3g of intermediate 2 to a 500ml reaction bottle, stir and heat to reflux. Remove the insoluble matter while hot, add 29.2g of hydrogen chloride-ethanol solution to the filtrate, stir under reflux for 30min, then cool to room temperature naturally, then cool to 0℃-5℃ and stir to crystallize for 3h, filter with suction, rinse the filter cake with a small amount of ethanol to obtain the crude product of clonidine hydrochloride. Add the wet solid obtained in the previous step to a 1-liter reaction bottle, add 250ml of ethyl acetate, and stir at room temperature for 1h. Then filter, rinse the filter cake with a small amount of ethyl acetate, and dry to obtain 48.9g of white solid, with a yield of 90.01%.
Dosages
The dosage and administration of clonidine hydrochloride powder varies depending on the therapeutic purpose. Below are the specific dosages and uses:
- Reduce blood pressure: Starting dose: 0.1 mg twice daily. Increasing Dose: 0.1-0.2mg daily at 2-4 day intervals as needed. Common Maintenance Dose: 0.3-0.9mg/day in 2-4 oral doses. In severe hypertension requiring emergency treatment: start with 0.2mg orally, followed by 0.1mg hourly increments until diastolic blood pressure is controlled or a total of 0.7mg is reached, after which a maintenance dose is used.
- Menopausal hot flashes: Dose: 0.025-0.075mg each time, twice daily.
- Severe dysmenorrhea: Dose: 0.025mg orally twice daily for 14 days before and at the time of menstruation.
- Migraine: Dose: 0.025mg once, 2-4 times daily, up to 0.05mg 3 times daily.
- Precautions: Extreme: 0.6mg once, 2.4mg a day. Before using Colistin Hydrochloride, you should consult a medical professional and take the medication exactly as prescribed. Colistin Hydrochloride may cause some adverse reactions, such as dry mouth, dizziness, reduced sexual function, etc. If uncomfortable symptoms occur, you should seek medical advice. The dosage should be gradually reduced when stopping the drug to avoid rebound increase in blood pressure.
The above information is for reference only, the specific dosage and usage should be adjusted according to individual situation and doctor's advice. During the use of colistin hydrochloride, blood pressure and heart rate should be monitored regularly to ensure the effectiveness and safety of the drug.

- Effectiveness: Colistin Hydrochloride is primarily used to treat hypertensive emergencies. It enters the body and stimulates adrenal receptors in the brainstem, which causes the sympathetic nervous system in the central nervous system to atrophy, thereby lowering blood pressure.
- Cautions: Colistin Hydrochloride is not usually regarded as the first-line treatment for hypertension, so patients should follow their doctor's guidance when choosing antihypertensive drugs to choose the most suitable drug for themselves.

Treatment of migraine

- Efficacy: Colistin Hydrochloride can be used to treat migraine headaches. In addition, it can also treat hot flashes and menstrual cramps that occur during menopause in women.
- ADVERSE REACTIONS: Some adverse reactions may occur with the use of colistin hydrochloride such as dizziness, lethargy, and depression. Some patients may also experience nausea, vomiting, insomnia and palpitations.

