Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of gonadorelin pills in China. Welcome to wholesale bulk high quality gonadorelin pills for sale here from our factory. Good service and reasonable price are available.
Gonadorelin pills are an oral form of gonadotropin-releasing hormone (GnRH), with its active ingredient being a synthetic ten-peptide that has the exact same structure as the naturally produced GnRH in the human body. This drug acts by mimicking the pulsatile secretion of the hypothalamus and stimulating the anterior pituitary gland, thereby triggering the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). These two hormones are crucial for regulating the function of the gonads: in men, they promote testosterone synthesis and sperm production; in women, they regulate follicle development, ovulation, and estrogen secretion.
|
|
|
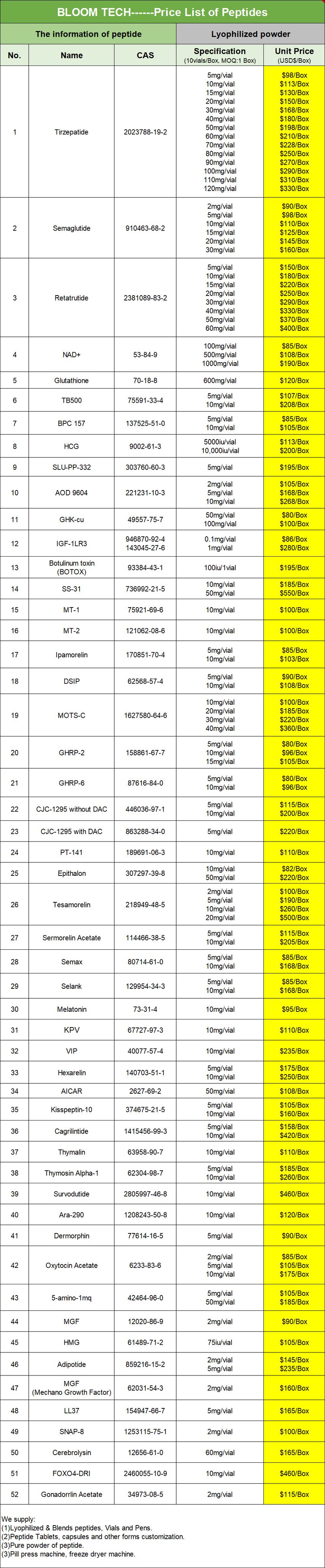


Gonadorelin COA


Suggestions for the rational selection of dietary supplements
Key Difference: The Positioning of Drugs and Dietary Supplements
Gonadorelin Pills are synthetic analogues of gonadotropin-releasing hormone (GnRH) and fall under the category of prescription drugs. Their primary function is to regulate the functions of the hypothalamus-pituitary-gonadal axis, achieving bidirectional regulation of sex hormone levels: for short-term use, it stimulates the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), promoting ovulation or sperm production; for long-term use, it inhibits sex hormone secretion through receptor desensitization, and is used to treat precocious puberty, endometriosis, and hormone-dependent tumors. The use of these drugs requires strict adherence to medical advice, and the indications cover the diagnosis and treatment of reproductive endocrine diseases, such as amenorrhea, infertility, and pituitary function assessment.

