Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of hmg tablets in China. Welcome to wholesale bulk high quality hmg tablets for sale here from our factory. Good service and reasonable price are available.
HMG Tablets contain two hormones, follicle stimulating hormone (FSH) and luteinizing hormone (LH), typically extracted from the urine of postmenopausal women. In assisted reproductive technologies such as in vitro fertilization (IVF), HMG is commonly used in the early stages of the ovulation cycle to stimulate the development of multiple follicles and increase the number of available eggs. For example, data from 2020 showed that about 2% of infertility treatments used HMG, which is also suitable for oligospermia, asthenozoospermia, or azoospermia caused by hypogonadotropic hypogonadism, by stimulating the development of testicular seminiferous tubules and promoting sperm production. In addition, HMG usually contains statins (such as rosuvastatin) and other ingredients, which reduce cholesterol synthesis by inhibiting the activity of HMG CoA reductase in the liver.
At the same time, our company not only provides pure powders, but also tablets and injections. If needed, please feel free to contact us at any time.
Our products



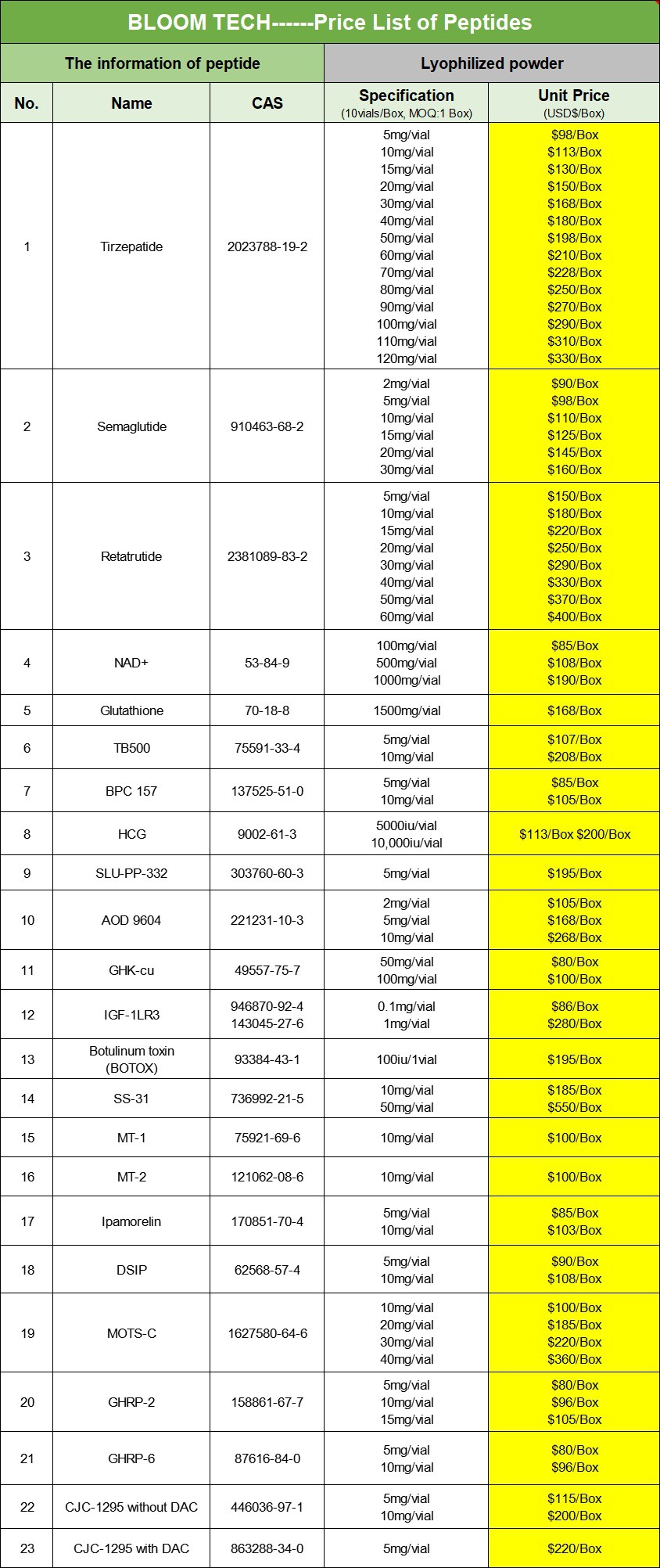
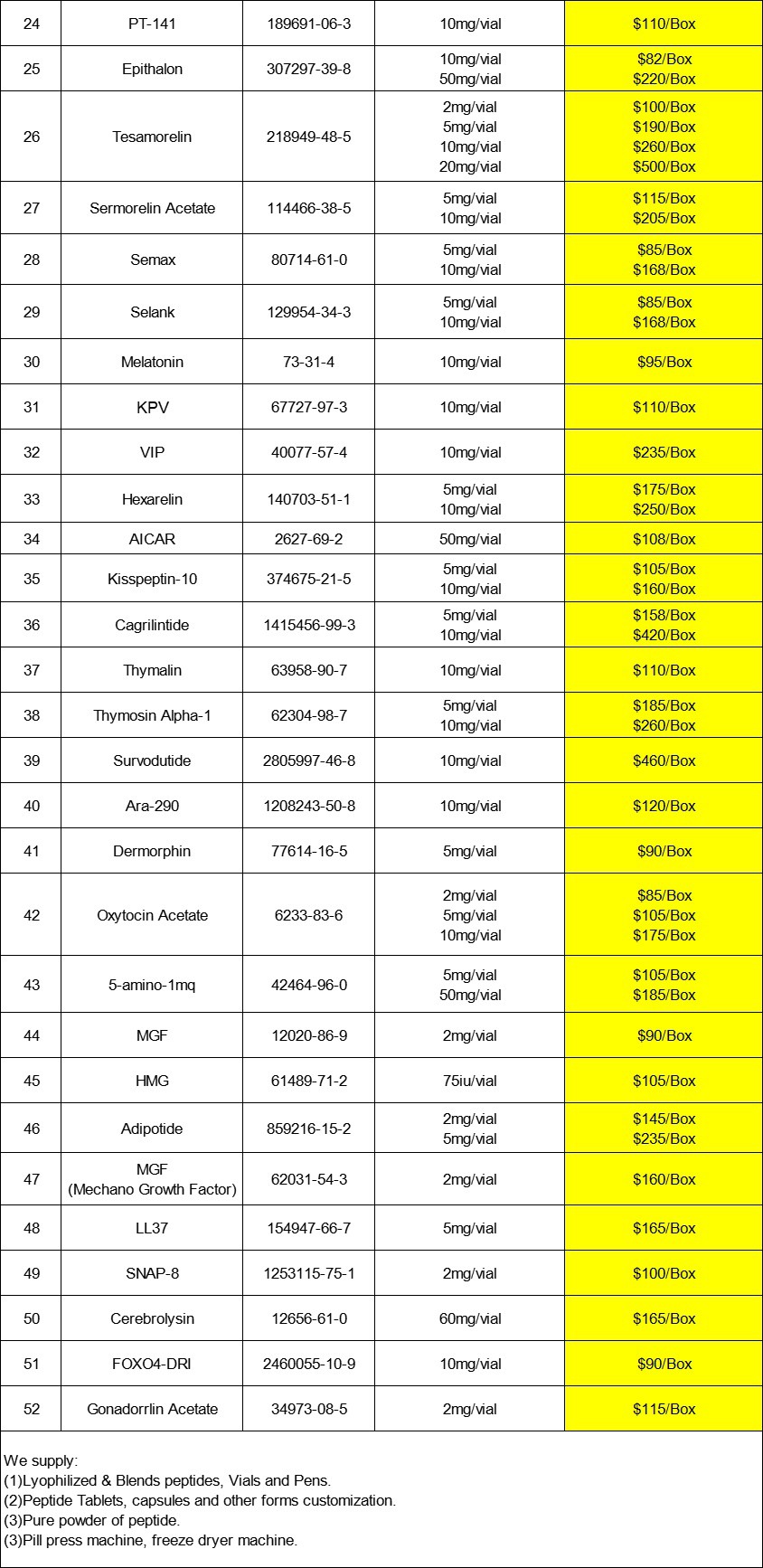
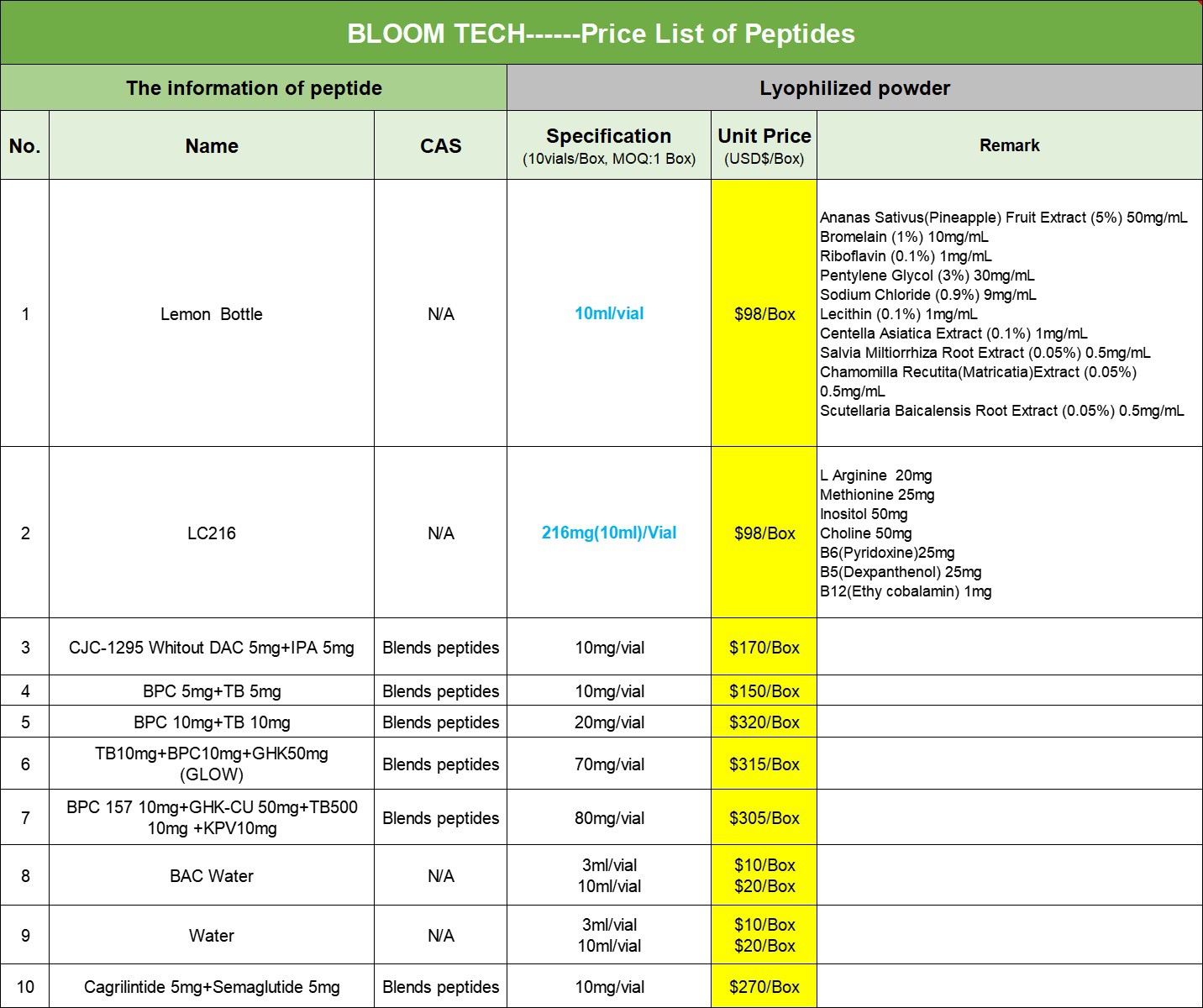