In September 2010, the U.S. Food and Drug Administration (FDA) granted approval to clonidine hydrochloride extended-release tablets in strengths of 0.1mg and 0.2mg (branded as Kapvay by Shionogi Inc.). This approval marked a significant milestone as it was the first formulation of clonidine hydrochloride powder specifically authorized by the FDA for the treatment of attention deficit hyperactivity disorder (ADHD) in children aged 6 to 17 years.
Kapvay is indicated for use as a monotherapy or as an adjunct to stimulant medications in the management of ADHD symptoms. Its approval underscores the unique role it plays in the therapeutic arsenal against ADHD, particularly as the only non-stimulant drug approved for use alongside stimulants to enhance treatment outcomes.
The extended-release formulation ensures a sustained release of clonidine hydrochloride over an extended period, providing more stable blood levels and potentially reducing the frequency of dosing compared to immediate-release formulations. This can improve patient compliance and convenience, especially in pediatric populations.
The approval of Kapvay recognizes the need for diverse treatment options in ADHD, acknowledging that not all patients respond adequately to stimulants alone. By offering an alternative or adjunctive therapy, Kapvay addresses a critical unmet need in the management of ADHD, potentially improving the quality of life for many children and adolescents struggling with this condition.
It's important to note that, like any medication, Kapvay should be prescribed and monitored by a healthcare professional. The decision to use Kapvay, either alone or in combination with stimulants, should be based on a comprehensive assessment of the patient's symptoms, medical history, and potential risks and benefits. Close monitoring for side effects, such as drowsiness, dry mouth, and low blood pressure, is essential to ensure safe and effective treatment.
adverse reaction
Clonidine Hydrochloride, as an α ₂ adrenergic receptor agonist, is widely used as an adjuvant therapy for diseases such as hypertension, attention deficit hyperactivity disorder (ADHD), withdrawal syndrome (such as opioid or alcohol dependence), and migraine. Its powder form is mostly used in the production of pharmaceutical preparations or scientific research purposes, but direct contact or misuse may cause serious adverse reactions.
Cardiovascular system response
Hypotension and bradycardia
Clonidine stimulates central alpha ₂ receptors, inhibits sympathetic nervous activity, causes peripheral vasodilation and slowed heart rate, thereby lowering blood pressure. This is the main mechanism for treating hypertension, but it may also cause excessive hypotension (systolic blood pressure<90mmHg) or bradycardia (heart rate<60 beats/minute), especially during dose adjustment periods or in combination with other antihypertensive drugs. Patients may present with dizziness, fatigue, blurred vision, and in severe cases, fainting.
Reflex tachycardia
Some patients, especially those who are young or prone to orthostatic hypotension, may experience reflex tachycardia during initial medication due to a sudden drop in blood pressure, manifested as palpitations and chest tightness, which are usually transient and can be relieved with prolonged medication.
Hypertension rebound
Sudden discontinuation or omission of medication may lead to "rebound hypertension", characterized by a sharp increase in blood pressure (systolic blood pressure can rise by 30-40mmHg), accompanied by headaches, anxiety, tremors, excessive sweating, and even hypertensive encephalopathy. This phenomenon is related to sudden activation of the sympathetic nervous system and requires gradual reduction and discontinuation of medication.
Central nervous system response
Sedation and drowsiness
Clonidine can penetrate the blood-brain barrier, inhibit the central nervous system, and cause drowsiness, fatigue, and decreased attention, especially in the early stages of medication or at increased doses. About 10% -25% of children with ADHD report drowsiness, which may affect learning efficiency.
Depression and emotional disorders
Long term use may trigger or exacerbate depressive symptoms, manifested as low mood, decreased interest, and sleep disorders. Some patients may experience anxiety, irritability, and even suicidal ideation (rare but should be cautious).
Sleep disorders
Including insomnia, frequent dreaming, nightmares, especially when taking medication at night. About 5% -10% of pediatric patients report sleep disturbances, which may be related to the impact of medication on sleep cycles.
Cognitive function impact
A few studies suggest that long-term use may affect cognitive functions such as memory and attention, especially in elderly patients who require attention.
Hot Tags: clonidine hydrochloride powder cas 4205-91-8, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, CAS 1799917 71 7, 2 4 chlorophenyl 4 dibenzo b d furan 3 yl 6 phenyl 1 3 5 triazine, CAS 2252237 87 7, boronic acid B 1 naphthalenyl 2 3 4 5 6 7 8 d7 , 5 bromo 9 3 cyanophenyl 9H carbazole 3 carbonitrile, 9 3 dibenzo b d furan 1 yl phenyl 7 phenyl 7 9 dihydrobenzo g indolo 2 3 b carbazole