Clinically, it is mainly used for diagnosing the function of the hypothalamic-pituitary-gonadal axis or treating endocrine disorders caused by GnRH deficiency, such as hypogonadotropic hypogonadism (HH). Compared to the injectable it, the oral formulation improves the convenience of administration, but faces challenges such as first-pass effect and low bioavailability, and often requires special administration techniques (such as enteric-coated or absorption enhancers) to ensure efficacy. The use must be strictly in accordance with medical advice to avoid risks such as pituitary desensitization or abnormal hormone levels.
Dietary supplements fall under the category of food and are designed to supplement nutrients that may be lacking in one's daily diet (such as vitamins, minerals, amino acids, etc.). They cannot claim to treat or prevent diseases. Their core characteristics include:
Non-medicinal properties: Can be purchased without a prescription. The dosage of ingredients is subject to food regulations and must not contain active pharmaceutical ingredients.
Functional limitations: Only used to improve nutritional deficiencies, such as increased risk of osteoporosis due to vitamin D deficiency, or fatigue caused by B-vitamin deficiency.
Safety priority: Requires Good Manufacturing Practice (GMP) certification to avoid the risk of excessive intake (such as excessive vitamin A may cause liver damage).
Principles for the rational use of Gonadorelin Pills
Strict indication management
Fertility disorder diagnosis: Through the stimulation test (such as measuring the LH peak after intravenous injection of 100 μg), differentiate amenorrhea caused by hypothalamic or pituitary dysfunction. If the LH peak is < 5 IU/L, it indicates pituitary insufficiency (such as Sheehan syndrome); if the peak is normal, it may be due to hypothalamic dysfunction.
Treatment of precocious puberty: Children with precocious puberty need continuous medication to inhibit the gonadal axis and delay the process of sexual development. Usually, growth hormone monitoring of bone age progress is required.
Adjuvant treatment for tumors: Prostate cancer patients need to receive subcutaneous injection of 0.5 mg it daily, reduce the dosage to 0.1 mg/day after 7 days, continuously suppress testosterone to the castration level (< 50 ng/dL), and regularly monitor bone density and lipid levels.
Key points for risk control
Contraindication screening: Pregnant women, patients with hormone-dependent tumors, and those allergic to Gonadorelin Pills are prohibited from using.
Side effect management: Long-term use may lead to hot flashes, bone pain, and decreased libido. Calcium supplements (1000-1200 mg/day) and vitamin D (800-1000 IU/day) should be combined to prevent osteoporosis.
Drug interactions: Avoid using in combination with gonadotropin or sex hormone preparations to prevent counter-effect or enhancement of the effect.
Framework for rational selection of dietary supplements
Demand-oriented ingredient selection
Customization for specific populations:
Pregnant women: Supplement with folic acid (400-800 μg/day) and DHA (200-300 mg/day) to reduce the risk of neural tube defects in the fetus.
The elderly: Preferentially choose vitamin D (800-1000 IU/day) and calcium (1000-1200 mg/day), combined with resistance training to prevent osteoporosis.
Vegetarians: Supplement with vitamin B12 (2.4 μg/day), iron (18 mg/day), and Omega-3 (algal source, 250-500 mg/day).
Symptom-specific intervention:
Fatigue: Choose a compound supplement containing B vitamins (B1, B2, B6, B12) and magnesium (400 mg/day).
Weak immunity: Supplement with vitamin C (500-1000 mg/day) and zinc (15-30 mg/day), but avoid long-term high doses (zinc > 40 mg/day may inhibit immune function).
Safety assessment criteria
Third-party certification: Preferentially choose products that have passed USP (United States Pharmacopeia), NSF Certified for Sport, or Informed Choice certification to ensure purity of ingredients and consistency with labels.
Formula transparency: Avoid products containing "patented mixtures", choose supplements that clearly indicate the dosage of each ingredient. For example, probiotics should indicate the strain number (such as Lactobacillus rhamnosus GG) and the number of live bacteria (≥ 10^9 CFU/day).
Allergen control: Check if the label indicates common allergens (such as gluten, soy, dairy products). Gluten-sensitive individuals need to choose "Gluten-Free" certified products.

Scientific use norms
Dosage control: Follow recommended intake (RNI) or the maximum tolerated daily intake (UL), avoid overlapping intake. For example, the UL (Upper Limit) for vitamin A is 3000 μg/day. Excessive intake may increase the risk of fractures.
Taking timing: Fat-soluble vitamins (A, D, E, K) should be taken with meals to improve absorption rate; water-soluble vitamins (B, C) can be taken on an empty stomach.
Course management: After short-term use (such as 3 months), the effect needs to be evaluated. For long-term use, relevant indicators (such as vitamin D levels, bone density) need to be monitored regularly.
Key Conclusion: Drugs and supplements are not interchangeable
Gonadorelin Pills, as a prescription drug, its use requires strict medical diagnosis and course design. The dosage cannot be adjusted or stopped by oneself. Dietary supplements should be selected based on individual nutritional needs and safety assessment. Prioritize meeting basic nutritional requirements and avoid blindly pursuing "efficacy" components. There are essential differences in positioning, regulation, and functions between the two. Health management should follow the principle of "treating diseases with drugs and preventing deficiencies with nutrition", avoiding confusion in use.
First-pass effect and enzymatic degradation
First Pass Effect: The Natural Barrier of Oral Administration
The First Pass Effect refers to the phenomenon where, after a drug is absorbed through the gastrointestinal tract, it is metabolized by the enzyme system in the liver (such as cytochrome P450 enzymes), resulting in a significant reduction in the amount of the original drug that enters the systemic circulation. This effect has a decisive impact on the efficacy of oral medications:
Bioavailability sharply decreases: If the first-pass metabolism rate is 90%, only 10% of the original drug can enter the systemic circulation, and the dose needs to be significantly increased to achieve an effective concentration.
Significant individual differences: Liver enzyme activity is affected by factors such as genetic polymorphisms (such as CYP3A4 gene variations), age, and liver function status (such as liver cirrhosis), resulting in extremely large fluctuations in bioavailability among different patients.
Typical case: The oral bioavailability of nitroglycerin is less than 10%. Because it is rapidly inactivated by glutathione and organic nitrate reductase systems in the liver, it is necessary to use sublingual administration (to bypass the liver) to achieve rapid onset of action.
The first-pass effect challenge of Gonadorelin Pills
Chemical structure characteristics
Gonadorelin is an artificially synthesized ten-peptide. Its chemical name is 5-oxidized proline-L-tryptophan-L-phenylalanine-L-tyrosine-L-glutamine-L-leucine-L-arginine-L-proline-glycineamide, with a molecular weight of 1182.29. Its structure consists of:
N-terminal glutamic acid: Protects the peptide chain from being degraded by amino peptidase.
C-terminal amidation: Enhances the binding affinity with GnRH receptor.
Aromatic amino acids (tryptophan, tyrosine): Provide ultraviolet absorption properties (at 280nm), but are easily attacked by oxidizing enzymes.
Defects in oral absorption
Gastrointestinal barrier:
Gastric acid degradation: In an environment with a pH of 1.5-3.5 in the stomach, peptide bonds are easily hydrolyzed, resulting in the inactivation of the drug.
Protease attack: Pancreatic enzymes such as trypsin and chymotrypsin can specifically cut peptide chains, destroying the active structure.

Double blow of first-pass metabolism:
Intestinal enzyme degradation: The brush border membrane of the small intestine is rich in peptidases (such as aminopeptidase and dipeptidyl peptidase), which can gradually hydrolyze Gonadorelin.
Liver metabolism: Even a small amount of the drug entering the portal vein will be further decomposed by the CYP450 enzymes (such as CYP3A4) and peptidases within the liver cells.
Clinical data support:
Animal experiments have shown that the bioavailability of Gonadorelin after oral administration is less than 1%, which is much lower than that of injection administration (100%).
In human studies, an oral dose needs to be 50-100 times that of the injection dose to observe similar efficacy, but it is accompanied by severe gastrointestinal side effects (such as nausea and vomiting).
Enzyme Degradation Mechanism: Comprehensive Analysis from Molecular to Clinical
Key Metabolic Enzyme System
CYP450 enzyme family:
CYP3A4: Accounts for 30% of the total CYP450 content in the liver. It can oxidize the tryptophan and tyrosine residues in Gonadorelin, generating hydroxylated or deaminated metabolites.
CYP2D6: Participates in the metabolism of the arginine side chain, generating nitrosylated or demethylated products.
Peptidase system:
Amino peptidase: Removes amino acids step by step from the N-terminal to generate short peptide fragments.
Carnitine peptidase: Hydrolyzes peptide bonds from the C-terminal to release free amino acids.
Fate of metabolites
Inactive fragment: Short peptides and amino acids lack the ability to bind to GnRH receptors and cannot exert the effect of stimulating gonadotropin release.
Potential toxicity: A very small number of metabolites (such as oxidized tryptophan) may damage liver cells through oxidative stress, but no serious adverse reactions have been reported in clinical practice.
Breaking through the first-pass effect: Innovative directions in formulation technology




1. Injection formulation: The mainstream choice in clinical practice
Intravenous injection: Directly enters the systemic circulation, with a bioavailability of 100%, but requires strict aseptic operation.
Subcutaneous/muscular injection: Slow but stable absorption, suitable for long-term treatment (such as for the treatment of precocious puberty and infertility).
2. New drug delivery systems: Exploratory solutions under development
Nanoparticle carriers: Gonadorelin is encapsulated by poly(lactic-co-glycolic acid) (PLGA) to protect it from enzymatic degradation, but the drug loading capacity is low (typically <10%) and the release controllability is poor.
Transdermal patches: Micro-needle arrays are used to penetrate the stratum corneum, but the molecular weight of peptide drugs (>500Da) is too large, resulting in extremely low transdermal efficiency (<1%).
Oral mucosal administration: Sublingual administration can bypass the liver, but the bitterness and irritation of Gonadorelin limit patient compliance.
3. Chemical modification strategies
Cyclization modification: Cyclize linear peptides (such as cyclogonadorelin) to enhance resistance to enzymatic degradation, but this may reduce receptor binding activity.
Non-natural amino acid substitution: Replace L-type amino acids with D-type amino acids to slow down the rate of enzymatic hydrolysis, but the pharmacokinetic properties need to be re-evaluated.


The stability of Gonadorelin pills, a synthetic polypeptide drug, is critical to ensuring its therapeutic efficacy and safety, as Gonadorelin is prone to degradation due to its peptide structure. Its stability is mainly affected by internal factors such as chemical structure and external factors including storage conditions, formulation excipients, and preparation processes. Gonadorelin is extremely hygroscopic, so moisture absorption during storage can easily cause peptide chain hydrolysis and reduce drug activity. Additionally, extreme pH values and high temperatures accelerate its degradation, with the most stable state achieved at pH 5.0-5.5.

To maintain stability, the product adopt optimized formulation and packaging designs. Excipients such as mannitol are added as protective agents to form a glassy matrix that wraps peptide molecules and inhibits degradation. The pills are packaged in tight, well-sealed containers to prevent moisture and oxygen exposure, and brown packaging can avoid ultraviolet-induced oxidation. Under standard storage conditions (2-8℃, dry and dark environment), the pills can maintain stable efficacy for 12-24 months, with a degradation rate controlled below 1%.

Notably, repeated temperature fluctuations and moisture exposure should be avoided during storage and transportation, as they can increase the content of degradation products and reduce bioactivity. Accelerated stability tests show that the pills remain stable under 40℃ and 75% relative humidity for 6 months, meeting pharmacopeial standards. Proper stability control ensures that the product retain their ability to regulate the hypothalamic-pituitary-gonadal axis during clinical use.
FAQ
Is gonadorelin safe for long-term use?
+
-
Some patients may experience palpitations or an increase in blood pressure. These symptoms should be reported to a healthcare provider, especially if they persist or worsen. Long-term use of Gonadorelin Acetate has not been extensively studied, so the long-term side effects are not well understood.
Does gonadorelin restore testicle size?
+
-
Patients on testosterone who wish to stem or reverse testicular shrinkage may use either gonadorelin or low-dose hCG. Gonadorelin may come with fewer estrogenic effects compared to hCG and is generally considered the drug of choice in younger patients with fertility aspirations.
Hot Tags: gonadorelin pills, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale
















