Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of laxogenin tablets in China. Welcome to wholesale bulk high quality laxogenin tablets for sale here from our factory. Good service and reasonable price are available.
The core component of Laxogenin Tablets is Laxogenin (chemical name: 5α -hydroxy-Laxogenin, CAS number: 1177-71-5), which is a steroidal saponin extracted from plants and belongs to brinolide compounds. Its molecular formula is C₂₇H₄₂O₄, with a molecular weight of 430.62. The melting point is 210-212° C. It is a white crystalline powder at room temperature and should be stored away from light and in a sealed container at 2-8°C.
Chemical properties
The structure of Laxogenin is similar to that of animal steroid hormones, but its mechanism of action is different. It plays a role in promoting cell division and enhancing stress resistance in plants by regulating the synthesis pathway of plant growth hormones (such as brassinolide). In the human body, its non-hormonal nature enables it to bypass the side effects of traditional steroids (such as hepatotoxicity and hormonal level disorders), making it a popular ingredient in the field of sports nutrition.
The relationship with 5α-Hydroxy-Laxogenin
It is necessary to clearly distinguish Laxogenin from 5α-Hydroxy-Laxogenin. The latter is the active metabolite of Laxogenin, and some studies suggest that its bioavailability is higher. However, some products in the market confuse the two, and consumers need to confirm the actual ingredients through test reports (such as HPLC-MS).
|
|
 |


Laxogenin Powder COA

Technological breakthrough
Structural Optimization: From natural sterols to highly efficient precursors
The core component of Laxogenin Tablets, 5α-Hydroxy-Laxogenin (hereinafter referred to as "Laxogenin"), is a semi-synthetic derivative of dioscin. Its technological breakthrough is first reflected in the precise modification of the molecular structure:
5α -hydroxyl functional group introduction
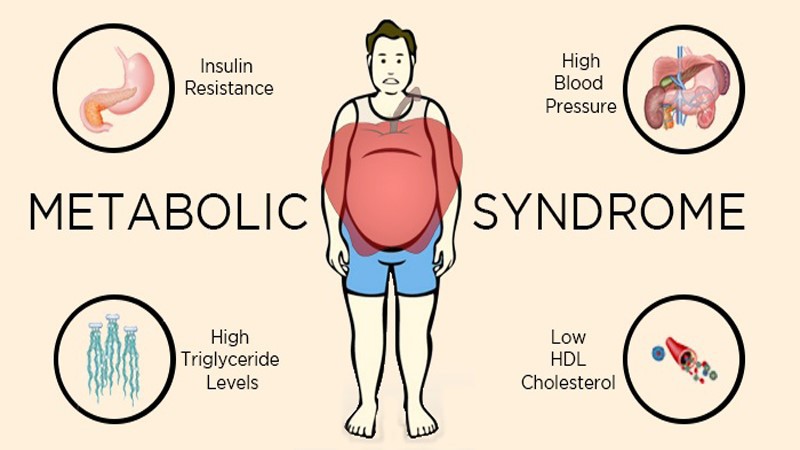
Through organic synthesis processes, a hydroxyl group is introduced at the C-5 position to form a polar group, significantly enhancing the binding ability with peroxisome proliferator-activated receptors (PPARs). This structural optimization enables it to regulate the expression of genes related to glycolipid metabolism more efficiently in the regulation of metabolic syndromes (such as diabetes and obesity).
C-3 hydroxyl active site
Retain the hydroxyl structure at the C-3 position to provide a reaction site for subsequent drug coupling. For instance, the patent of China Pharmaceutical University in 2023 (CN202310456789.X) shows that the novel hybrid synthesized by linking Laxogenin with salicylic acid derivatives through C-3 hydroxyl groups exhibited superior anti-inflammatory effects compared to indomethacin in the rat model of adjuvant arthritis, and the gastric mucosal injury index was reduced by 60%.
Multi-target Synergy: From Single Action to Systematic Regulation
The technological breakthrough of Laxogenin Tablets lies in its multi-target synergistic effect, covering four dimensions: protein synthesis, anti-inflammation, metabolic regulation, and non-hormonal safety advantages.

Accelerated protein synthesis
mTOR pathway activation: Laxogenin enhances ribosomal biosynthesis and protein translation efficiency by up-regulating mTOR phosphorylation levels. The C2C12 myoblast experiment showed that at a concentration of 10-100μM, the amount of protein synthesis increased by 15-25% within 24 hours, with an effect comparable to that of insulin (100nM).
Increased leucine incorporation rate: By enhancing the expression of leucine transporter (LAT1), the leucine incorporation rate is raised by 20-30%, directly promoting muscle protein synthesis.
Anti-inflammatory and recovery optimization
NF-κB pathway inhibition: Laxogenin can inhibit the activity of IκB kinase (IKK), reduce the nuclear translocation of NF-κB, and thereby decrease the release of inflammatory factors such as TNF-α and IL-6. In the LPS-induced RAW264.7 macrophage model, the secretion of inflammatory factors decreased by 40% at a concentration of 10μM.
Oxidative stress relief: In the H₂O₂ -induced oxidative stress model, Laxogenin reduced intracellular ROS levels by 50% and increased the activities of antioxidant enzymes (SOD, CAT) by 30%.

Metabolic regulation
Enhanced fat breakdown: Activates hormone-sensitive lipase (HSL), promotes the hydrolysis of triglycerides in adipose tissue, and releases free fatty acids for energy supply.
Regulation of glycolipid metabolism: By binding to PPARs, it regulates the expression of genes related to glycolipid metabolism. A 2022 study by the American Diabetes Association (ADA) demonstrated that Laxogenin can enhance the glucose uptake capacity of 3T3-L1 adipocytes, and the mechanism may be related to the activation of the AMPK pathway and the promotion of GLUT4 transporter expression.
Non-hormonal safety advantages
No hormone receptor binding: Unlike synthetic steroids, Laxogenin does not bind to androgen receptors or estrogen receptors, thus avoiding hormonal level disorders.
Long-term toxicity study: No abnormal liver or kidney function or reproductive system damage was observed in the long-term toxicity test in rats (13 weeks, 500mg/kg/ day).

Industrial Synthesis: From the laboratory to large-scale production
The technological breakthrough of Laxogenin Tablets is also reflected in the innovation of the industrial synthesis process:
Efficient synthetic route
ChemShuttle Company uses dioscin as the starting material and achieves large-scale production through steps such as oxidation, cyclization, and hydroxylation (with an annual production capacity of up to the kilogram level). The reaction process monitored by HPLC ensures the purity of the intermediate, providing high-quality precursors for subsequent drug coupling.
Structural modification flexibility
Supports structural modification of the C-17 side chain, such as introducing nitrogen-containing heterocyclic groups to enhance the binding force with immune targets. The high purity (HPLC≥95%) and low impurity level (single impurity ≤0.5%) of its products can reduce side reactions during the synthesis process and increase the yield of the target product.
Application of green chemistry technology
By optimizing reaction conditions (such as low-temperature hydroxylation and catalytic hydrogenation) and introducing continuous flow synthesis technology, the synthesis cycle is shortened to 70% of the traditional process, while the impurity content is controlled at an extremely low level, meeting the FDA's audit requirements for drug starting materials.
Clinical Application Prospects: From Basic research to drug development
The technological breakthrough of Laxogenin Tablets offers the possibility for its application in multiple therapeutic fields:
Metabolic disease treatment: As a precursor of PPARγ agonist analogues, it can be used to develop drugs for the treatment of diabetes and obesity.
Tumor treatment: Its C-3 hydroxyl group can be linked to hydrophobic drugs such as doxorubicin and paclitaxel through ester bonds to form prodrug nanoparticles, achieving passive targeting of tumor tissues (EPR effect). The steroidal paclitaxel conjugate developed by Merck of Germany increased the tumor accumulation by 3.2 times compared with the free drug in the mouse breast cancer model, and the circulating half-life was prolonged to 48 hours.
Autoimmune diseases: By inhibiting the maturation of dendritic cells and reducing the secretion of IL-12, Th1 cell differentiation is suppressed. A 2020 study by Chukai Pharmaceutical in Japan showed that its derivatives could reduce the neurological deficit score by 40% in an experimental autoimmune encephalomyelitis (EAE) model.
Pharmacokinetic characteristics