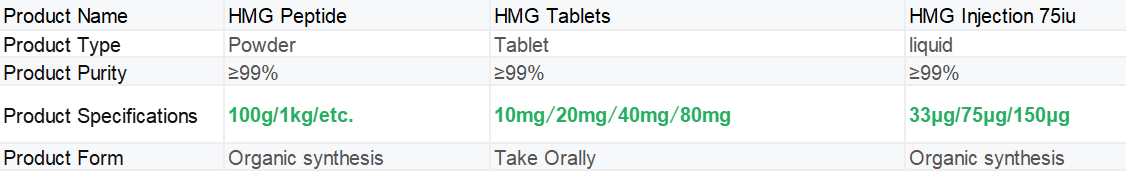
 |
 |
HMG COA
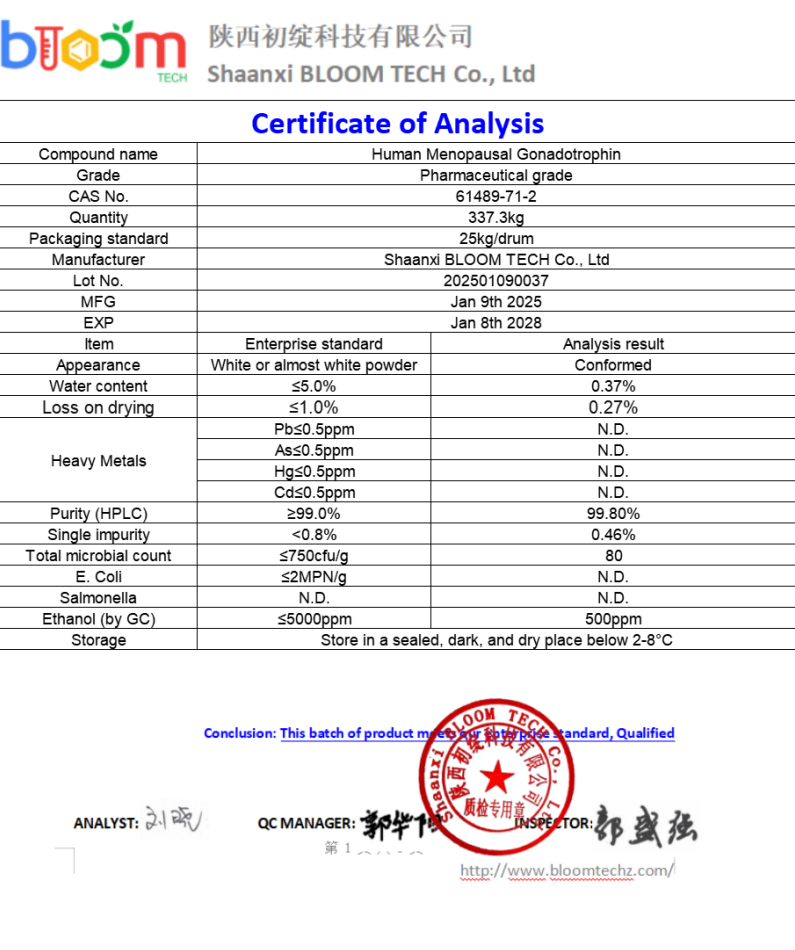

HMG (Human Menopausal Gonadotropin) is a hormone mixture extracted from the urine of postmenopausal women, mainly containing follicle stimulating hormone (FSH) and luteinizing hormone (LH), with a small amount of human chorionic gonadotropin (HCG). HMG Tablets, as an important drug for treating fertility disorders, involve complex biological extraction, purification, formulation processes, and strict quality control in their manufacturing process.
Raw material sources and pretreatment

Source of raw materials
The main ingredient of HMG tablet is the urine of postmenopausal women. These urine samples need to undergo strict screening to ensure the donor's health, absence of infectious diseases, and compliance with relevant regulatory requirements. The collection of urine is usually carried out through specialized collection stations to ensure the hygiene and quality of the raw materials.
Raw material pretreatment
The collected urine needs to undergo preliminary treatment to remove impurities and potential contaminants. The preprocessing steps may include:
Filtering: Removing large particulate impurities from urine through physical filtration.
Centrifuge: Using centrifugal force to separate solid and liquid components in urine, further purifying urine.
Adjust pH value: Adjust the pH of urine according to the needs of subsequent extraction processes.

Biological extraction and purification
Extraction process
The extraction of HMG usually uses biological separation technologies such as ion exchange chromatography and gel filtration chromatography. These technologies can separate HMG from urine based on the characteristics of protein charge, molecular size, etc.
Ion exchange chromatography: using ion exchange resin to adsorb and desorb proteins in urine, achieving preliminary separation of HMG.
Gel filtration chromatography: HMG is further purified by separating the pore size of gel particles according to the different size of protein molecules.


Purification steps
The crude HMG extracted needs to undergo multiple purification steps to improve its purity and activity. The purification steps may include:
Ultrafiltration: using ultrafiltration membrane to concentrate and desalinate HMG solution, removing small molecule impurities.
Affinity chromatography: Utilizing the affinity between HMG and specific ligands to achieve efficient purification of HMG.
High performance liquid chromatography (HPLC): As the final purification step, HPLC can further remove impurities and improve the purity of HMG.
Purity and activity assay
During the purification process, real-time monitoring of the purity and activity of HMG is required. Common detection methods include:
Electrophoretic analysis: such as SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis), used to detect the molecular weight and purity of HMG.
Biological activity assay: Determine the ability of HMG to stimulate follicular development and corpus luteum formation through in vitro cell experiments or animal experiments.

preparation technology
Accessory selection
The manufacturing of HMG Tablets requires the addition of various excipients to improve the stability, solubility, and taste of the drug. Common auxiliary materials include:
Fillers
Such as lactose, microcrystalline cellulose, etc., used to increase the volume and weight of tablets.
Adhesive
Such as hydroxypropyl methylcellulose (HPMC), polyvinylpyrrolidone (PVP), etc., used to bond drugs and excipients together to form a uniform mixture.
Lubricants
Such as magnesium stearate, talcum powder, etc., used to reduce friction between tablets and molds, facilitating compression.
Disinfectant
Such as cross-linked carboxymethyl cellulose sodium (CCNa), carboxymethyl starch sodium (CMS Na), etc., used to promote the disintegration and drug release of tablets in the gastrointestinal tract.
Formulation formula design
The design of formulation should consider the stability, solubility, taste, and patient convenience of the drug. Taking a certain HMG tablet as an example, its formulation may be as follows:
| Ingredient | Dosage (mg/tablet) | Effect |
| HMG | 75IU (calculated based on FSH and LH activity) | Main drug, stimulates follicular development and corpus luteum formation |
| lactose | 100 | Filler, increase tablet volume |
| HPMC | 10 | Adhesive, bonding drugs and excipients together |
| Magnesium stearate | 2 | Lubricant reduces friction between tablets and molds |
| CCNa | 5 | Disinfectant, promotes the disintegration of tablets in the gastrointestinal tract |
Manufacturing process

1
Ingredients and Mixing
Accurately weigh various raw materials and excipients according to the formulation formula, and mix them thoroughly in the mixer to ensure even distribution of the drug and excipients.

2
Granulation
Add the mixed powder to an appropriate amount of adhesive solution to make soft materials. Then, the soft material is made into granules through a granulator to improve the flowability and compressibility of the drug.

3
Drying
Place wet particles in a drying oven to dry, remove moisture, and improve the stability of the particles.

4
Granulation and mixing
The dried particles are granulated by a granulator to remove oversized or undersized particles. Then mix again to ensure the uniformity of the particles.

5
Tablet pressing
Put the mixed granules into a tablet press to make tablets with a certain shape and hardness.

6
Coating (optional)
Tablets can be coated as needed to improve the taste, stability, and appearance of the drug. Coating materials may include sugar coating, thin film coating, etc.
quality control
Raw material quality control
Strictly control the quality of raw urine and extracted HMG crude products to ensure they meet relevant standards. Quality control projects may include:
Microbial limit test:
Ensure that the raw materials are free from microbial contamination.
Heavy metal detection:
Detect the content of heavy metals in raw materials to ensure compliance with safety standards.
Purity and activity detection:
As mentioned earlier, the purity and activity of HMG are detected through methods such as electrophoresis analysis and biological activity determination.
Intermediate quality control
During the manufacturing process, quality control is carried out on intermediates (such as extraction solutions, purification solutions, particles, etc.) to ensure the stability and reliability of each step of the process. Quality control projects may include:
Content determination:
Detect the content of HMG in the intermediate to ensure compliance with the formulation requirements.
Impurity detection:
Detect the impurity content in intermediates, such as protein impurities, endotoxins, etc.
Particle size distribution:
For intermediate particles, detect their particle size distribution to ensure compliance with tablet requirements.
Finished product quality control
Conduct comprehensive quality control on finished HMG tablet to ensure compliance with relevant standards and regulations. Quality control projects may include:
Appearance inspection:
Check the shape, color, surface smoothness, etc. of the tablet.
01
Weight difference inspection:
Detect the weight difference of tablets to ensure compliance with pharmacopoeia regulations.
02
Content uniformity check:
Check the uniformity of HMG content in tablets to ensure that the content of each drug is consistent.
03
Dissolution test:
Detect the dissolution of tablets in a specified medium to ensure rapid release and absorption of the drug.
04
Stability study:
Conduct long-term stability research on finished products, test their stability under different conditions, and provide a basis for drug storage and expiration date.
05
Quality standards and regulatory compliance
The manufacturing of HMG must comply with relevant quality standards and regulatory requirements, such as the Chinese Pharmacopoeia, the United States Pharmacopoeia, and relevant guidelines of the International Organization for the Coordination of Drug Registration (ICH). These standards and regulations provide detailed requirements for the raw materials, manufacturing processes, quality control, and other aspects of drugs to ensure their safety and effectiveness.
Packaging And Storage
Selection of packaging materials
The packaging material of HMG Tablets needs to have good sealing, moisture resistance, and light avoidance to protect the drugs from external environmental influences. Common packaging materials include:
Bubble wrap packaging:
Place the tablet in a bubble wrap and seal it with aluminum foil, which has good sealing and moisture resistance.
Bottled:
Put the tablets into a glass or plastic bottle and seal it with a bottle cap. Bottled packaging should be equipped with desiccants to absorb the moisture inside the bottle.
Packaging process
The packaging process needs to ensure the integrity and sterility of the tablets. During the packaging process, it is necessary to clean and disinfect the packaging materials to avoid microbial contamination. At the same time, strict quality control is required for the packaging process to ensure that each drug is packaged correctly.
Storage conditions
HMG Tablet should be stored in a dry, cool, and dark place to avoid the effects of high temperature and humidity on the medication. The storage temperature is usually controlled between 2-8 ℃ to ensure the stability of the drug. At the same time, regular monitoring of the storage environment is required to ensure compliance with storage requirements.
Hot Tags: hmg tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale