Ethyl methanesulfonate, with molecular formula C3H8O3S and CAS number 62-50-0, also known as EMS, is an important organic compound. It is a colorless to light yellow transparent oily liquid. Its clear and transparent appearance indicates high purity, without impurities or suspended solids. At standard atmospheric pressure (760 mmHg), its boiling point is 214.4 ± 9.0 ° C. At lower pressures, such as 10 mmHg, its boiling point drops to 85-86 ° C. This characteristic results in different volatility in different application environments. Its vapor pressure is about 0.2 ± 0.4 mmHg at 25 ° C, indicating that its volatility is relatively low at room temperature and pressure. It is a DNA ethylating agent with mutagenicity to plants and animals. It has been used as a model alkylating agent in DNA repair process research, inducing the substitution of guanine cytosine (G/C) bases with adenine thymine (A/T) bases. MES is a potential chemical mutagen used to induce mutations in rice, wheat, and Arabidopsis. Point mutations and single nucleotide polymorphisms can occur in the genome. It is also used to induce mutations in mouse embryonic fibroblasts, yeast human embryonic kidney cell lines, and human lymphocytes.

|
|
|
|
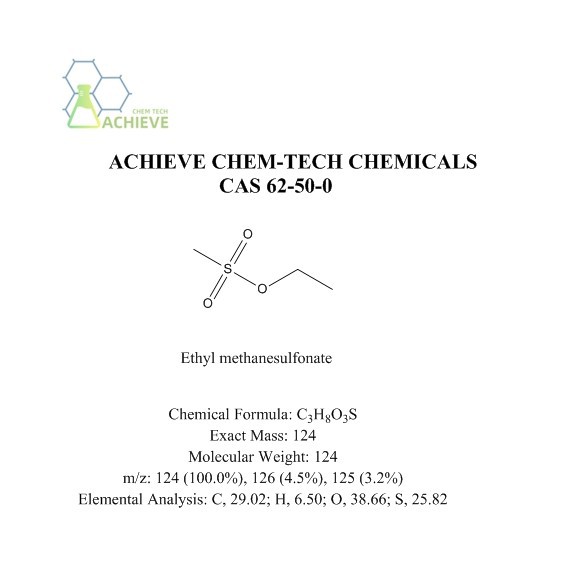
Chemical Formula |
C3H8O3S |
|
Exact Mass |
124 |
|
Molecular Weight |
124 |
|
m/z |
124 (100.0%), 126 (4.5%), 125 (3.2%) |
|
Elemental Analysis |
C, 29.02; H, 6.50; O, 38.66; S, 25.82 |

Ethyl Methanesulfonate (EMS), with the chemical formula C3H8O3S, is an important organic compound. It has a wide range of applications in various fields, including biological research, organic synthesis, and research as a potential carcinogen.
(1) Genetic research
As an efficient mutagen, it is widely used in genetic research. By processing experimental materials such as plant seeds, microbial cells, etc. through EMS, a large number of random mutants can be generated to study the relationship between gene function, genetic variation, and phenotype. This method provides a powerful tool for revealing the genetic laws of organisms and analyzing complex genetic traits.
(2) Molecular biology experiments
In molecular biology experiments, it can be used to prepare cell lines or strains with specific gene mutations. These mutants can be used to study biological processes such as gene expression regulation, protein function, and signal transduction pathways. By comparing the differences between wild-type and mutant types, we can gain a deeper understanding of the interactions and regulatory mechanisms between biomolecules.

2. Organic synthesis

(1) Drug synthesis
It has potential application value in the field of drug synthesis. Due to its unique chemical properties, EMS can be used as a synthetic raw material or intermediate for certain drug molecules. By participating in chemical reactions through EMS, drug molecules with specific pharmacophores can be constructed, providing important support for the development of new drugs.
(2) Preparation of functional materials
In addition to drug synthesis, it may also be used to prepare certain functional materials. For example, through the modification effect of EMS, nanomaterials with special surface or optical properties can be prepared. These materials have potential application prospects in fields such as optoelectronic devices, sensors, catalysts, etc.
(1) Research on Carcinogenic Mechanisms
Listed as a Class 2B carcinogen by the International Agency for Research on Cancer of the World Health Organization, indicating its potential carcinogenicity to humans. However, this characteristic also makes EMS an important tool for studying carcinogenic mechanisms. By inducing cell mutations and tumorigenesis through EMS, the pathogenesis of cancer can be deeply explored, potential anticancer targets can be identified, and the risk of cancer can be evaluated.
(2) Screening of anti-cancer drugs
Due to its ability to induce gene mutations, EMS can also be used for screening anti-cancer drugs. By treating cancer cell lines or animal models with EMS, anti-cancer drugs that are sensitive to specific gene mutations can be screened. This method provides a fast and effective screening platform for the development of new anti-cancer drugs.

4. Other potential applications

(1) Agricultural breeding
In addition to being directly used as a mutagen, it may also play an important role in agricultural breeding. By treating crop seeds or seedlings with EMS, mutants with excellent traits can be induced, providing a new approach for crop genetic improvement and new variety cultivation.
(2) Environmental Science
In the field of environmental science, volatility and biotoxicity make it an important object for studying environmental pollution and ecotoxicology. By monitoring the distribution and transformation process of EMS in the environment, its impact and potential risks on the ecosystem can be evaluated.

Ethyl methanesulfonate (EMS), as an important organic compound, has a wide range of applications in fields such as biology, chemistry, and medicine. The following is an introduction to a method for synthesizing high-purity EMS using methanesulfonic acid (MSA) and triethyl orthoformate (TEOF) as raw materials through a multi-step distillation and purification process. This method has the advantages of low cost, simple operation, high efficiency, and high yield.
1. Main raw materials and reagents
Methanesulfonic acid (MSA):
As a sulfonating agent, it provides sulfonic acid groups.
01
Triethyl orthoformate (TEOF):
As one of the substrates in esterification reactions.
02
Ethanol:
Used for azeotropic distillation to help remove residual TEOF.
03
Solvents (such as ethyl acetate):
Used in extraction and purification processes.
04
Other auxiliary reagents:
Such as dehydrating agents, catalysts (if necessary), etc.
05
2. Synthesis steps
Step description:
Add an appropriate amount of methanesulfonic acid and triethyl orthoformate to the reactor equipped with a stirrer, thermometer, and condenser. Control the reaction temperature within a certain range (usually from room temperature to reflux temperature), stir the reaction for a period of time, and allow the two to fully react. During the reaction process, products such as EMS, ethyl formate, and ethanol may be generated.
Chemical equation (schematic):
MSA + TEOF → EMS + Ethyl Formate + Ethanol
(Note: This equation is a schematic representation, and actual reactions may involve more complex intermediates and side reactions.)
Step description:
After the reaction is completed, transfer the reaction mixture to the distillation unit for atmospheric distillation. Due to the lower boiling points of ethyl formate and ethanol, they will be steamed out before EMS. By controlling the distillation temperature and time, ethyl formate and ethanol can be collected as by-products, respectively.
Step description:
After atmospheric distillation, the remaining reaction mixture contains unreacted TEOF and generated EMS. Transfer the mixture to a vacuum distillation device for vacuum distillation. At lower pressures, the boiling point of TEOF decreases and can be separated and recovered from the mixture. By controlling the distillation pressure and time, TEOF can be maximally recovered.
Step description:
To further remove residual TEOF, add an appropriate amount of ethanol to the remaining reaction mixture. Ethanol and TEOF can form an azeotrope, which can be more effectively removed from the mixture through azeotropic distillation. During the distillation process, control the temperature and pressure to evaporate and collect the azeotrope of ethanol and TEOF.
Step description:
After the above steps, high-purity EMS is mainly left in the mixture. Perform vacuum distillation again to remove any residual impurities and incompletely separated components. By precisely controlling distillation conditions such as temperature, pressure, and time, EMS with a purity of over 99.5% can be obtained.
3. Purification and post-treatment
After obtaining the crude EMS, further purification processes such as recrystallization, drying, etc. may be required to improve the purity and stability of the product. Specific methods can be selected and optimized according to actual needs. This method achieves effective utilization of raw materials and recovery of by-products through multi-step distillation and purification processes, and has the advantages of low cost, simple operation, high efficiency, and high yield. At the same time, this method also conforms to the concept of green chemistry and is of great significance for promoting the industrial production of ethyl methanesulfonate.
What are the side effects of this compound?
Potential side effects
Mutability and carcinogenicity
It is a DNA ethylating agent that can cause changes in DNA structure, thereby inducing gene mutations. This mutagenicity has been confirmed in animal experiments. Long term exposure or ingestion may increase the risk of cancer, especially in mammals. Therefore, for personnel engaged in related work, strict protective measures must be taken to avoid long-term exposure.
Damage to the reproductive system
It may have adverse effects on the reproductive system. Animal experiments have shown that this substance can cause damage to reproductive cells and a decline in reproductive function. For humans, long-term exposure or ingestion may have a negative impact on fertility. Therefore, pregnant and lactating women should avoid contact in particular.
Irritation to the skin and eyes
It has a pungent odor and may cause irritation to the skin and eyes. Direct contact may cause symptoms such as redness, itching, and pain in the skin; Splashing into the eyes may cause eye pain, tearing, redness, and other reactions. Therefore, appropriate personal protective equipment such as goggles, gloves, and respirators must be worn during use.
Damage to the digestive system
Although there is limited research on its direct damage to the digestive system, considering its chemical properties and irritability, long-term intake may have adverse effects on the digestive system. This may lead to the occurrence of digestive system symptoms such as nausea, vomiting, and loss of appetite. Therefore, when using, it is necessary to strictly follow the operating procedures to avoid ingestion or inhalation.
Potential impact on the nervous system
Although research on the direct effects of this substance on the nervous system is limited, some chemicals have the potential to affect the nervous system. Long term exposure or ingestion may have adverse effects on the nervous system, such as headaches, dizziness, memory loss, and other symptoms. However, these speculations require further experimental evidence to support them.
Potential damage to liver and kidney function
Liver and kidney function are important metabolic and excretory organs in the human body. Long term exposure or ingestion may have adverse effects on liver and kidney function, leading to the occurrence of liver dysfunction symptoms such as elevated transaminase levels and jaundice. At the same time, it may also cause damage to the kidneys, leading to symptoms of abnormal kidney function such as abnormal urine and edema. These speculations also require further experimental evidence to verify.

The development prospects of Ethyl Methanesulfonate (EMS) can be analyzed from the following aspects:
Market Trends and Growth Potential
With the continuous expansion of EMS application fields, its market size is expected to further expand. Especially in the field of plant genetics and breeding, the market demand for EMS will continue to grow. With the advancement of technology, the production process and purification technology of EMS will continue to improve, improving product quality and reducing production costs. This will help enhance the market competitiveness of EMS and further expand its application areas.
Policy and regulatory support
The government's support policies for technological innovation and agricultural development will provide strong guarantees for the development of EMS. For example, increasing investment in agricultural technology innovation and supporting the breeding of new crop varieties will help promote the application and development of EMS in the field of plant genetics and breeding.
Challenges and Risks
EMS has certain volatility and carcinogenicity, so it is necessary to strictly follow safety operating procedures during production and use to prevent harm to the environment and human health. With the continuous expansion of the EMS market, competition will become increasingly fierce. Enterprises need to enhance their market competitiveness through technological innovation, improving product quality and service levels.
Development prospects and outlook
EMS will continue to play an important role in genetic research and mutagenesis breeding, providing strong support for crop improvement and new variety breeding. Meanwhile, with the continuous advancement of technology and the improvement of policies and regulations, the production process and application areas of EMS will also continue to expand, and the market prospects are promising. However, enterprises also need to pay attention to challenges such as environmental and safety risks, as well as market competition, and strengthen technological innovation and risk management to ensure sustainable development.
Frequently Asked Questions
What chemicals can change your DNA?
+
-
In-vitro, animal, and human investigations have identified several classes of environmental chemicals that modify epigenetic marks, including metals (cadmium, arsenic, nickel, chromium, methylmercury), peroxisome proliferators (trichloroethylene, dichloroacetic acid, trichloroacetic acid), air pollutants (particulate ...
What is the disease caused by enzyme deficiency?
+
-
Enzyme deficiency diseases are genetic conditions where a missing or faulty enzyme disrupts metabolism, causing harmful substances to build up or essential ones to lack, leading to various symptoms like developmental delays (Tay-Sachs), severe intellectual disability (PKU), muscle weakness (Pompe disease, McArdle's), or digestive issues (lactose intolerance, Sucrase-Isomaltase deficiency). Examples include PKU, Tay-Sachs, Gaucher, Pompe, Fabry, and Galactosemia, often managed with strict diets, enzyme replacement, or supportive care.
What is the rarest disease on Earth?
+
-
The rarest disease is often cited as Ribose-5-phosphate isomerase deficiency (RPID), with only a handful of reported cases globally, characterized by severe neurological issues, but other contenders include Field's Disease (only two cases) and conditions like Fibrodysplasia Ossificans Progressiva (FOP), with about 700 known individuals. Defining the absolute "rarest" is difficult as new conditions are discovered, but these are some of the least common human ailments known.
Hot Tags: ethyl methanesulfonate cas 62-50-0, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 5 chloro 1 1 2 1 terphenyl 3 yl boronic acid, CAS 50548 40 8, 10 bromobenzo b naphtho 2 1 d furan, CAS 1332939 27 1, 4 Methylpropiophenone, 1 3 5 Triazine 2 1 1 biphenyl 4 yl 4 3 bromophenyl 6 phenyl