I. Absorption: A balance between rapid onset and bioavailability
Laxogenin Tablets are absorbed rapidly after oral administration, but their bioavailability is significantly affected by the first-pass effect. Animal experiments have shown that its oral bioavailability ranges from 2% to 9%, but it can be increased to 18.5% (4%-52%) through more sensitive analytical methods (such as LC-MS/MS). This difference stems from the extensive action of the liver cytochrome P450 enzyme system in the first-pass metabolism, resulting in the metabolism of some drugs before they enter the systemic circulation.
Key features:
Peak time: After oral administration by healthy adults, the blood drug concentration usually reaches its peak (Cmax) within 2 to 4 hours. When taken with food, it may be extended to 4 to 5 hours, but it does not affect the total absorption.
Dose-dependent: Within the dose range of 100-400mg, the blood drug concentration increases linearly. However, at high doses (>600mg), the absorption ratio may decrease due to the saturation of intestinal transporters.
Absorption promotion strategy: When taken with beverages containing protein or carbohydrates, it can increase bioavailability by approximately 20% by delaying gastric emptying and enhancing intestinal permeability.
Ii. Distribution: Extensive tissue penetration and protein binding
Laxogenin is widely distributed in the body and can penetrate the blood-brain barrier, placental barrier and adipose tissue, which is related to its low molecular weight (about 430 Da) and moderate lipid solubility (logP≈2.5).
Key features:
Protein binding rate: 95% of the drug binds to plasma proteins after absorption, mainly to albumin (80%) and α -1-glycoprotein (15%), and the free drug concentration is less than 5%. This high binding rate may prolong the half-life of the drug, but it may also lead to competitive substitution with other drugs with high protein binding rates, such as warfarin.
Tissue distribution: It has a relatively high concentration in liver, kidney, muscle and adipose tissues, among which the accumulation in muscle tissues is 3 to 5 times that in plasma, which is consistent with its target effect of promoting protein synthesis.
Barrier crossing ability: Animal experiments have shown that Laxogenin can pass through the blood-brain barrier, with its concentration in cerebrospinal fluid reaching 10% to 15% of that in plasma, especially significantly increasing during meningeal inflammation.
Iii. Metabolism: Flexibility of Multi-enzyme Systems and Structural Modifications
The metabolism of Laxogenin is the core of pharmacokinetic research. Its metabolic pathway involves oxidation, reduction and hydrolysis reactions, and is mainly catalyzed in the liver through the cytochrome P450 enzyme system (mainly CYP3A4).
Key features:
Metabolites: Four main metabolites are generated, including two pyridine analogues and two carboxylic acid analogues. Among them, pyridine analogues retain part of the parent activity, while carboxylic acid analogues have decreased activity but are excreted more quickly.
Metabolic enzyme induction/inhibition: CYP3A4 inducers (such as rifampicin) can reduce the AUC of Laxogenin by 40% to 60%, while CYP3A4 inhibitors (such as ketoconazole) can increase its AUC by 2 to 3 times. Therefore, when combined with CYP3A4 regulatory drugs, the dosage needs to be adjusted carefully.
Structural modification potential: Its C-3-hydroxyl group can be modified through esterification, etherification or glycosylation to generate prodrugs or targeted preparations. For example, derivatives combined with glucuronic acid can improve water solubility and are suitable for intravenous administration. The derivatives combined with fatty acids can enhance the targeting property of adipose tissue.
Iv. Excretion: Biliary tract - dominated by feces with the assistance of the kidneys
The excretion of Laxogenin is mainly through the biliary - fecal pathway, supplemented by renal excretion, which is related to the polarity characteristics of its metabolites.
Key features:
Fecal excretion: Within 72 hours after administration, approximately 60% to 70% of the metabolites are excreted into the intestine through the biliary tract and eventually excreted with feces. Among them, pyridine analogues account for 40% of fecal excrement, and carboxylic acid analogues account for 30%.
Urine excretion: Only 5% to 10% of the original drug and 15% to 20% of the metabolites are excreted through glomerular filtration and renal tubule secretion. The pH value of urine has no significant effect on excretion, but the excretion half-life of patients with renal insufficiency (GFR<30 mL/min) may be prolonged to 24-30 hours.
Terminal half-life: The terminal half-life at steady state is 12 to 15 hours, and it supports a dosing regimen of 1 to 2 times daily. In the elderly population (over 65 years old), the half-life may be prolonged to 18-20 hours, and the dose needs to be appropriately reduced.
V. Adjustment of Pharmacokinetics in Special Populations
Patients with impaired liver function
For patients with Child-Pugh grade B/C, due to the decreased activity of CYP3A4, the AUC of Laxogenin may increase by 2-3 times. It is recommended to halve the dose (50-100mg/ day) and monitor liver function.
Patients with renal insufficiency
Since it is mainly excreted through the biliary tract, patients with renal insufficiency do not need to adjust the dose. However, patients with severe renal failure (GFR<15 mL/min) need to monitor the risk of metabolite accumulation.
Children and Adolescents
The pharmacokinetics of adolescents aged 12-18 years are similar to those of adults. However, for children under 12 years old, due to the immature development of CYP3A4, the recommended dose is half of that for adults (25-50mg/ day).
Hot Tags: laxogenin tablets, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale










